A. Menarini Farmaceutica Internazionale SRL has provided an educational grant for the production of this e-learning programme and has had no editorial control or input. The views and content expressed within this programme are solely those of the authors.
PP-CA-UK-0155 Date of preparation: March 2020
Introduction
Imaging – with computerised tomography coronary angiography (CTCA) – has become a recommended first-line investigation by the National Institute for Health and Care Excellence (NICE) for patients with new onset chest pain.1 This was one of several new recommendation in the 2016 update to the 2010 guidance.
This earlier 2010 NICE Guidance2 recommended that based on age, the type of chest pain, and the presence or absence of risk factors, a pre-test probability (PTP) risk score (RS), could be used to identify the investigation of choice and whether an individual is at low or high risk of coronary artery disease (CAD).
It also recommended that findings from the clinical assessment and the resting 12-lead electrocardiogram (ECG) are taken into account when making the estimate, and further diagnostic testing was advised as follows:
- Patients with an RS <10% do not warrant cardiac investigation
- If the estimated likelihood of CAD is 10–29%, offer computed tomography (CT) calcium scoring as the first-line diagnostic investigation
- If the estimated likelihood of CAD is 30–60%, offer functional imaging as the first-line diagnostic investigation e.g. myocardial perfusion scintigraphy (MPS), magnetic resonance imaging (MRI), stress echocardiography
- If the estimated likelihood of CAD is 61–90%, offer invasive coronary angiography as the first-line diagnostic investigation if appropriate.
A detailed critique of the 2016 NICE update has been covered by Alfakih K et al.3 Briefly there are three key changes to the 2010 Guidance:
- First is the recommendation for a clinical assessment of the likelihood of CAD, based on the typicality of the chest pain into typical, atypical or non-cardiac, instead of the previous PTP RS.
- Second, a zero calcium score is no longer used to rule out CAD in patients with low PTP.
- Thirdly, all patients with new onset chest pain with atypical or typical anginal features, as well as those with noncardiac chest pain and an abnormal resting ECG, should first be investigated with CTCA using a 64-slice (or above) CT scanner.
Functional imaging tests are now reserved for the assessment of patients with chest pain symptoms who are known to have CAD and for patients where the CTCA has been non-diagnostic or has shown CAD of uncertain significance.
When offering non-invasive functional imaging for myocardial ischaemia NICE recommends to use:
- MPS with single-photon emission computerized tomography (SPECT) or
- stress echocardiography or
- first-pass contrast-enhanced magnetic resonance perfusion or
- MRI for stress-induced wall motion abnormalities.
When deciding on the imaging method, this should take account of the locally available technology and expertise, the person and their preferences, and any contraindications (for example, disabilities, frailty, limited ability to exercise).
The 2016 update recommends that the following investigations should not be used:
- magnetic resonance (MR) coronary angiography for diagnosing stable angina.
- exercise ECG to diagnose or exclude stable angina for people without known CAD.
This module describes imaging techniques in more detail.
Calcium and coronary artery imaging by computed tomography
In the assessment of patients with symptoms of obstructive CAD, coronary calcium imaging with CT is indicated after functional tests are either not possible or inconclusive. Current NICE guidelines,2 however, recommend coronary calcium imaging as a first-line test in symptomatic patients with a low (<30%) pre-test probability of CAD. Calcium deposits within the coronary arterial wall correlate with the presence of atherosclerosis and hence represent a highly sensitive marker for CAD.
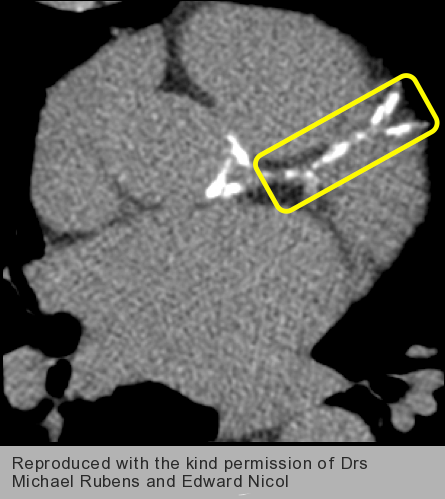
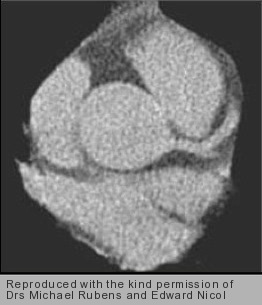
Coronary calcification can be quantified using non-enhanced CT imaging (figure 1). Calcium causes hyperattenuation of X-rays; pixels with a density above a pre-defined threshold of 130 Hounsfield Units (HU) are assumed to represent calcium deposits (figure 2). Areas within the coronary arteries with >130HU are selected and added up to estimate the individual’s coronary calcium score (CCS) or Agatston Score.
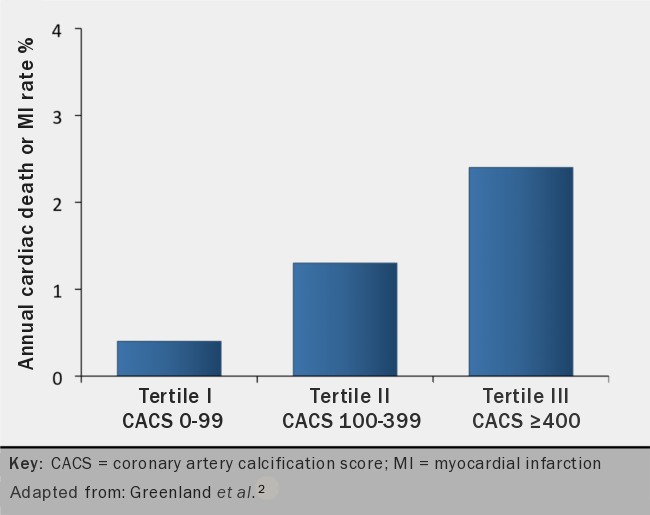
Coronary calcification not only reveals the presence of atherosclerosis but also predicts a worse outcome. It is possible to risk-stratify patients according to the total amount of calcium present in their coronary arteries. Hence, patients with a CCS of zero are deemed at very low risk of cardiac events (<1% risk of myocardial infarction [MI] or cardiac death/year; figure 1) while patients with CCS ≥1 have a higher risk. As the calcium score increases so does the risk of future adverse events (figure 3).4
Patients with CCS ≥1 are considered to have CAD and hence further assessment is recommended. NICE advocates the use of CT angiography (CTA) as second-line test in patients with CCS ≥1 but ≤400. CTA allows visualisation of the coronary vasculature and detection of coronary luminal narrowing with the use of contrast-enhanced imaging (see below). Patients with CCS >400 should be considered for functional imaging (discussed later) or invasive X-ray coronary angiography.
CTA offers the best resolution to date and hence it is the preferred imaging technique for the non-invasive assessment of the coronary arteries. CTA is performed by injecting an intravenous iodine-based contrast agent to enhance visualisation of the coronary arterial wall. Most studies are currently performed using multi-detector CT systems.
Image acquisition is synchronised to the patient’s ECG and images obtained during a pre-selected phase of the cardiac cycle (prospective gating) or throughout the cardiac cycle and then retrospectively aligned to the ECG tracing (retrospective gating). The former method reduces radiation exposure. The acquired data is reconstructed into a stack of transaxial slices that can be processed further using different post-processing methods (figure 4).
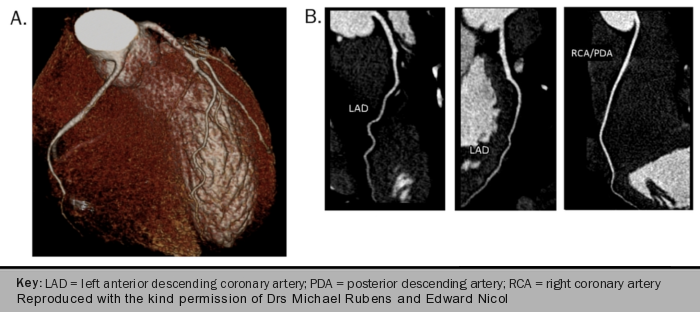
CTA allows plaque characterisation by means of differences in X-ray attenuation caused by the various components of the atherosclerotic plaque. This provides an accurate estimation of the site, extent and severity of underlying coronary disease (figure 5A–C).
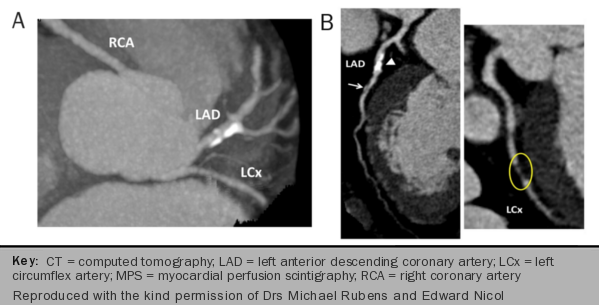
A thorough review of the images may include:
- a brief reference to the anatomical arrangement of the vessels and a more detailed description of the origin and course of the coronary arteries if clinically relevant
- the site, extent and severity of luminal narrowing (a binary cut-off value of 50% or 70% diameter stenosis is commonly used to define clinically important coronary stenosis) and
- individual plaque characterisation (e.g. calcified, soft or mixed plaque; eccentric or concentric).
Some reports might include a brief statement on the overall quality of the study, the presence of artefacts and their effect on image quality and interpretation.
Other indications for cardiac CT imaging
Cardiac CT imaging can be used for the following indications:
- as part of the workup of anomalous coronary arteries
- for evaluation of coronary artery bypass grafts
- for assessment of cardiovascular anatomy and morphology
- for assessment of left ventricular volumes and function
- as a roadmap for electrophysiological procedures such as catheter ablation.
Advantages and limitations of CT imaging
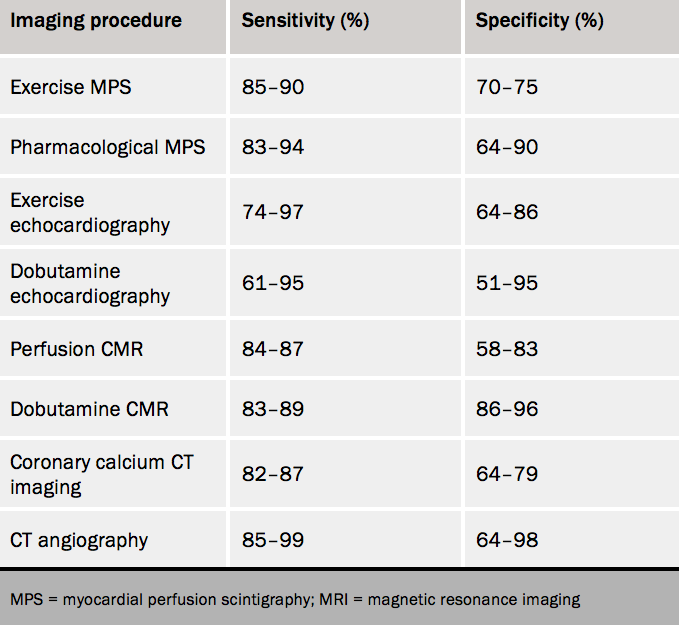
As for most diagnostic tests, CT imaging is validated against non-invasive X-ray coronary angiography. Coronary calcium imaging by CT has a high sensitivity for the detection of angiographically significant CAD (90–95%) as well as a high negative predictive value (>95%) but a low specificity for ruling out coronary obstruction (table 1).
In other words, the presence of calcium in the coronary arteries does not necessarily relate to the presence of flow-limiting stenosis. Although a calcium score of zero predicts a low probability of adverse cardiac events, it does not rule out CAD. Calcium imaging may underestimate the presence of CAD as soft (non-calcified) plaques may go undetected. This is particularly relevant to the symptomatic low-risk population since younger patients are more likely to have soft plaque stenosis than calcified lesions. Non-calcified atheroma can result in plaque rupture, acute thrombosis, MI and death.
CTA provides a comprehensive assessment of coronary artery stenosis including plaque composition and total plaque burden. It also provides unparalleled views of anatomical structures and permits a study of the relations of the coronary arteries to the other structures and tissues. CTA has a very high sensitivity and a high negative predictive value (>95%) for excluding CAD (table 1). Accuracy to predict obstructive CAD however is lower and this is partly due to difficulties in determining the true extent of luminal narrowing at any given site in the presence of calcification.
Large deposits of calcium may have significant partial volume effect and may cause blooming artefact, obscuring the true vessel lumen. Quality and interpretation of CT images can be affected by other sources of artefact including respiratory and patient motion during the acquisition, heart rhythm and heart rate variability, beam hardening, and ECG-triggering errors. Many of these can be prevented by adequate patient preparation and use of appropriate scan protocols. Meticulous review of the images using different post-processing methods also helps to distinguish artefacts from true abnormalities hence improving diagnostic performance.
One of the most important aspects to consider when requesting a CT scan is radiation exposure. Calcium imaging by CT exposes individuals to a relatively small amount of ionizing radiation (table 1). Higher radiation exposure is associated with CTA but this can be reduced significantly by optimising scan techniques and protocols. In this regard, attention should be paid to scanner design and imaging performance, which influence total dose administered to the patient.
Finally, administration of iodine-based contrast agents for the purpose of enhanced CT imaging may cause some adverse reactions including anaphylaxis and nephrotoxicity. Routine documentation of kidney function before contrast injection is recommended in all patients with renal disease as well as in those with diabetes. In patients with known renal impairment, possible benefits must be weighed against risks and consideration should be given to non-contrast CT imaging or other alternative imaging techniques.
Patients with chest pain and an intermediate probability of angiographically significant CAD
First-line diagnostic test: functional non-invasive imaging
Myocardial perfusion scintigraphy
MPS is a well-established technique for the diagnosis of obstructive CAD by means of the detection of an inducible myocardial perfusion abnormality or myocardial ischaemia. MPS provides functional rather than anatomical information on the significance of coronary stenosis, which assists clinical decision-making with regard to the need for optimal medical therapy and/or coronary revascularisation.
MPS is best performed in symptomatic patients with an intermediate pre-test probability of angiographically significant coronary disease according to current NICE recommendations.1 MPS helps characterise the extent and severity of disease and provides incremental prognostic information over clinical history, cardiovascular risk factors and exercise ECG results. It also acts as a cost-effective gatekeeper to coronary catheterisation by reducing the number of unnecessary angiograms.
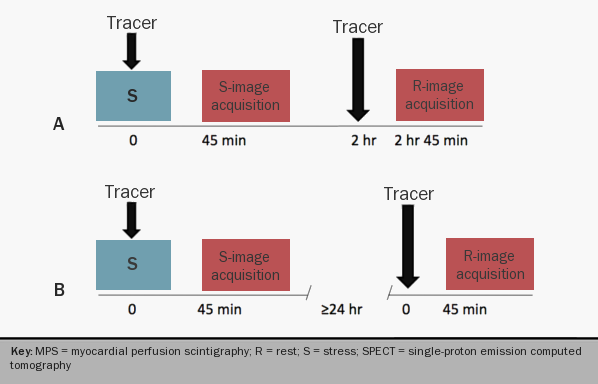
A conventional MPS study consists of an intravenous injection of a radiopharmaceutical or perfusion tracer (i.e. 201Tl, 99mTc-sestamibi, and 99mTc-tetrofosmin) at rest and at maximal or near-maximal coronary hyperaemia. The latter is achieved during cardiac stress with dynamic exercise or intravenous dobutamine, or after injection of a primary coronary vasodilator, such as dipyridamole, adenosine or regadenoson (figure 6).
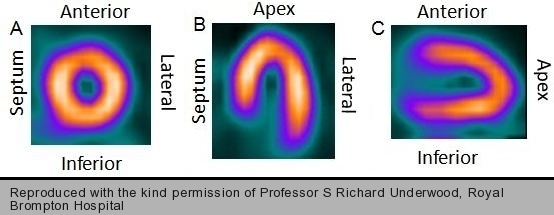
In most nuclear cardiology centres, distribution of tracer activity within the left ventricular myocardium is detected using single-photon emission computed tomographic (SPECT) technology. The raw cardiac data is reconstructed into a stack of transaxial tomograms that are realigned to obtain three standard views of the left ventricle (figure 7).


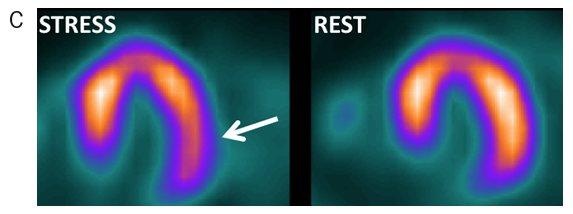
Differences in hyperaemic flow between normal and abnormal vascular territories due to coronary stenosis produce heterogeneous distribution of tracer activity in the myocardium on the stress images (figures 8 and 9). Side by side comparison with the resting tomograms provides information on the size and severity of inducible perfusion abnormality and hence on the extent of flow-limiting CAD (figures 8 and 9).
At present, most gamma-camera systems allow ECG-gating of the tomographic acquisition for simultaneous measurement of left ventricular volumes and ejection fraction, and assessment of regional myocardial wall motion and thickening (figure 10).
Indications for MPS
The following are indications for MPS:
- To assess the extent and severity of flow-limiting coronary stenosis in patients with an intermediate pre-test probability of CAD
- To assess myocardial viability and hibernation
- To determine the functional significance of non-atherosclerotic causes of myocardial ischaemia (e.g. anomalous coronary arteries, myocardial bridging)
- Initial assessment of patients with acute chest pain and nonspecific ECG changes and no history of CAD (this indication is most popular in the US)
- To aid management of patients with known CAD by guiding strategies of revascularisation and providing incremental information for risk stratification and prognostic assessment.

Advantages and limitations of MPS
MPS is not only a highly accurate test for the detection of obstructive CAD (sensitivity, 83–94%; specificity, 64–90%; table 1) but also a powerful prognostic tool. MPS helps stratify patients according to their likelihood of future coronary events. An abnormal scan predicts a high risk of events and risk increases in proportion to the extent and severity of inducible perfusion abnormality, while a normal MPS study predicts a low probability of events (<1%/year). MPS also represents a highly cost-effective strategy by directing invasive coronary angiography to patients who are more likely to benefit from coronary revascularisation.
MPS can be combined with all forms of stress currently available, and this may represent an important advantage over other functional imaging techniques. Visual assessment of tracer activity within the myocardium is the commonest approach to image interpretation in clinical practice. With the use of automated programmes, quantification of relative- rather than absolute-perfusion is also possible by comparing individual count profile to a normal database. This approach adds objectivity to image interpretation and enhances reproducibility of results, which is most useful in the assessment of serial studies.
Conventional MPS accurately identifies patients with disease in one or two of the three major coronary arteries but may be less effective at identifying patients with disease of similar severity in all three vascular territories. This is because the images are normalised to maximum tracer activity within the heart and hence balanced reduction in blood flow would appear normal. The presence of non-perfusion markers of extensive ischaemia such as transient ischaemic left ventricular dilation and increased lung tracer uptake raises suspicion of severe multivessel or left main stem disease.
To enhance diagnostic performance, electrocardiographic, symptomatic and haemodynamic changes during stress must complement image interpretation. MPS is vulnerable to count attenuation by extra-cardiac tissue. Indeed, image quality and interpretation can be affected by photon absorption, photon scatter and patient motion during the acquisition. It is now possible to minimise the degrading effect of these on the final tomograms by applying the appropriate reconstruction algorithms.
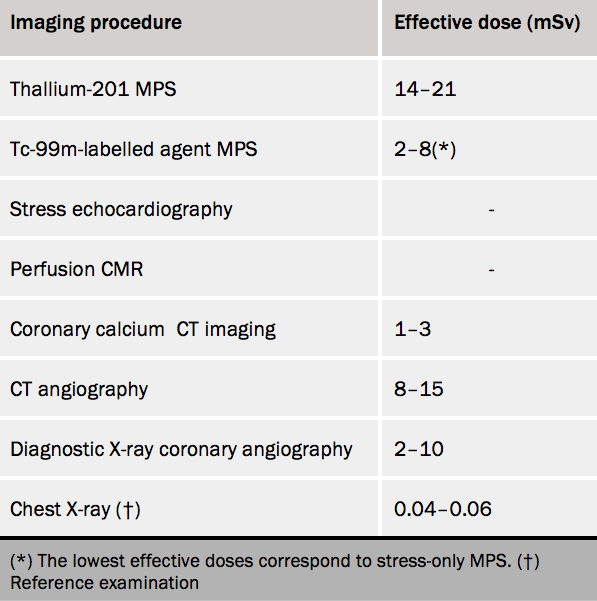
The most important factor to consider when indicating and performing an MPS study is exposure to ionising radiation. Radiation exposure varies from 6 to 21 mSv depending on patient characteristics, imaging protocol and type of radionuclide (table 2). At the doses currently administered in the UK, the individual risk of malignancy associated with conventional MPS is relatively small.
Moreover, when indicated appropriately, the incremental diagnostic and prognostic information provided by the study outweighs the risk associated with radiation exposure. Recent studies using new generation camera systems have shown that total radiation exposure can be reduced to as low as 3–4 mSv for a stress/rest ECG-gated MPS study. Radiation-free techniques such as MRI and echocardiography may be preferred over MPS in young subjects (see below).
A position paper from the European Society of Cardiology (ESC)5 aims to summarise knowledge on radiation effective doses (and risks) related to cardiac imaging procedures. It reviews the literature on radiation doses, ranging from the equivalent of 1–60 milliSievert (mSv) around a reference dose average of 15 mSv (corresponding to 750 chest X-rays) for a percutaneous coronary intervention, a cardiac radiofrequency ablation, a multidetector coronary angiography, or a myocardial perfusion imaging scintigraphy. It provides a European perspective on the best way to play an active role in implementing into clinical practice the key principle of radiation protection that “each patient should get the right imaging exam, at the right time, with the right radiation dose”.
Cardiovascular magnetic resonance imaging
Cardiovascular magnetic resonance imaging (CMR) is a high-resolution, cross-sectional imaging technique that combined with cardiac stress provides a comprehensive assessment of patients with suspected CAD in a single examination. As for MPS, perfusion CMR is best indicated in patients with an intermediate pre-test likelihood of CAD.
To obtain the corresponding images, the patient is positioned inside a superconducting magnet that generates a strong static magnetic field. Protons in the body align with the direction of the field and precess at the resonance frequency (e.g. 64 MHz for a 1.5 Tesla scanner). Images are produced by applying a pulse sequence, a combination of magnetic field gradients and radiofrequency pulses that excite the precessing protons and generate a radio signal as the protons relax and return to their former pre-excitation position.
New ‘open’ scanners are now available (pictured) which can help overcome some of the noise and claustrophobia sometimes associated with earlier scanners.
The quality and intensity of the signal varies according to inherent magnetic properties of the tissue. This difference in signal intensity allows delineation of cardiac anatomy, and enables the distinction between physiological and pathological changes. Most acquisitions are synchronised to the patient’s ECG to minimise artefact due to cardiac motion. In addition and whenever possible, breath-holding is performed to avoid the degrading effect of respiratory motion on the images.
For the purpose of perfusion imaging, a contrast media is injected intravenously at rest and at peak stress and images acquired during first-pass of contrast through the heart. The most commonly used contrast media are gadolinium chelates, such as gadolinium-DTPA. These are paramagnetic substances that once injected intravenously distribute throughout the intravascular and extracellular space and enhance signal intensity.
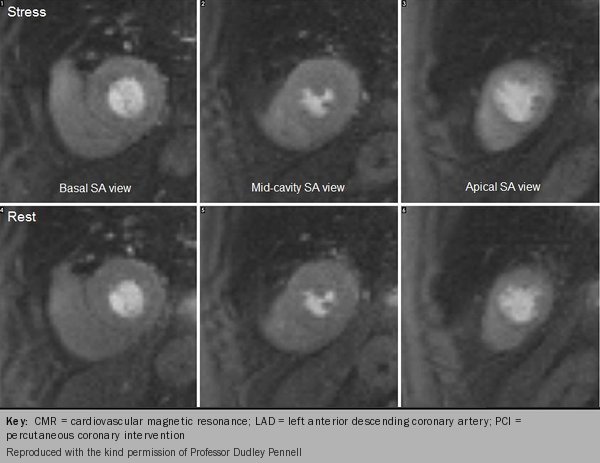
To stress the myocardial perfusion system, a continuous intravenous infusion of adenosine is administered for at least three minutes before injecting the contrast agent with the use of a power injector. The first-pass stress and rest images usually consist of three to four short-axis views equally spaced from base to apex and these may be accompanied by one or two long-axis views (figure 11). The stress and rest images are displayed side-by-side for visual assessment.
Myocardial regions supplied by a stenotic coronary artery will show delayed arrival of contrast to the myocardium resulting in regional hypo-enhancement, which will appear as areas of reduced signal intensity (figures 12-16).
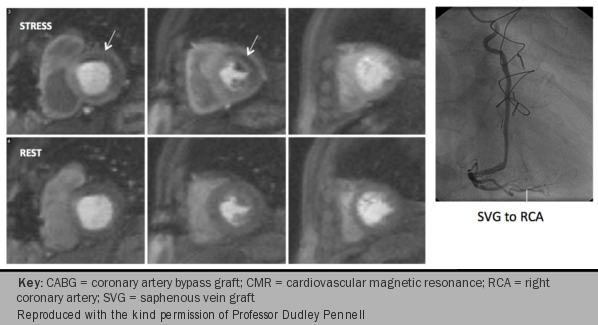
Delayed acquisition of contrast-enhanced images complements the assessment of perfusion by identifying areas of myocardial fibrosis and infarct (figures 17-19). CMR is superior to other imaging techniques for the detection of small subendocardial infarcts because of its higher spatial resolution.

In addition to myocardial perfusion and delayed-enhancement images, cine recordings of short-axis and long-axis views of the heart at rest are almost universally obtained for the assessment of ventricular volumes, global and regional myocardial function (figures 14, 18 and 19). Detection of ischaemia-induced wall motion abnormality and hence flow-limiting CAD is also possible by combining CMR with dobutamine stress (see dobutamine stress echocardiography).


Other indications for CMR
The following are indications for CMR:
- Initial evaluation and follow-up of patients with complex congenital heart disease
- Quantification and monitoring of ventricular function in patients with heart failure or suspected ventricular dysfunction, especially in cases of limited echocardiographic access, inconclusive or unexpected results
- As part of the diagnostic workup of cardiomyopathy and myocarditis
- Assessment of myocardial viability in patients with ischaemic left ventricular dysfunction
- Diagnosis and characterisation of cardiac masses
- Assessment of vascular morphology with the acquisition of three-dimensional datasets during magnetic resonance angiography (MRA).
Advantages and limitations of perfusion CMR
The diagnostic accuracy of myocardial perfusion CMR imaging for the detection of flow-limiting CAD is similar to that of radionuclide MPS (table 2). CMR allows accurate assessment of ventricular volumes and function, cardiac and vascular anatomy and morphology. Hence, it may be preferred over other functional imaging techniques in selected patients.
CMR does not involve the use of iodinated contrast agents. Since it is free of ionising radiation, perfusion CMR may be preferred over radionuclide MPS in young patients, particularly in women of child-bearing age, and in patients undergoing sequential studies who may otherwise receive a significant cumulative radiation dose. With the use of CMR imaging, it is possible to document the presence of myocardial hypoperfusion due to disease of the coronary microcirculation in patients with suspected microvascular angina.
Risks inherent to the technique and to the administration of contrast media must be considered. CMR is a safe procedure and no significant adverse events due to exposure to a high-strength magnetic field (≤ 1.5 Tesla) have been described in the literature. But CMR should be avoided in patients with ferromagnetic implants. Allergic reactions may also occur after injection of gadolinium-based contrast but these are usually mild and respond to supportive medical therapy.
Nephrogenic systemic fibrosis, a disorder characterised by fibrotic changes of the skin and other organs, has been attributed to administration of gadolinium chelates. Patients with renal impairment (eGFR <30 ml/min/1.73 m2) are at an increased risk and so a routine check of renal function (i.e. serum creatinine level) is recommended in all patients before gadolinium injection. Because most magnets feature a long tunnel with a narrow bore, CMR may not be suitable for patients with claustrophobia.
Myocardial perfusion imaging by CMR may be affected by cardiac and respiratory motion artefacts, differences in signal intensity between blood pool and subendocardium, and ECG triggering errors. Adequate patient preparation, including a thorough explanation of the procedure, appropriate selection of imaging protocol and careful attention to ECG triggering before gadolinium administration helps to minimise the impact of these potential sources of artefact on image interpretation.
Stress echocardiography
The combination of echocardiography with stress provides an accurate method for the assessment of the functional significance of CAD by means of detection of ischaemic changes of regional myocardial contractile function. As for other functional imaging techniques, stress echocardiography is best performed in symptomatic patients with an intermediate pre-test probability of flow-limiting coronary disease.
During a standard transthoracic echocardiogram, a piezoelectric transducer is placed on the chest wall. The transducer produces sound waves of ultra-high frequency (i.e. approx. 2.25 MHz in adults) that penetrate the body and are reflected by the tissues. The reflected waves are received by the transducer and converted back into an electrical signal. Most examinations include single-dimension or M-mode (motion mode), two-dimensional (cross-sectional), and Doppler imaging.
Although three-dimensional echocardiography may overcome some of the shortcomings of two-dimensional imaging, it has yet to become part of the routine echocardiographic examination. Stress echocardiography is usually performed with harmonic imaging in the two parasternal and three apical views and it is combined with one of the several forms of cardiac stress currently available.
Dynamic exercise on a treadmill or a bicycle ergometer has the advantage of inducing a physiological response to cardiovascular stress. The main shortcoming of this form of stress is that images cannot be obtained throughout the test and hence wall motion can only be assessed immediately after completion of the test. Dynamic exercise is performed in an identical way to conventional exercise ECG with image acquisition at baseline and at peak stress.
In patients who are unable to exercise appropriately, pharmacological stress is an alternative. Dobutamine is the most commonly used stress agent because of its dose-dependent effect on myocardial oxygen needs, increasing oxygen demand by means of its positive inotropic and chronotropic effects. Dobutamine is administered intravenously using a step-wise protocol starting at 5–10 µg/kg/min, increasing by 5–10 µg/kg/min every three to five minutes up to a maximum of 40 µg/kg/min.
Intravenous atropine may be administered to achieve target heart rate. Short- and long-axis views of the left ventricle are obtained at baseline and at each dobutamine dose step. The test is continued until a stress end point is reached (figures 20 and 21). A normal response to stress involves a progressive increase in regional contractile function. A new or worsening regional wall motion abnormality during stress is a sensitive marker of significant coronary obstruction.


Injection of contrast media (gas-filled microbubbles) can be used for better delineation of left ventricular endocardial border by means of left ventricular cavity opacification, which improves image quality and enhances visualisation of regional wall motion abnormalities (figure 21).
Vasodilator agents are less efficient at inducing regional wall motion abnormalities and hence they are preferred for assessment of perfusion by myocardial contrast echocardiography (MCE). For this, a microbubbles-based contrast agent is injected intravenously at rest and at peak stress; myocardial perfusion abnormalities appear as areas of slow contrast replenishment within the ultrasound beam after initial destruction of the microbubbles using high energy ultrasound. Quantification of flow velocity and myocardial blood volume is possible and the product of the two is proportional to myocardial blood flow.
Other indications for stress echocardiography:
The following are indications for stress echocardiography:
- Assessment of cardiac morphology (e.g. congenital heart disease)
- Evaluation of valve anatomy and function
- Assessment of global and regional left ventricular function
- Detection of viable and hibernating myocardium
- Diagnosis of pericardial effusion and cardiac tamponade
- Evaluation of constrictive and restrictive disorders of the heart
Advantages and limitations of stress echocardiography
Echocardiography provides a rapid and reliable method for visual and quantitative assessment of cardiac morphology and function. It is a safe technique and no adverse effects have been documented from exposure to ultrasound frequencies currently used. The diagnostic performance of stress echocardiography is similar to that of other functional imaging techniques (table 2).
More importantly, it provides valuable incremental prognostic information that allows stratification of patients into low risk (<1% annual cardiac event rate) if normal, or high risk (>10%) if abnormal. Stress echocardiography is also a versatile technique that can be combined with almost all forms of stress available although some modalities of stress are preferred over others (e.g. dobutamine vs. dipyridamole stress).
Stress echocardiography has a lower sensitivity for the detection of multi-vessel as well as single-vessel CAD, especially if coronary stenoses are mild in severity or affect a small region of myocardium. The lower sensitivity but relatively higher specificity is mainly attributed to the fact that stress echocardiography relies on ischaemia provocation and its immediate consequences. Moreover, abnormalities of contractile function occur late in the ischaemic cascade in the presence of flow-limiting CAD.
Myocardial perfusion by MCE may enhance the sensitivity of stress echocardiography for the detection of CAD as perfusion abnormalities precede wall motion abnormalities. The diagnostic yield of echocardiography also depends on image quality. A stress echocardiogram may not be diagnostic in cases of poor tissue penetration or limited access and hence other imaging modalities may be considered.
Although it is a robust and reliable diagnostic tool, echocardiography is highly operator-dependent and stress echocardiography, in particular, relies heavily on skilful and experienced users. The detection of ischaemia-induced contractile abnormalities can be challenging in the presence of resting wall motion abnormalities as seen in patients with left bundle branch block, permanent pacing, Wolf-Parkinson-White syndrome and post-CABG.
Patients with chest pain and a high probability of significant CAD
Coronary angiography (figure 8) is the definitive method of establishing whether or not a patient has CAD. Although it clearly shows the severity of anatomical lesions, it does not tell us about myocardial perfusion. Information relating to the degree of flow restriction can now be obtained using a pressure-flow wire (see Angina module 7: revascularisation in stable coronary artery disease) to measure fractional flow reserve (FFR), which is an index of physiological significance.
Angiography is a prerequisite for percutaneous coronary intervention (PCI, angioplasty). The technique is generally simply and easily performed, in 15–20 minutes, under local anaesthetic, often as a day-case procedure. The major complications, such as provocation of MI, stroke, or death, are fortunately rare; their risk estimated at about one or two in every 1,000.6
Haemorrhage from the puncture site is a more common complication and this can be reduced by using the radial artery access, although many operators continue to use the femoral artery approach. After the introduction of a small sheath into the artery, a series of catheters are introduced into the heart and using radio-opaque contrast injections both left ventricular contraction and coronary anatomy is established.

A series of coronary pictures are obtained in differing projections to avoid overlap of vessels and to carefully examine for high grade stenoses. Following the procedure, the sheath is withdrawn and haemostasis is established by direct pressure to the artery or by using a compression assist device such as that pictured, which offers haemostasis management using hands-free compression of the femoral artery after diagnostic or therapeutic catheterisation.
Non-invasive testing in women with suspected CAD
A statement from the American Heart Association (AHA) on the role of noninvasive testing in the clinical evaluation of women with suspected CAD7 includes an evidence synthesis on the abundant diagnostic and prognostic accuracy data for the diagnostic evaluation of women with ischaemic symptoms. Firstly it recommends that, low-risk women, with some exceptions, are not candidates for diagnostic testing.
Second, it recommends an initial exercise ECG–first strategy for women at low and intermediate IHD risk. Third, for symptomatic women with functional disability, an indeterminate ETT, or an abnormal rest ECG, echocardiography or MPI is recommended or CMR may be considered a reasonable test option. CCTA may also be considered reasonable for women at intermediate IHD risk. Premenopausal women at intermediate CAD risk who are functionally disabled generally should undergo echocardiography or CMR but may undergo MPI or CCTA if an effective radiation dose of <3 mSv is possible. Risk stratification is based on the extent and severity of inducible abnormalities noted on the stress examination.
Moreover, evidence supports CMR and CCTA as being accurate in the detection of obstructive CAD and for IHD risk assessment of symptomatic women, according to this AHA statement. CCTA can uniquely provide information on the obstructive and non-obstructive burden of CAD, which may be reasonable to guide post-test management approaches for women.
There is a growing evidence base with vasodilator stress myocardial perfusion positron emission tomography (PET), and evidence supports a high diagnostic and prognostic accuracy for both women and men.8
close window and return to take test
References
1. National Institute for Health and Care Excellence. Chest pain. NICE pathway. Manchester: NICE, 2017. https://pathways.nice.org.uk/pathways/chest-pain [Accessed 6 January 2020]
2. National Institute for Health and Care Excellence (NICE). Chest pain of recent onset. Clinical guideline 95: London: National Clinical Guideline Centre for Acute and Chronic Conditions, 2010. http://guidance.nice.org.uk/CG95
3. Alfakih K, Greenwood JP, Plein S. The 2016 update to NICE CG95 guideline for the investigation of new onset stable chest pain: more innovation, but at a cost? Clin Med 2017;17:20911. https://doi.org/10.7861/clinmedicine.17-3-209
4. Greenland P, Bonow RO, Brundage BH, et al. ACCF/AHA 2007 Clinical expert consensus document on coronary artery calcium scoring by computed tomography in global cardiovascular risk assessment and in evaluation of patients with chest pain. Circulation 2007;115:402–26. http://dx.doi.org/10.1161/CIRCULATIONAHA..107.181425
5. Picano E, Vañó E, Rehani MM, et al. The appropriate and justified use of medical radiation in cardiovascular imaging: a position document of the ESC Associations of Cardiovascular Imaging, Percutaneous Cardiovascular Interventions and Electrophysiology. Euro Heart J 2014. http://dx.doi.org/10.1093/eurheartj/eht394
6. Coronary heart disease – diagnosis. NHS Choices. (http://www.nhs.uk/Conditions/Coronary-heart-disease/Pages/Diagnosis.aspx)
7. Mieres JH, Gulati M, Merz NB et al. Role of noninvasive testing in the clinical evaluation of women with suspected ischemic heart disease. A consensus statement from the American Heart Association. Circulation 2014;130:350–79. http://dx.doi.org/10.1161/CIR.0000000000000061
8. Baldassarre LA,Raman SV, Min JK et al. Noninvasive imaging to evaluate women with stable ischemis heart disease. JACC Cardiovasc Imag 2016:9:421-35. https://doi.org/10.1016/j.jcmg.2016.01.004
Recommended further reading:
Pennell DJ. Cardiovascular magnetic resonance. Circulation 2010;121:692–705. http://dx.doi.org/10.11.61/circulationaha.108.811547
Kaufmann PA, Knuuti J. Ionizing radiation risks of cardiac imaging: estimates of the immeasurable. European Heart Journal (2011) 32, 269–271. http://dx.doi.org/10.1093/eurheartj/ehq298
Stokes MB, Roberts-Thompson R. The role of cardiac imaging in clinical practice. Aust Prescr 2017;40(4):151–5. https://dx.doi.org/10.18773/austprescr.2017.045
Carrabba N, Migliorini A, Pradella S, et al. Old and new NICE Guidelines for the evaluation of new onset stable chest pain: a real world perspective. BioMed Res Int 2018;2018,Article ID 3762305. https://doi.org/10.1155/2018/3762305
close window and return to take test
All rights reserved. No part of this programme may be reproduced, stored in a retrieval system, or transmitted in any form or by any means, electronic, mechanical, photocopying, recording or otherwise, without the prior permission of the publishers, Medinews (Cardiology) Limited.
It shall not, by way of trade or otherwise, be lent, re-sold, hired or otherwise circulated without the publisher’s prior consent.
Medical knowledge is constantly changing. As new information becomes available, changes in treatment, procedures, equipment and the use of drugs becomes necessary. The editors/authors/contributors and the publishers have taken care to ensure that the information given in this text is accurate and up to date. Readers are strongly advised to confirm that the information, especially with regard to drug usage, complies with the latest legislation and standards of practice.
Healthcare professionals should consult up-to-date Prescribing Information and the full Summary of Product Characteristics available from the manufacturers before prescribing any product. Medinews (Cardiology) Limited cannot accept responsibility for any errors in prescribing which may occur.

All rights reserved. No part of this programme may be reproduced, stored in a retrieval system, or transmitted in any form or by any means, electronic, mechanical, photocopying, recording or otherwise, without the prior permission of the publishers, Medinews (Cardiology) Limited.
It shall not, by way of trade or otherwise, be lent, re-sold, hired or otherwise circulated without the publisher’s prior consent.
Medical knowledge is constantly changing. As new information becomes available, changes in treatment, procedures, equipment and the use of drugs becomes necessary. The editors/authors/contributors and the publishers have taken care to ensure that the information given in this text is accurate and up to date. Readers are strongly advised to confirm that the information, especially with regard to drug usage, complies with the latest legislation and standards of practice.
Healthcare professionals should consult up-to-date Prescribing Information and the full Summary of Product Characteristics available from the manufacturers before prescribing any product. Medinews (Cardiology) Limited cannot accept responsibility for any errors in prescribing which may occur.