![]() This article is available as a ‘Learning with reflection’ CPD activity
This article is available as a ‘Learning with reflection’ CPD activity
In November 2016, the National Institute for Health and Care Excellence (NICE) published an update of its guideline for the investigation of chest pain of recent onset (CG95), bringing computed tomography coronary angiography (CTCA) to the forefront as the first-line investigation. CTCA has a high negative-predictive value for the identification of obstructive coronary artery disease (CAD), but its positive-predictive value may be as low as 48%. Moreover, until recently it was unable to determine the functional significance of stenoses identified by CTCA. Using advanced computational fluid dynamics (CFD), HeartFlow® has pioneered a system that can predict the invasive fractional flow reserve (FFR) from a standard CTCA acquisition (FFRCT). The PLATFORM study has demonstrated that the use of CTCA with FFRCT was associated with equivalent clinical outcomes in terms of major adverse cardiovascular events (MACE) and quality of life at one year compared with usual testing. The global ADVANCE registry, and other long-term follow-up studies in over 9,000 patients, have demonstrated extremely good long-term outcomes when patients with CAD but negative FFRCT are managed conservatively without invasive testing. In a technology appraisal (MTG32), NICE projected that the adoption of this technology within the NHS in England could save at least £9.1 million by 2022. While it is accepted that the HeartFlow® FFRCT shows a lot of promise, there are a number of limitations that need to be considered.
Introduction
In 2010, the UK’s National Institute for Health and Care Excellence (NICE) published a landmark guideline for the investigation of patients with chest pain of recent onset (CG95). This placed a major emphasis on determining the pre-test probability of coronary artery disease (CAD) and then using this information to select an appropriate imaging test, the exact choice depending to some extent on local expertise.1
In November 2016, NICE published an update of CG95, and made substantial changes to the guidelines. As well as eliminating the clinically derived pre-test probability as a means of directing further investigations, NICE brought computed tomography (CT) coronary angiography (CTCA) to the forefront as the first-line investigation for all patients with new-onset typical or atypical anginal chest pain, and non-anginal chest pain with potentially ischaemic electrocardiogram (ECG) changes. Functional imaging is reserved for the assessment of patients with chest pain and known CAD, and for patients where CTCA is non-diagnostic or has shown CAD of uncertain significance.2
The primary reason for extending the role of CTCA as a first-line test relates to its unparalleled ability to exclude the presence of obstructive CAD when compared with invasive coronary angiography (ICA) as a gold standard. From this perspective, CTCA has been shown to have a diagnostic sensitivity of 89%. CTCA, therefore, has an excellent diagnostic accuracy for the identification of CAD, with a high negative-predictive value.3
CTCA
The justification for CTCA, based on its ability to predict the anatomical findings on ICA, remains controversial, but CTCA has an increasing evidence base for more clinically relevant end points. Two large ‘test and treat’ randomised-controlled trials, SCOT-HEART (Scottish COmputed Tomography of the HEART) and PROMISE (PROspective Multicentre Imaging Study for Evaluation of chest pain), have supported the value of CTCA as a first-line investigation for patients with suspected angina pectoris.
The SCOT-HEART trial randomised 4,146 outpatients with suspected angina due to CAD to standard investigation (mainly exercise ECG) or standard investigation plus CTCA.4 Follow-up at five years has demonstrated a significant reduction in CAD death or nonfatal myocardial infarction (MI) in the CTCA group (2.3% vs. 3.9% in the standard investigation group; p=0.004). This may reflect a greater use of preventive medication (19.4% vs. 14.7%; 95% confidence interval [CI] 1.19 to 1.65). Early rates of ICA and coronary revascularisation were higher in the CTCA group, but the difference had disappeared by five years.5
The PROMISE trial was a US-based study and recruited 10,003 symptomatic stable outpatients randomised to undergo either anatomical assessment with CTCA or functional testing. Although this large clinical trial found no difference in outcome between the arms overall at one year, over a three-year follow-up patients with diabetes were 62% less likely to have MI or death from a cardiovascular cause if they underwent CTCA initially than if they underwent functional testing. There was also an increase in the proportion of patients undergoing ICA and coronary revascularisation in the CTCA group compared with the functional imaging group.6,7 These findings are not surprising given that a major strength of CTCA is its ability to identify anatomical stenoses. However, its ability to identify flow-limiting stenosis is limited. To this end, there is evidence to suggest that the specificity of this strategy may improve when functional assessment by fractional flow reserve (FFR) from a standard CTCA acquisition (FFRCT) is adopted.8,9
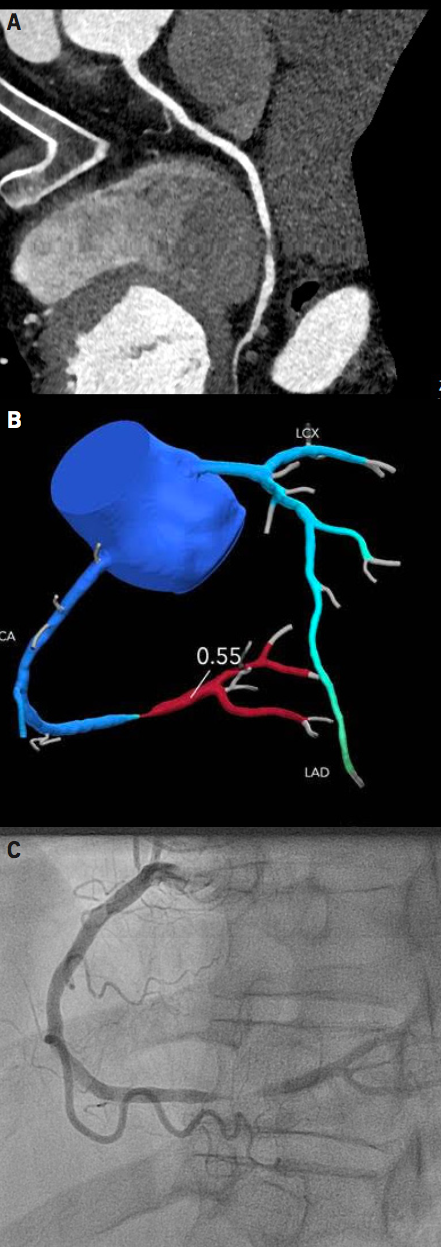
HeartFlow®
In recent years, HeartFlow®, a company based in Redwood City, California, USA, has developed software using advanced computational fluid dynamics (CFD) to predict FFR from standard CTCA datasets (FFRCT) (figure 1). Initial studies with early versions of the analysis process and technology – DISCOVER-flow (Diagnosis of Ischemia-Causing Stenoses Obtained Via Noninvasive Fractional Flow Reserve) and DeFACTO (Determination of Fractional Flow Reserve by Anatomic Computed Tomographic Angiography) – compared FFRCT with invasive FFR and demonstrated promising results.10,11 The subsequent NXT (Analysis of Coronary Blood Flow Using CT Angiography: Next Steps) study of the approach taken into clinical use evaluated the diagnostic accuracy of FFRCT in 251 patients with suspected CAD using invasive FFR as a gold standard. The per-patient diagnostic accuracy of FFRCT was significantly higher for FFRCT than for CTCA alone (81% vs. 53%, p<0.001). This was primarily due to a much higher specificity (79% vs. 34%, p<0.001). Sensitivity was similarly high for both FFRCT and CCTA (86% vs. 94%, p=not significant).12 The NXT study data led to clearance of FFRCT by the US Food and Drug Administration (FDA) in 2014, making HeartFlow® the market leader in this arena.
The HeartFlow® process
The HeartFlow® process first requires patients to undergo CTCA at their local hospital. The minimum stipulated requirement is for a scanner with at least 64 detectors, and for image acquisition in accordance with Society of Cardiovascular Computed Tomography Guidelines.13 Anonymised data are then transferred electronically to HeartFlow® via the Cloud.
Using advanced algorithms that incorporate artificial intelligence, a three-dimensional model of the patient’s coronary arteries is created and then edited. Calculation of FFRCT is performed throughout the coronary tree using CFD. The algorithms are able to take into account myocardial resistance, and then predict changes in coronary resistance during hyperaemia. By applying the Navier-Stokes equations, which describe how the pressure, velocity, density and temperature of a moving fluid are related, coronary flow can be predicted during hyperaemia. No vasodilator drug is required in this process, as estimates of virtual hyperaemia are used for the calculation. A form of artificial intelligence known as deep learning is used to optimise and enhance performance continually. Results are returned to the referrer via the Cloud within less than five hours.14,15
Evidence-base for the clinical utility of HeartFlow® FFRCT
The initial clinical study of FFRCT was the PLATFORM (Prospective LongitudinAl Trial of FFRCT: Outcome and Resource iMpacts) study, which was published in 2018. This was a prospective cohort study of 585 patients. Consecutive patients with stable chest pain of recent onset were managed by either usual testing (n=287) or CTCA (n=297) with selective FFRCT (submitted in 201, analysed in 177). Patients were classified according to whether initial ICA (n=380) or non-invasive testing (n=204) had been planned, and the primary end point of the PLATFORM was related only to the invasive group. There was a parallel exploratory ‘non-invasive’ group enrolled, which did not contribute to the primary end point and was not powered for clinical outcomes.16
The primary end point of PLATFORM, related to the invasive group only, showed that the proportion undergoing ICA but finding only unobstructed coronary arteries was reduced by 83% (from 73% to 12% absolute) in the CTCA/FFRCT arm, and that planned ICA was avoided in 61% of patients who underwent CTCA with FFRCT: 90-day costs were correspondingly lower in the FFRCT patients. Moreover, the use of FFRCT was safe. Of the 117 patients whose ICA was deferred, there were no adverse cardiac events after one year. The rate of cardiac events in either group was very low.
Because rates of death and MACE are very low in patients with stable CAD, one could argue about the fundamental merits of referring such patients for advanced imaging. With that in mind, the ADVANCE (Assessing Diagnostic Value of Non-invasive FFRCT in Coronary Care) registry explored the utility and safety of this new diagnostic pathway in much larger numbers of patients and centres in real-world routine clinical practice. This was a large international prospective examination of using a CTCA and FFRCT diagnostic pathway in 5,083 patients at 38 centres in North America, Europe, and Japan with symptomatic CAD. The study enrolled patients who had CAD apparent on CTCA. Over one-year follow-up, the vast majority of those who had a negative FFRCT analysis were managed without invasive testing and had extremely good clinical outcomes, with adverse cardiac events seen in only 0.2%.17 While there was a low rate of events in all patients in this registry, there was less revascularisation and a trend towards lower MACE and significantly lower cardiovascular death or MI in patients with a negative FFRCT when compared with patients with a positive FFRCT.
CTCA with FFRCT compared with other approaches to non-invasive investigation
The clinical literature strongly suggests that CTCA with FFRCT is preferable to first-line ICA, but current international guidelines do not yet endorse the latter approach in the vast majority of patients.18 A more important issue is how CTCA with FFRCT compares with more traditional non-invasive functional testing, but framing the relevant question is not easy. Applying the logic of the HeartFlow® business model, FFRCT cannot be regarded as a simple adjunct to routine CTCA analysis. This is because, while no additional patient imaging is required, a significant cost is incurred, which may exceed that of performing a second in-house stress-based test. In that sense, FFRCT should be regarded as a distinct second-line functional test for a subset of patients following first-line anatomical CTCA, in competition with other stress-based tests that could be used in the same situation.
PACIFIC (Prospective Comparison of Cardiac PET/CT, SPECT/CT Perfusion Imaging and CT Coronary Angiography With Invasive Coronary Angiography) was a prospective head-to-head study in 208 patients which compared CTCA, 99mTc-tetrofosmin single-photon emission CT (SPECT) perfusion imaging and 15O-H2O positron-emission tomography (PET) perfusion imaging with a functional gold standard of invasive FFR on a per-vessel basis.19 In a recently published substudy, the performance of FFRCT, SPECT and PET was compared by means of area under the receiver-operating characteristic curve (AUC) using invasive FFR as a gold standard. FFRCT analysis was successful in 87% of patients, and had a significantly higher sensitivity (90%) than either SPECT (42%; p<0.001) or PET (81%; p=0.03). Its specificity (86%) was similar to that of PET (76%; p=not significant), though lower than that of SPECT (97%; p<0.001).20
It is perhaps not surprising that FFRCT outperformed SPECT and PET in PACIFIC, given that it directly models the gold standard, invasive FFR. Invasive FFR has become popular with interventional cardiologists because studies, such as FAME (Fractional Flow Reserve Versus Angiography for Multivessel Evaluation), FAME-2 and DEFER, suggested that the outcome of patients with FFR-positive (but not FFR-negative) disease can be improved by percutaneous coronary intervention (PCI). This benefit is driven almost entirely by a reduction in unscheduled urgent revascularisations rather than ‘hard’ clinical end points.21-23 When considering invasive FFR as a gold-standard test, it is important to take the following factors into consideration:
- FFR was itself originally validated against stress-based imaging tests, such as SPECT, creating a logical paradox.
- There is an extensive literature demonstrating the ability of functional imaging to predict hard cardiac end points, whereas few such data exist for FFR.
- FFR assesses only the significance of epicardial CAD, whereas PET and SPECT integrate both epicardial and microvascular function.
Cost
A retrospective analysis by Rajani et al. projected the potential cost saving that would be accrued if FFRCT was used upfront following assessment of patients at a rapid-access chest pain clinic, instead of care guided by the original 2010 NICE guidelines. When NHS tariffs were applied, it was predicted that the use of CTCA with FFRCT as the primary diagnostic test would result in a saving of £200 per patient. This is driven by a 48% reduction in the number of ICAs in the 60–90% pre-test probability group who would now have CTCA, and a 49% reduction in the number of PCI procedures (invasive FFR), together with a reduction in the number of adverse events of 4%.24
In February 2017, NICE released a medical technology guidance (MTG32), which concluded that HeartFlow® FFRCT is “non-invasive and safe, and has a high level of diagnostic accuracy”. In addition, the evaluation concluded that FFRCT is cost-effective and predicted that its use could lead to a saving of £214 per patient. NICE projected that adoption of this technology could save the NHS in England a minimum of £9.1 million by 2022.25 It was suggested that FFRCT could be used in place of a second-line functional test in patients being investigated using the CG95 pathway.
MTG32 can be criticised on a number of levels. FFRCT is a single-vendor technology, and a non-specialist NICE committee accepted the submission from HeartFlow® with rather little criticism. The cost-effectiveness analysis was based on NICE’s original CG95 guideline from 2010, and, hence, most of the cost-savings can be assumed to derive from avoiding first-line ICA in the majority of patients with a pre-test probability of 60–90%. Finally, NICE’s Medical Technology Advisory Committee might not be entitled to make recommendations about the incorporation of FFRCT into the revised version of CG95. Regardless of these reservations, HeartFlow® FFRCT has been enthusiastically endorsed by NHS Improvement, which has made it one of only four technologies eligible for an Innovation and Technology Payment in 2018/19.
Pros and cons
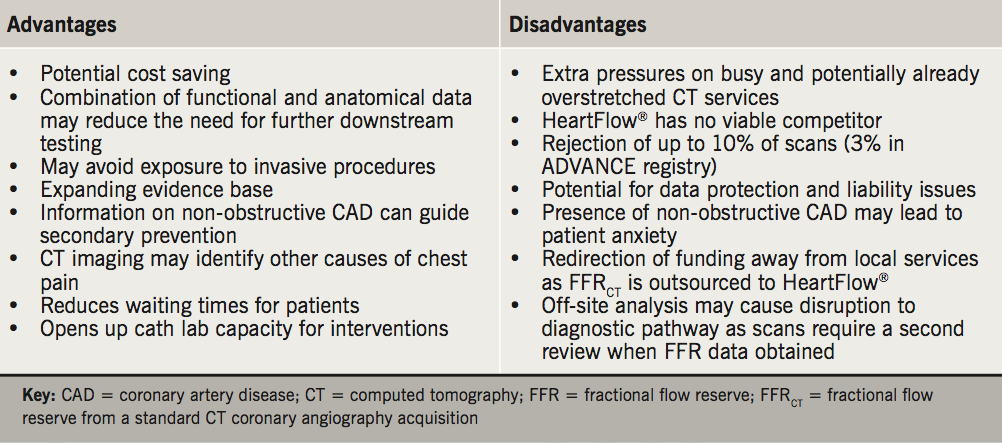
A single non-invasive investigation for CAD that provides both anatomical and functional data has great appeal for the investigation of patients with stable chest pain. While HeartFlow® FFRCT added to CTCA shows a lot of promise, there are a few limitations that need to be considered (table 1).
In the DeFACTO, NXT and PLATFORM studies, 11%, 13% and 12% of CTCA datasets, respectively, were unsuitable for analysis by FFRCT. This was mainly due to technical issues, such as phase misalignment artefact, coronary motion artefact and extensive coronary calcification. These can largely be overcome with attention to acquisition technique and patient preparation, as evidenced by the ADVANCE registry of over 5,000 patients in which the rejection rate was only 3%. HeartFlow® remains under evaluation by the NHS with more than 30 hospitals adopting HeartFlow® in 2018.
The future
Results from the SYNTAX III Revolution trial, published in 2018, demonstrated very promising results for the potential application of FFRCT in the context of the heart team to aid decision-making with regards to PCI versus surgery (CABG). In this study, heart teams were randomised to two arms with heart team A using only non-invasive investigations, i.e. FFRCT, while heart team B used conventional investigations, i.e. ICA. The investigators found that the treatment decisions between the two randomised heart teams were in almost perfect agreement (Cohen’s kappa 0.82), highlighting the potential of CTCA to not only diagnose coronary disease, but also assist in subsequent heart team decision-making.26,27
Another potential future application of FFRCT is its potential utility in the management of patients with acute coronary syndrome (ACS). One recent study demonstrated that change in FFRCT across a lesion (ΔFFRCT) may have an important role in predicting ACS risk in non-obstructive lesions. This study showed that specific features on CTCA, such as ΔFFRCT, low-attenuation plaque and plaque volume, showed the highest ability in ACS prediction (AUC 0.725, 95%CI 0.724 to 0.727). However, of these features, ΔFFRCT was the strongest predictor of ACS risk in non-obstructive lesions and, as such, may in the future improve the non-invasive prediction of ACS risk in non-obstructive lesions.28
Conclusion
The addition of FFRCT to standard CTCA provides a simultaneous anatomical and functional assessment of CAD. FFRCT also offers a potential cost saving by reducing the number of patients referred for ICAs, and in doing so also reduces risk for patients. Initial data suggest that FFRCT improves the accuracy of CTCA. Guidance from NICE supports the use of FFRCT in England, but it remains to be seen if its routine use will be eventually commissioned by the NHS.
Key messages
- The National Institute for Health and Care Excellence (NICE) published an update of the chest pain of recent onset guideline (CG95) bringing computed tomography coronary angiography (CTCA) to the forefront as the first-line investigation
- CTCA has been shown to have a sensitivity of 89% for the detection of obstructive coronary artery disease (CAD)
- CTCA has a relatively low positive-predictive value of 48%, and studies have shown that its use is associated with higher rates of referral for invasive coronary angiography compared with functional imaging
- A single non-invasive investigation for CAD that provides both anatomical and functional data, such as fractional flow reserve (FFR) from a standard CTCA acquisition (FFRCT) with HeartFlow® FFRCT, would be a great step forward in the investigation of patients with stable chest pain
- The advantages of HeartFlow® FFRCT, such as potential cost savings and a reduction in the need for further downstream testing, need to be weighed against disadvantages, including rejection of some scans due to low image quality (3% in the ADVANCE registry of over 5,000 patients)
Conflicts of interest
None declared.
Funding
The authors have received no funding for this article. Medinews (Cardiology) Limited, the publishers of the BJC, approached HeartFlow® after this article was accepted for publication, to request funding for making it available for CPD accreditation.
Acknowledgement
We acknowledge the support and comments of Dr Campbell Rogers, HeartFlow®, who checked the article for technical accuracy only.
References
1. National Institute for Health and Care Excellence. Chest pain of recent onset: assessment and diagnosis of recent onset chest pain or discomfort of suspected cardiac origin. CG95. London: NICE, 2010. Available from: https://www.nice.org.uk/guidance/CG95
2. National Institute for Health and Care Excellence. Chest pain of recent onset: assessment and diagnosis. London: NICE, 2016. Available from: https://www.nice.org.uk/guidance/CG95
3. Miller JM, Rochitte CE, Dewey M et al. Diagnostic performance of coronary angiography by 64-row CT. N Engl J Med 2008;359:2324–36. https://doi.org/10.1056/NEJMoa0806576
4. Newby DE, Williams MC, Flapan AD et al. Role of multidetector computed tomography in the diagnosis and management of patients attending the rapid access chest pain clinic, the Scottish computed tomography of the heart (SCOT-HEART) trial: study protocol for randomized controlled trial. Trials 2012;13:184. https://doi.org/10.1186/1745-6215-13-184
5. The SCOT-HEART Investigators. Coronary CT angiography and 5-year risk of myocardial infarction. N Engl J Med 2018;379:924–33. https://doi.org/10.1056/NEJMoa1805971
6. Douglas PS, Hoffmann U, Patel MR et al. Outcomes of anatomical versus functional testing for coronary artery disease. N Engl J Med 2015;372:1291–300. https://doi.org/10.1056/NEJMoa1415516
7. Sharna A, Coles A, Sekaran NK et al. Stress testing versus CT angiography in patients with diabetes and suspected coronary artery disease. J Am Coll Cardiol 2019;73:893–902. https://doi.org/10.1016/j.jacc.2018.11.056
8. Lu MT, Ferencik M, Roberts RS et al. Non-invasive FFR derived from coronary CT angiography: management and outcomes in the PROMISE trial. JACC Cardiovasc Imaging 2017;10:1350–8. https://doi.org/10.1016/j.jcmg.2016.11.024
9. Budoff MJ, Dowe D, Jollis JG et al. Diagnostic performance of 64-multidetector row coronary computed tomographic angiography for evaluation of coronary artery stenosis in individuals without known coronary artery disease: results from the prospective multicenter ACCURACY (Assessment by Coronary Computed Tomographic Angiography of individuals undergoing invasive coronary angiography) trial. J Am Coll Cardiol 2008;52:1724–32. https://doi.org/10.1016/j.jacc.2008.07.031
10. Koo BK, Erglis A, Doh JH et al. Diagnosis of ischemia-causing coronary stenoses by noninvasive fractional flow reserve computed from coronary computed tomographic angiograms. Results from the prospective multicenter DISCOVER-FLOW (Diagnosis of Ischemia-Causing Stenoses Obtained Via Noninvasive Fractional flow Reserve) study. J Am Coll Cardiol 2011;58:1989–97. https://doi.org/10.1016/j.jacc.2011.06.066
11. Min JK, Leipsic J, Pencina MJ et al. Diagnostic accuracy of fractional flow reserve from anatomic CT angiography. JAMA 2012;308:1237–45. https://doi.org/10.1001/2012.jama.11274
12. Nørgaard BL, Leipsic J, Gaur S et al. Diagnostic performance of noninvasive fractional flow reserve derived from coronary computed tomography angiography in suspected coronary artery disease: the NXT trial (analysis of coronary blood flow using CT angiography: next steps). J Am Coll Cardiol 2014;63:1145–55. https://doi.org/10.1016/j.jacc.2013.11.043
13. Abbara S, Blanke P, Maroules CD et al. SCCT guidelines for the performance and acquisition of coronary computed tomographic angiography: a report of the society of Cardiovascular Computed Tomography Guidelines Committee: Endorsed by the North American Society for Cardiovascular Imaging (NASCI). J Cardiovasc Comput Tomogr 2016;10:435–49. https://doi.org/10.1016/j.jcct.2016.10.002
14. Taylor CA, Fonte TA, Min JK. Computational fluid dynamics applied to cardiac computed tomography for noninvasive quantification of fractional flow reserve: scientific basis. J Am Coll Cardiol 2013;61:2233–41. https://doi.org/10.1016/j.jacc.2012.11.083
15. Min JK, Taylor CA, Achenbach S et al. Noninvasive fractional flow reserve derived from coronary CT angiography: clinical data and scientific principles. JACC Cardiovasc Imaging 2015;8:1209–22. https://doi.org/10.1016/j.jcmg.2015.08.006
16. Hlatky MA, De Bruyne B, Pontone G et al. Quality-of-life and economic outcomes of assessing fractional flow reserve with computed tomography angiography: PLATFORM. J Am Coll Cardiol 2015;66:2315–23. https://doi.org/10.1016/j.jacc.2015.09.051
17. Patel MR, Nørgaard DL, Fairbairn TA et al. 1-year impact on medical practice and clinical outcomes of FFRCT. JACC: Cardiovascular Imaging 2019;online first. https://doi.org/10.1016/j.jcmg.2019.03.003
18. Montalescot G, Sechtem U, Achenbach S et al. 2013 ESC guidelines on the management of stable coronary artery disease: the Task Force on the management of stable coronary artery disease of the European Society of Cardiology. Eur Heart J 2013;34:2949–3003. https://doi.org/10.1093/eurheartj/eht296
19. Danad I, Rajimakers PG, Driessen RS et al. Comparison of coronary CT angiography, SPECT, PET, and hybrid imaging for diagnosis of ischemic heart disease determined by flow fractional reserve. JAMA Cardiol 2017;2:1100–07. https://doi.org/10.1001/jamacardio.2017.2471
20. Driessen RS, Danad I, Stuijfzand WJ et al. Comparison of coronary computed tomography angiography, fractional flow reserve, and perfusion imaging for ischemia diagnosis. J Am Coll Cardiol 2019;73:161–73. https://doi.org/10.1016/j.jacc.2018.10.056
21. De Bruyne B, Pijls NH, Kalesan B et al. Fractional flow reserve-guided PCI versus medical therapy in stable coronary disease. N Engl J Med 2012;367:991–1001. https://doi.org/10.1056/NEJMoa1205361
22. Pijls NH, Fearon WF, Tonino PA et al. Fractional flow reserve versus angiography for guiding percutaneous coronary intervention in patients with multivessel coronary artery disease: 2 year follow-up the FAME (Fractional Flow Reserve versus Angiography for Multivessel Evaluation) study. J Am Coll Cardiol 2010;56:177–84. https://doi.org/10.1016/j.jacc.2010.04.012
23. Pijls NH, van Schaardenburgh P, Manoharan G et al. Percutaneous coronary intervention of functionally nonsignificant stenosis: 5-year follow-up of the DEFER study. J Am Coll Cardiol 2007;49:2105–11. https://doi.org/10.1016/j.jacc.2007.01.087
24. Rajani R, Webb J, Marciniak A et al. Comparative efficacy testing – fractional flow reserve by coronary computed tomography for the evaluation of patients with stable chest pain. Int J Cardiol 2015;183:173–7. https://doi.org/10.1016/j.ijcard.2015.01.035
25. National Institute for Health and Care Excellence. HeartFlow® FFRCT for estimating fractional flow reserve from coronary CT angiography. London: NICE, 2016. Available from: https://www.nice.org.uk/guidance/mtg32/documents/consultation-document
26. Cavalcante R, Onuma Y, Sotomi Y et al. Non-invasive heart team assessment of multivessel coronary disease with coronary computed tomography angiography based on SYNTAX score II treatment recommendations: design and rationale of the randomised SYNTAX III Revolution trial. EuroIntervention 2017;12:2001–08. https://doi.org/10.4244/EIJ-D-16-00612
27. Collet C, Onuma Y, Andreini D et al. Coronary computed tomography angiography for heart team decision-making in multivessel coronary artery disease. Eur Heart J 2018;39:3689–98. https://doi.org/10.1093/eurheartj/ehy581
28. Park J, Lee JM, Koo B-K et al. Relevance of anatomical, plaque, and hemodynamic characteristics of non-obstructive coronary lesions in the prediction of risk for acute coronary syndrome. Eur Radiol 2019;online first. https://doi.org/10.1007/s00330-019-06221-9
