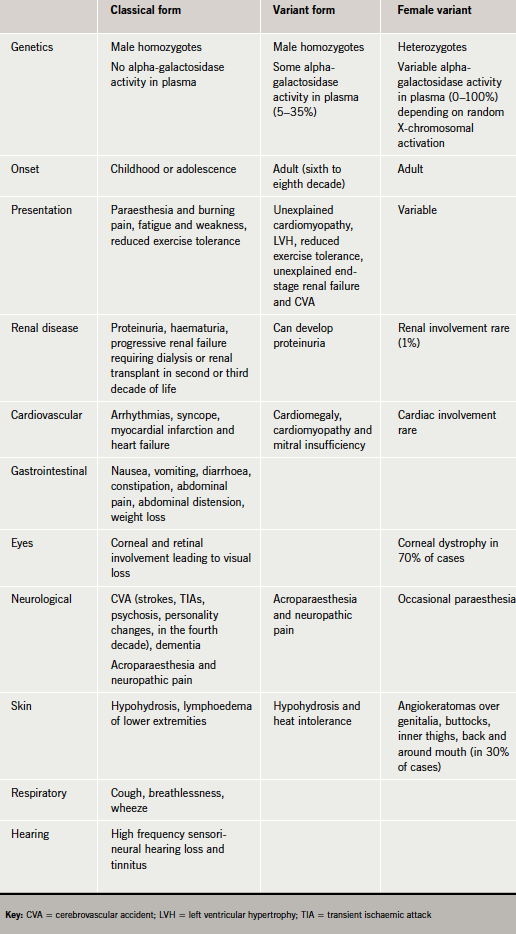
Anderson-Fabry disease is a rare X-linked recessive lysosomal storage disease that may cause a wide range of symptoms affecting multiple systems. It is due to a DNA mutation in the enzyme alpha-galactosidase A; this causes an accumulation of a glycolipid, globotriaosylceramide, within blood vessels, tissues, and organs, impairing their function.
Typically, males experience severe symptoms, but the impact on women is variable, with some being asymptomatic and others having severe symptoms. Although the diagnosis can often be readily made in males by measuring the blood level of alpha-galactosidase activity, in females, gene sequencing is preferred as enzyme activity may be within the normal range. However, the disease may not be suspected as many symptoms are shared with other disease processes. Important clues are multi-system symptoms that vary in age of onset, severity and manner of progression; early onset of kidney failure; and stroke or heart disease in the absence of conventional vascular risk factors. Enzyme replacement therapy is available.
Introduction
Fabry disease, or Anderson-Fabry disease (AFD), is an X-linked lysosomal storage disorder, second only in frequency to Gaucher disease.1 All daughters of a hemizygous male will be affected but none of the sons. An affected woman has a 50% chance of passing the disorder on to both male and female offspring.
There are over 350 mutations in the gene coding for the enzyme galactosidase A resulting in pathological accumulation of the lipid globotriaosylceramide (Gb3) in many body organs including vascular endothelium and vascular smooth muscle cells, skin, eyes, heart, kidneys, brain and peripheral nervous system.2,3 With disease progression, organs fail and premature death can occur, often from renal failure or a cardiac or cerebrovascular event, typically in the late fifties or sixties.3 Life expectancy can be shortened by 20 years in men and 15 years in women.
The disease presents in three forms: classical form, variant form or female variant (table 1).
In Caucasians, Fabry disease affects between one in 40,000 and one in 117,000 individuals.4,5 Male patients present at an earlier age and tend to
have more symptoms than female patients. However, due to variations and random inactivation of the X chromosome, females tend to have more severe disease.6
When to suspect Fabry disease
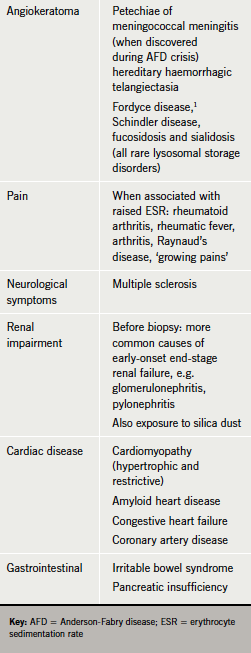
Fabry disease is rare and presents in many ways. Symptoms can be very non-specific,7,8 sharing many of the features of rheumatic fever, arthritis, fibromyalgia, dermatomyositis, multiple sclerosis, idiopathic hypertrophic cardiomyopathy, renal failure of unknown aetiology, Meniere’s disease and irritable bowel.9,10 This may delay diagnosis.
The clinician should suspect Fabry disease in any patient with multi-system symptoms varying in age of onset, severity and manner of progression (table 2). Kidney failure, or stroke or heart disease occurring early or in the absence of conventional vascular risk factors should direct the physician to take a family history, which may identify other similarly affected relatives with undiagnosed Fabry disease.
Screening should be considered in all family members of patients with Fabry disease, young patients with unexplained left ventricular hypertrophy, cerebrovascular disease or unexplained end-stage renal failure requiring dialysis, and patients with multiple renal cysts of unknown aetiology.11

Diagnosis
Several diagnostic test kits are available to measure plasma alpha-galactosidase A activity. In males, enzyme activity may be absent in classical, or low in variant, Fabry disease. DNA analysis confirms the diagnosis. In females, levels of alpha-galactosidase activity may be within the normal range, and gene sequencing is the preferred test.12,13
Exceptionally, skin biopsy or culture of skin fibroblasts may demonstrate the characteristic glycolipid deposits. Renal biopsy can also be helpful in reaching a diagnosis but is rarely necessary.11 Prenatal diagnosis can be obtained by detecting deficient enzyme activity measured from chorionic villus biopsy in the first trimester or cultured amniotic cells in the second trimester.14
Fabry disease and the heart
Fabry cardiomyopathy is characterised by reduced myocardial contraction, resulting from stiffening of cardiomyocytes, and relaxation velocities at tissue Doppler imaging (TDI). Such abnormalities are caused by Gb3 deposition and are detectable before the development of left ventricular hypertrophy (LVH).15 As the disease progresses, myocardial fibrosis becomes prominent, accompanied by disturbance of the myofilamental structure.16 In many patients, ejection fraction is well preserved and cardiac disease progresses without symptoms.
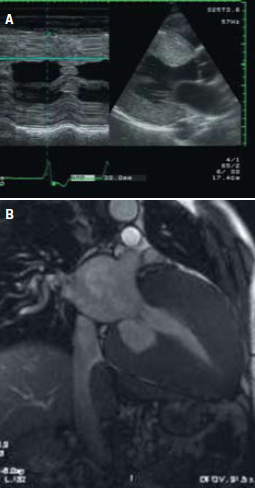
Echocardiographic studies show a nonspecific binary appearance of the endocardial border corresponding to endomyocardial sphingolipid deposition, creating a two-layered appearance of the myocardium. This binary appearance is more evident in the left side of the interventricular septum but is observable in most cases all along the left ventricular (LV) chamber contour and, in some patients with marked LVH, even in the right side of the interventricular septum and the free right ventricular wall.15
Echocardiographic studies conducted by Chimenti and others on patients with Fabry disease show an increase in LV septum and posterior wall thickness, LV mass, and LV atrial volume and mild right ventricular hypertrophy, compared with control subjects. Doppler studies show an increased LV filling pressure.13
Even though transthoracic echocardiograms can be completely normal, systolic and diastolic dysfunction can be recognised with TDI. Biventricular impairment can be responsible for the progression of cardiac disease.16
Cardiac symptoms
Patients commonly present with symptoms that include palpitations, angina, dyspnoea and syncope, and investigations may reveal ventricular hypertrophy, cardiomyopathy, conduction defects, aortic and mitral valve abnormalities, coronary artery disease, aortic root dilatation and heart failure (figure 1).17,18 Half of the patients with Fabry disease report angina, though coronary arteries are normal at angiography. Regional wall motion abnormalities suggest endothelial dysfunction due to Gb3 deposition.19,20

The difficulty lies in identifying an index case of Fabry disease from among those referred for a cardiology opinion. Some features may lead the clinician to suspect Fabry disease. Symptoms indicating Fabry disease include:
- angina, dyspnoea or stroke in young adults with no cardiovascular risk factors
- unexplained syncope
- unexplained renal impairment leading to end-stage renal disease.
LVH in young patients: structural changes may resemble hypertrophic cardiomyopathy (HCM) and around 3% to 9% of male patients with myopathy have been found to have low levels of Gal-A,21 but in Fabry disease the hypertrophy is symmetrical.22,23
Conduction defects: Gb3 can be deposited anywhere in the conduction system but suspect Fabry when isolated repolarisation abnormalities (ST-T wave changes in leads I, II, aVL, and V4−6) occur in the absence of other causes such as hypertension or aortic stenosis. A short PR interval with RBBB pattern is also suspicious.
Autonomic dysfunction: Reduced heart rate variability in patients with Fabry disease indicates impairment of the autonomic system.
Valvular defects: Aortic and mitral valve dysfunction, predominantly regurgitation due to valve thickening, occurs in approximately half of Fabry patients. Aortic root dilatation is not severe enough to require valve or root replacement.24-26
Cardiac investigations and differential diagnoses are provided in tables 3 and 4.27
Management of Fabry disease
Enzyme replacement therapy
Recombinant alpha-galactosidase A has been available in Europe since 2001 and in the USA since 2003.
There are two enzyme preparations, both well tolerated. Agalsidase alfa is produced in a human cell line by gene activation. It is administered by intravenous infusion at 0.2 mg/kg every other week over approximately 40 minutes, usually without routine premedication. Agalsidase beta is produced in Chinese hamster ovary cells by recombinant techniques and is administered by intravenous infusions every other week at a dose of 1.0 mg/kg. Premedication (antipyretic and/or antihistamine) is routinely used. Both enzymes are available in Europe and many other countries. The regulatory approval of both drugs was based on the results of randomised, placebo-controlled clinical trials of each preparation.16
The key in the treatment of Fabry disease is early diagnosis and early start of replacement therapy, as late recognition and diagnosis may mean that end organ damage is irreversible.12,28 Referral to a specialist centre designated by the Department of Health National Commissioning Group for patients with lysosomal storage disorders including Fabry disease is recommended for confirmation of the diagnosis and appropriate treatment and monitoring.
Both forms of the enzyme may clear Gb3 from affected tissues with improvement in clinical parameters including LV thickness and improved LV function; QRS duration and heart failure symptoms; decreased frequency in pain crisis; improvement of gastrointestinal and pulmonary symptoms; increased sweating; and improved hearing and sensation. Those with mild kidney dysfunction or mild LVH respond better to treatment.12,29,30
Between 55% and 80% of patients on treatment develop antibodies against agalsidase alfa and beta. The clinical effect of these antibodies is still unclear.12
All affected males, irrespective of age, should be treated with enzyme replacement. The recommendations for heterozygous females are different; only females with cerebrovascular disease, reduced glomerular filtration rate, proteinuria, LVH or significant valvular regurgitation should be treated.12
Symptomatic treatment
Whatever the response to enzyme replacement therapy, treatment of symptoms is necessary.12
- Chronic pain is treated with anticonvulsants (e.g. carbamazepine, gabapentin, phenytoin), non-steroidal anti-inflammatory drugs or opiates, and minimisation of activities that trigger painful crises, e.g. physical exertion, temperature changes, emotional stress.
- Angiokeratoma are removed with argon laser therapy.
- Early stage renal impairment is treated with angiotensin-converting enzyme (ACE) inhibitors (in patients without renal artery stenosis). Renal failure is treated with dialysis or transplantation.
- Chest pain is treated with anti-anginals; heart failure is managed with diuretics, ACE inhibitors, digoxin, angiotensin II (AII) receptor blockers.
- Tachyarrhythmia is managed with anti-arrhythmics, anticoagulants or implantable cardio-defibrillators; symptomatic bradycardia requires pacemaker insertion.
- Gastrointestinal symptoms are managed with a low-fat diet and motility agents.
- Neurovascular disease is treated with aspirin and clopidogrel.27
Yearly follow-up should be instituted for patients undergoing treatment and should include a detailed physical examination, routine haematology and chemistry, urinalysis with urinary protein and creatinine clearance, echocardiogram and electrocardiogram, pain and quality-of-life assessment.12
More studies are necessary to establish the effect of long-term treatment on the different aspects of Fabry disease.
Conclusion
Fabry disease can lead to significant morbidity, multi-system compromise and premature mortality, making it an important condition to diagnose and treat. Recent evidence suggests that early enzyme replacement therapy improves outcome, so clinicians should have a low threshold for considering Fabry disease as a possible diagnosis in young patients with a characteristic clinical phenotype or with unexplained cerebrovascular, cardiac or renal impairment.
Further research will help to identify protocols for investigation and lend support to the use of enzyme replacement therapy and other forms of management.
Acknowledgements
The authors are grateful to Dr Mike Sosin, Consultant Cardiologist, Nottingham University Hospitals for supplying the echo and MRI images.
Conflict of interest
None declared.
Key messages
- Fabry disease is difficult to diagnosis but should be suspected if multi-system disorders present in young people without conventional risk factors
- Referral to a specialist centre is recommended for confirmation of the diagnosis and appropriate treatment and monitoring
References
- Eng CM, Ashley GA, Burgert TS et al. Fabry disease: thirty-five mutations in the alpha-galactosidase A gene in patients with classic and variant phenotypes. Mol Med 1997;3:174.
- Desnick R, Ioannou Y, Eng C. Alpha-galactosidase A deficiency: Fabry disease. In: Scriver CR, Beaudet AL, Sly WS et al. (eds). The metabolic and molecular bases of inherited disease, 8th ed. New York: McGraw Hill, 2001;3733.
- MacDermot KD, Holmes A, Miners AH. Anderson-Fabry disease: clinical manifestations and impact of disease in a cohort of 98 hemizigous males. J Med Genet 2001;38:750. http://dx.doi.org/10.1136/jmg.38.11.750
- Branton MH, Sciffmann R, Sabnis SG et al. Natural history of Fabry renal disease: influence of alpha-galactosidase A activity and genetic mutations on clinical course. Medicine (Baltimore) 2002;81:122. http://dx.doi.org/10.1097/00005792-200203000-00003
- Meikle PJ, Hopwood JJ, Clague AE, Carey WF. Prevalence of lysosomal storage disorders. JAMA 1999;281:249. http://dx.doi.org/10.1001/jama.281.3.249
- Wilcox WR, Oliviera JP, Hopkin RJ et al. Females with Fabry disease have major organ involvement: lesson from the Fabry registry. Mol Genet Metab 2008;93:112–28. http://dx.doi.org/10.1016/j.ymgme.2007.09.013
- Deegan PB, Baehner AF, Barba Romero MA, Hughes DA, Kampmann C, Beck M. Natural history of Fabry Disease in females in the Fabry outcome survey. J Med Genet 2006;43:347–52. http://dx.doi.org/10.1136/jmg.2005.036327
- Eng CM, Fletcher J, Wilcox WR et al. Fabry disease: baseline medical characteristics of a cohort of 1765 males and females in the Fabry Registry. J Inherit Metab Dis 2007;30:184–92. http://dx.doi.org/10.1007/s10545-007-0521-2
- Houge G, Skarbovik AJ. Fabry disease – a diagnostic and therapeutic challenge. Tidsskr Nor Laegeforen 2005;125:1004.
- Metha A, Ricci R, Widmer U et al. Fabry disease defined: baseline clinical manifestations of 366 patients in the Fabry Outcome Survey. Eur J Clin Invest 2004;34:236. http://dx.doi.org/10.1111/j.1365-2362.2004.01309.x
- Sunder-Plassmann G, Födinger M. Diagnosis of Fabry disease: the role of screening and case-finding studies. Chapter 17 in Fabry Disease: perspectives from 5 years of FOS. Mehta A, Beck M, Sunder-Plassmann G, editors. Oxford: Oxford PharmaGenesis; 2006.
- Zarate YA, Hopkin RJ. Lysosomal storage disease 3. Lancet 2008;372:1427–35. http://dx.doi.org/10.1016/S0140-6736(08)61589-5
- Elliott P. Anderson–Fabry disease: an important differential diagnosis in patients with unexplained left ventricular hypertrophy. Heart Metab 2006;33:25–9.
- Hughes DA, Ramaswami U, Elliott PM, et al. Guidelines for the diagnosis and management of Anderson-Fabry disease. www.dh.gov.uk/en/Publicationsandstatistics/Publications/PublicationsPolicyAndGuidance/DH_4118404
- Pieroni M, Chimenti C, De Cobelli F et al. Echocardiographic detection of endomyocardial glycosphingolipid compartmentalization. J Am Coll Cardiol 2006;47:1663–71. http://dx.doi.org/10.1016/j.jacc.2005.11.070
- Lidove O, West M, Pintos-Morell G et al. Effects of enzyme replacement therapy in Fabry disease – A comprehensive review of the medical literature. Genet Med 2010;12:668–79.
- Nagueh SF. Fabry disease. Heart 2003;89:819. http://dx.doi.org/10.1136/heart.89.8.819
- Kampmann C, Baehner F, Ries M, Beck M. Cardiac involvement in Anderson-Fabry disease. J Am Soc Nephrol 2002;13(suppl 2):S147.
- Kampmann C, Baehner F, Ries M, Beck M. Cardiac involvement in Anderson-Fabry Disease. J Am Soc Nephrol 2002;13(suppl 2):S147.
- Barbey F, Brakch N, Linhart A et al. Increased carotid intima-media thickness in the absence of atherosclerotic plaques in an adult population with Fabry Disease. Acta Paediatr 2006;95(suppl):63. http://dx.doi.org/ 10.1080/08035320600618924
- Hughes DA, Elliott PM, Shah J et al. Effects of enzyme replacement therapy on the cardiomyopathy of Anderson-Fabry disease: a randomised, double-blind, placebo-controlled clinical trial of agalsidase alfa. Heart 2008;94:153–8. http://dx.doi.org/10.1136/hrt.2006.104026
- Linhart A, Palecek T, Bultas J et al. New insights in cardiac structural changes in patients with Fabry disease. Am Heart J 2000;139:1101. http://dx.doi.org/10.1067/mhj.2000.105105
- Von Scheidt W, Eng CM, Fitzmaurice TF et al. An atypical variant of Fabry disease with manifestations confined to the myocardium. N Engl J Med 1991;324:395–9. http://dx.doi.org/10.1056/NEJM199102073240607
- Linhart A, Palecek T, Bultas J et al. New insights in cardiac structural changes in patients with Fabry’s Disease. Am Heart J 2000;139:1101. http://dx.doi.org/10.1067/mhj.2000.105105
- Goldman ME, Cantor R, Schwartz MF et al. Echocardiographic abnormalities and disease severity in Fabry Disease. J Am Coll Cardiol 1986;7:1157. http://dx.doi.org/10.1016/S0735-1097(86)80238–8
- Desnick RJ, Blieden LC, Sharp HL et al. Cardiac valvular anomalies in Fabry Disease. Clinical, morphologic and biochemical studies. Circulation 1976;54:818.
- Hoffmann B. Fabry disease: recent advances in pathology, diagnosis, treatment and monitoring. Orphanet Journal of Rare Diseases 2009,4:21 http://www.ojrd.com/content/4/1/21
- Weidmann F, Niemann M, Breunig F et al. Long term effects of enzyme replacement therapy on Fabry cardiomyopathy, evidence for a better outcome with early treatment. Circulation 2009;119:524–9. http://dx.doi.org/10.1161/CIRCULATIONAHA.108.794529
- Schiffman RS, Kopp JB, Austin HA 3rd et al. Enzyme replacement therapy in Fabry disease: a randomized controlled trial. JAMA 2001;285:2743–9. http://dx.doi.org/ 10.1001/jama.285.21.2743
- Kampmann C, Whybra C, Baehner FA, Beck M. Enzyme replacement therapy in Anderson-Fabry cardiomyopathy. Heart Metab 2002;18:39–41.


