The authors describe a case of hyperkalaemia in a patient receiving trimethoprim.
Case report
An 86-year-old man presented to Accident and Emergency (A&E) feeling generally unwell and increasingly confused. Following an assessment, the doctors diagnosed a urinary tract infection (confirmed on dipstick) and discharged him with a standard course of trimethoprim. After a few days, he was readmitted with falls and increasing lethargy.
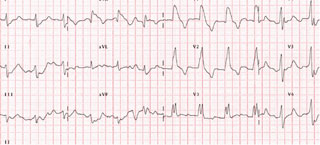
His electrocardiogram (ECG) on admission showed widened QRS complexes and significantly prolonged QT/QTc intervals (figure 1).
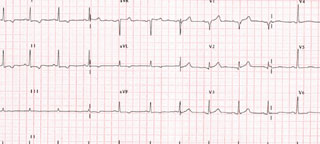
Blood tests demonstrated a potassium level of 9.2 mmol/L, having been 5.5 mmol/L on his first presentation. This was subsequently treated and his potassium level returned to normal. Repeat ECGs showed resolution of the initial abnormalities but unmasked non-specific ST–T changes, which also soon resolved (figure 2). On review of his history, the patient was not on any medications that were nephrotoxic nor any hyperkalaemic agents. He had a pre-morbid history of cognitive impairment, mild chronic obstructive pulmonary disease (COPD) and significant but stable renal impairment (estimated glomerular filtration rate [eGFR] 27 ml/min).
Discussion
Trimethoprim is a heterocyclic weak base that is structurally related to the potassium-sparing diuretic amiloride.1 Amiloride and trimethoprim block the reabsorption of sodium in the distal nephron through the inhibition of luminal sodium channels, which subsequently impairs renal potassium secretion.2 Hyperkalaemia due to trimethoprim typically affects elderly patients administered standard oral dosages, even in the presence of a normal serum creatinine level.3
Serum potassium concentrations should be monitored in patients with renal insufficiency who receive this drug, and the dose of trimethoprim should be titrated according to glomerular filtration rate.4 Concurrent angiotensin-converting enzyme (ACE) inhibitor therapy may increase the risk of hyperkalaemia. The prognosis is favourable with standard therapy for hyperkalaemia and withdrawal of trimethoprim.
The three classic ECG changes associated with raised potassium levels are:5
- Tall peaked T-waves (as a result of accelerated terminal hyperpolarisation)
- Reduction in amplitude and eventual loss of the P-wave
- Widening of the QRS interval (due to depression of conduction between adjacent
cardiac myocytes).
Non-specific ST–T changes that can mimic ischaemic pathology may also be recognised in hyperkalaemic patients.6
Conclusion
Trimethoprim should be prescribed with vigilance in patients with impaired renal function, the elderly, and those with a predisposition to hyperkalaemia.
Conflict of interest
None declared.
References
- Choi MJ, Fernandez PC, Patnaik AL et al. Brief report: trimethoprim-induced hyperkalemia in a patient
with AIDS. N Engl J Med 1993;328:703–06. - Velazquez H, Perazella MA, Wright FS et al. Renal mechanism of
trimethoprim-induced hyperkalemia. Ann Intern Med 1993;119:296–301. - Marinella MA. Trimethoprim-induced hyperkalemia: an analysis of reported
cases. Gerontology 1999;45:209–12. - Perazella MA. Trimethoprim-induced hyperkalaemia: clinical data, mechanism,
prevention and management. Drug Safety 2000;22:227–36. - Webster A, Brady W, Morris F. Recognising signs of danger: ECG changes
resulting from an abnormal serum potassium concentration. Emerg Med J 2002;19:74–7. - Jowett NI, Thompson DR. Comprehensive coronary care. London: Elsevier
Science Ltd, 2003.
