Introduction
In 1988 the ISIS-2 (Second International Study of Infarct Survival) study brought about a sea change in the management of the patient suffering a myocardial infarction (MI) and, in particular, those who had ST-elevation (STEMI) changes on their electrocardiogram (ECG).1 Prior to that landmark trial, general practitioners (GPs) were much more involved in the care of patients suffering MIs. They had to decide with what urgency the patient had to be admitted, or even if they would be admitted at all, in the light of how little could be done for the patient in hospital. The care provided has improved since then to the point that the GP is rarely involved in the acute care of the MI patient. The incidence of MI has also reduced to less than half of that in 1988 due to improvements in lifestyle, particularly smoking cessation, and control of risk factors such as high cholesterol and blood pressure.2 The impressive decline in ischaemic heart disease (IHD) mortality witnessed over the last 20 years is a direct consequence of the reduction in overall MI event rates, but is also related to substantially improved acute medical therapies.3
However, IHD (principally manifest through MI) as an individual disease entity, remains the number one cause of death in the UK, killing approximately 73,000 people annually, of which 23,000 deaths are premature.4 So, MI still occurs frequently, around one every seven minutes across the UK, remains one of the most common causes of acute hospitalisation and yields a great many survivors who inevitably require complex care after discharge. The GRACE (Global Registry of Acute Coronary Events) registry tells us that the risk of death is higher for STEMI (8%) compared with non-STEMI (NSTEMI) (5%) during the in-hospital phase, but that once the acute index event has been survived, this trend is reversed, with six-month mortality rates of 5% for STEMI and 6% for NSTEMI. For those patients who are still alive at six months after discharge, one in five will die within the subsequent five years.5 This clearly demonstrates the chronic, incurable nature of IHD and that, although there has been considerable investment in the STEMI emergency care pathway during recent years, much more is required if the ongoing burden of acute events and long-term morbidity is to be reduced.
The period in hospital has become very short, often less than 48 hours for STEMI patients, and the opportunity for a hospital follow-up is usually just one visit, if that, for the majority of MI patients. Therefore, the role of primary care has now become that of providing post-heart attack care. How well is that done, how good is the communication between secondary care and primary care and how often is there an assumption that everything is being done by the cardiac rehabilitation team? In addition, our patients are increasingly elderly with multiple, complex medical conditions, so it is often unclear as to what the short-, medium- and longer-term priorities are and where (and by whom) these are best delivered.
What needs to be done?
The majority of patients who have experienced a MI are either frightened and confused or are in a state of utter disbelief. There are many questions to answer. Why me? What happens next? Will it happen again? Will I die? Will I be disabled? Can I drive? Can I go back to work? What are all these pills for? The psychological impact can be immense. This may not be their first event or their only problem. How much exercise can they do? Who will help them stop smoking? What foods can they eat and can they drink alcohol? Can they have sex?
For first timers, they are suddenly taking many drugs. The beta blocker and angiotensin-converting enzyme (ACE) inhibitor will need up-titrating. They are likely to be taking a high-dose, high-intensity statin, which together with the ACE inhibitor will require blood tests in the near future. Will the statin have adequately reduced their cholesterol? Do they need to be considered for ezetimibe or even a proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor? When should they use their glyceryl trinitrate (GTN) spray and how long should they tolerate chest pain before calling 999? How long should they take dual anti-platelet therapy (DAPT)? What should they do about bruising or bleeding? After one year should they stop DAPT or step down their dose?
This is not only confusing for the patient but it can be equally difficult for the primary care team. Not all of these questions have straightforward answers and become increasingly difficult when there are comorbid conditions, such as atrial fibrillation.
The discharge process
Bed availability pressures mean that quick discharges are necessary, so having a cardiac liaison or discharge nurse has become a common and successful approach. Of even greater value is the presence of a member of the relevant cardiovascular prevention and rehabilitation programme (CPRP) during discharge planning, as this has been demonstrated to have a favourable impact on subsequent CPRP attendance.6 The patient and their relatives require an explanation of what has happened and what will happen next. There will be too much to take in at once and, therefore, literature, such as the British Heart Foundation (BHF) leaflets, is useful. They need to know who to contact next and what to do if there are any problems, with appropriate telephone numbers or email addresses. They need a reasonable amount of discharge medication with some idea as to what the medicines are, when to take them and what side effects they might expect.
When designing discharge summaries the obvious people to liaise with are doctors, nurses and pharmacists working in primary care or rehabilitation. But do not forget to ask the opinion of patients and lay people too. The language must be precise. If they are not expected to stop the medication say ‘lifelong’ not ‘indefinitely’. The latter word means different things to different people. You could ask groups such as the Plain English Campaign for help.7 For patients for whom English is not their first language the provision of computer-generated, translated literature should be feasible.
One great irritation to GPs is secondary care sending them a list of delegated tasks. All healthcare professionals are busy people and we need to be mindful of trying not to burden others with tasks we could do ourselves.

The existence of the Myocardial Infarction National Audit Project (MINAP) has undoubtedly promoted best practice. One downside has been the creation of integrated discharge summaries to answer MINAP questions. This has produced information unintelligible to anyone other than a specialist doctor or nurse. They can also be full of unexplained acronyms and very lengthy. Box 1 contains the desirable information in a discharge summary. In an ideal world the patient would leave hospital with a copy of their discharge summary that they could understand.
Post-discharge care
Initially, this is a shared activity between cardiology, rehabilitation and general practice. For most patients it will eventually become a general practice only annual assessment, performed by the nursing team. The first attendance post-discharge will most probably be with the GP. It will be a 10-minute appointment. Mainly it will involve a review of the medication changes and is an opportunity to plan up-titration of beta blockers and ACE inhibitors. There may be a need to discuss employment factors and to issue an unfit or fit note.
Some hospitals will offer a follow-up appointment at six to 12 weeks in the cardiology clinic. This appointment might be with a doctor or specialist nurse. In some areas, there may be no such follow-up due to commissioning constraints. This clinic appointment will be an opportunity to screen for MI complications such as angina, heart failure, arrhythmias or other forms of myocardial injury. It may be an opportunity to organise occupational help such as an exercise ECG for heavy goods or public transport drivers. For the majority, however, the consultation will be routine and rapid with little or no intervention undertaken. Sometimes primary care will assume that there is more ongoing follow-up because patients are attending hospital for other functions, such as pacemaker checks. But it is likely that such checks are very specific and functional.
CPRPs – adding years to life
The cost of adding one year of life to the patient via a CPRP is between a third and a sixth of the cost of the primary percutaneous coronary intervention procedure.8 For patients who have experienced MI and/or coronary revascularisation, attending and completing a course of exercise-based cardiac rehabilitation (CR) is associated with an absolute risk reduction in cardiovascular mortality from 10.4% to 7.6% when compared with those who do not receive CR, with a number needed to treat (NNT) of 37. In terms of all-cause mortality, recurrent MI and repeat revascularisation, the effect of CR would appear to be neutral, however, there is a significant reduction in acute hospital admissions (reduced from 30.7% to 26.1%, NNT 22) which is a key determinant of the intervention’s overall cost-efficacy. Access to a comprehensive CPRP is a class 1 recommendation (level of evidence A) according to the most recently updated European Society of Cardiology (ESC) prevention guidelines,9 and is also fully endorsed by the National Institute for Health and Care Excellence (NICE) within its Myocardial Infarction: cardiac rehabilitation and prevention of further cardiovascular disease clinical guideline published in 2013.10
According to both NICE and the British Association for Cardiovascular Prevention and Rehabilitation (BACPR), CR should start within 10 days of discharge11 in order to maximise uptake, achieve the evidence-based outcomes and to ensure maximum impact upon readmission rates. However, data from the National Audit of Cardiac Rehabilitation (NACR) 2016 report, which is funded by the BHF and hosted by the Health Sciences department of the University of York, demonstrates waiting times of between 23 and 43 days across the regions of England. In addition, overall CR attendance post-MI, while increasing gradually year on year, remains well below the 65% target set out within the Cardiovascular Disease Outcomes Strategy document,12 and currently stands at 43% for Northern Ireland, 49% for England and 54% for Wales (data for Scotland unavailable). The primary care team needs to be made aware of patients who do not take up the service so that they can try and encourage them to do so. The access route into a local CPRP via a community-based referral must also be efficient and well publicised.
The first anniversary
It is not usual practice to have a first-year post-MI check-up, but there may be value in such a visit. In fact, it could easily be argued that there is very little merit in offering early (6–12 week) ‘routine’ hospital-based follow-up appointments for the majority of uncomplicated post-MI patients. These visits often lack structure or purpose, and unless there have been specific tests or concerns (such as the possible need for an implantable cardioverter defibrillator [ICD] referral or the result of a stress scan to be relayed) then they may be viewed as a waste of both resources and patients’ time. At this six to 12 week time point, patients should be either undergoing or have completed their CPRP and, thus, be surrounded by a multi-disciplinary team of cardiac specialists who will have far more time, insight and skills to perform a continuous assessment of their progress. Perhaps commissioners – local and specialist – should review funding arrangements for these ‘routine’ hospital appointments and consider diverting resources to CR so that local programmes can increase capacity, shorten waiting times and improve flexibility. Acute providers should be encouraged (or even mandated) to view CR referral and uptake as an essential part of the treatment pathway, no less important than the inpatient care or the take-home medication.
Patients will be very aware of the impending anniversary and there may be associated psychological effects. They might celebrate this milestone, but equally it may be an unwelcome reminder of an unpleasant event.
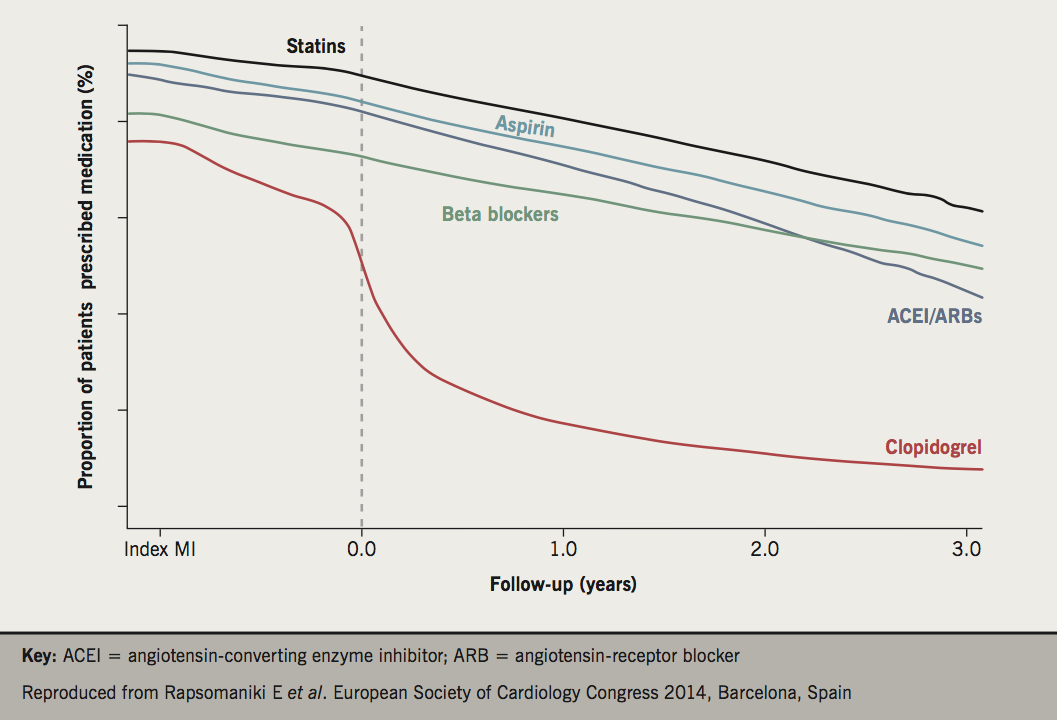
We know that compliance with secondary prevention medication will fall off to about 50–60% usage over a three-year period with a pronounced dip at one year (figure 1).13 Is their medication maximised? Is their cholesterol and blood pressure control adequate? Are they maintaining a healthy lifestyle? We can reinforce all these issues. In particular, we need to make decisions about antiplatelet medication at this point. Dual antiplatelet therapy finishes at this point for most patients. Should we continue aspirin in low-risk patients? Very high-risk patients could be considered for ongoing therapy with a combination of low-dose aspirin plus ticagrelor (at a reduced dose of 60 mg twice daily) following the PEGASUS-TIMI 54 (Prevention of Cardiovascular Events in Patients with Prior Heart Attack Using Ticagrelor Compared to Placebo on a Background of Aspirin-Thrombolysis in Myocardial Infarction 54) results which demonstrated a significant reduction in cardiovascular death, MI or stroke compared with those patients who did not receive ticagrelor beyond 12 months.14 Should we concentrate such a service on the very high-risk patients? In other words, offer this service to those with recurrent events, multiple comorbidities, diabetes, renal disease or older age groups.
Who should offer this service, primary or secondary care? Commissioners would be reluctant to support such a service without health economics evidence of benefit. Primary care might feel they are not qualified to make decisions about antiplatelet therapy for different groups of patients. A likely solution would be some form of shared care. Clearly, this is an area that needs research, but existing CPRPs already have the appropriate skill mix with which to offer a meaningful assessment at 12 months, particularly if supported by a local cardiologist or GP with a specialist cardiology interest.
Conclusion: action required…
As we have pointed out, there is ambiguity about the short-, medium- and long-term assessment of the post-MI patient and how well this is performed. That there is continued risk and a need for well-planned, shared care is without doubt. But how can we make this come about? Usually change happens in local National Health Service (NHS) healthcare provision because somebody champions the cause. In this case, the best placed people to do that will be the local interventional cardiologist, the director of cardiology services or a rehabilitation specialist. They need to reach out to primary care to begin a dialogue in order to design a clear local pathway with defined actions and measurable outcomes. It needs to be inclusive of a universally agreed discharge process, purposeful short-term follow-up for a selected minority, early uptake and completion of a CPRP for all and a first anniversary review, which may or may not target those deemed at highest risk for further acute events. It needs to include a robust communication strategy that involves all stake-holder professional groups and appropriate advertising to the general public. This could be a service design project or even a research project and at least in England, with the evolution of regional Sustainability and Transformation Plans, there exists a vehicle through which this might be delivered. A comprehensive strategy for the shared assessment of patients throughout the first year after the acute event should be a priority for all local healthcare providers and, whatever the chosen pathway, an audit of the process in terms of adherence, benefit and cost-efficacy will be required in order to ensure sustainability.
Conflict of interest
TM has received research grants, educational grants or speaker fees from Amgen, AstraZeneca, and MSD which may be relevant to this article. JM: none declared.
Key messages
- The management of the patient with an acute myocardial infarction (MI) has been revolutionised since the ISIS-2 study was published in 1988
- The GRACE registry tells us that for those patients still alive at six months after discharge, one in five will die within the subsequent five years
- Discharge might be best done using cardiac liaison or discharge nurse and should include written information using plain English
- Follow-up should involve a coordinated strategy, which includes cardiology, rehabilitation and general practice
- The National Audit of Cardiac Rehabilitation 2016 report demonstrates waiting times for rehabilitation of between 23 and 43 days across the regions of England
- The overall cardiac rehabilitation attendance post-MI currently stands at 43% for Northern Ireland, 49% for England and 54% for Wales
- A first-year anniversary follow-up consultation might be the best time to reinforce the need for lifestyle improvements, encourage drug adherence and plan changes to the antiplatelet regimen
References
1. ISIS-2 Collaborative Group. Randomised trial of intravenous streptokinase, oral aspirin, both, or neither among 17,187 cases of suspected acute myocardial infarction. Lancet 1988;332:349–60. https://doi.org/10.1016/S0140-6736(88)92833-4
2. Unal B, Critchley JA, Capewell S. Explaining the decline in coronary heart disease mortality in England and Wales between 1981 and 2000. Circulation 2004;109:1101–07. https://doi.org/10.1161/01.CIR.0000118498.35499.B2
3. Smolina K, Wright FL, Rayner M, Goldacre MJ. Determinants of the decline in mortality from acute myocardial infarction in England between 2002 and 2010; linked national database study. BMJ 2012;344:d8059. https://doi.org/10.1136/bmj.d8059
4. Heart UK. Key facts & figures. Available at: https://heartuk.org.uk/press/press-kit/key-facts-figures
5. Fox KAA, Carruthers KF, Dunbar DR et al. Underestimated and under-recognised: the late consequences of acute coronary syndrome (GRACE UK-Belgian Study). Eur Heart J 2010;31:2755–64. https://doi.org/10.1093/eurheartj/ehq326
6. Cossette S, Frasure-Smith N, Dupuis J, Juneau M, Guertin M. Randomized controlled trial of tailored nursing interventions to improve cardiac rehabilitation enrolment. Nursing Research 2012;61:111–20. https://doi.org/10.1097/NNR.0b013e318240dc6b
7. Plain English Campaign. Available at: www.plainenglish.co.uk
8. Fidan D, Unal B, Critchley J, Capewell S. Economic analysis of treatments reducing coronary heart disease mortality in England and Wales, 2000–2010. Q J Med 2007;100:277–89. https://doi.org/10.1093/qjmed/hcm020
9. Piepoli MF, Hoes AW, Agewall S et al. 2016 European guidelines on cardiovascular disease prevention in clinical practice. Eur Heart J 2016;37:2315–81. https://doi.org/10.1093/eurheartj/ehw106
10. National Institute for Health and Care Excellence. Secondary prevention in primary and secondary care for patients following a myocardial infarction. Clinical guideline 172. London: NICE, 2013. Available from: https://www.nice.org.uk/guidance/CG172
11. Buckley JP, Furze G, Doherty P et al. BACPR Scientific Statement. British standards and core components for cardiovascular disease prevention and rehabilitation. Heart 2013;99:1069–71. https://doi.org/10.1136/heartjnl-2012-303460
12. Department of Health. Cardiovascular disease outcomes strategy. London: Department of Health, March 2013. Available from: https://www.gov.uk/government/publications/improving-cardiovascular-disease-outcomes-strategy
13. Rapsomaniki E, Stogiannis D, Emmas C et al. Health outcomes in patients with stable coronary artery disease following myocardial infarction; construction of a PEGASUS-TIMI-54 like population in UK linked electronic health records. Eur Heart J 2014;35(suppl 1):363 abstract P2077.
14. Bonaca MP, Bhatt DL, Cohen M et al. Long-term use of ticagrelor in patients with prior myocardial infarction. PEGASUS-TIMI 54. N Engl J Med 2015;372:1791–1800. https://doi.org/10.1056/NEJMoa1500857
close window and return to take test
All rights reserved. No part of this programme may be reproduced, stored in a retrieval system, or transmitted in any form or by any means, electronic, mechanical, photocopying, recording or otherwise, without the prior permission of the publishers, Medinews (Cardiology) Limited.
It shall not, by way of trade or otherwise, be lent, re-sold, hired or otherwise circulated without the publisher’s prior consent.
Medical knowledge is constantly changing. As new information becomes available, changes in treatment, procedures, equipment and the use of drugs becomes necessary. The editors/authors/contributors and the publishers have taken care to ensure that the information given in this text is accurate and up to date. Readers are strongly advised to confirm that the information, especially with regard to drug usage, complies with the latest legislation and standards of practice.
Healthcare professionals should consult up-to-date Prescribing Information and the full Summary of Product Characteristics available from the manufacturers before prescribing any product. Medinews (Cardiology) Limited cannot accept responsibility for any errors in prescribing which may occur.




