Much effort has been expended assessing the relative merits of percutaneous coronary intervention (PCI) and coronary artery bypass graft (CAGB) surgery. Much less energy has been directed towards understanding the potential of these two interventions for causing additional myocardial damage during the procedure and the means to avoid this injury.
This review examines the impact of myocardial injury in elective PCI and CABG, principles of myocardial protection, and their efficacy in current coronary revascularisation. The objective of every coronary revascularisation should be a technically perfect result without producing myocardial damage. A patent graft that perfuses an area of myocardium with numerous pockets of myocyte necrosis serves no useful purpose.
Myocardial damage induced by PCI
Most of the clinical research studies on percutaneous coronary intervention (PCI) have focused on angiographic and clinical outcomes. Cardiomyocyte necrosis associated with impaired flow to side branches or occlusion after coronary stenting does occur, but has received little attention. Atherosclerotic debris can plug the downstream coronary microcirculation and ischaemia-reperfusion injury may occur after revascularisation of occluded vessels. Creatinine kinase myocardial band (CK-MB) and troponin T levels, and electrocardiogram (ECG) evidence of infarction have been used as markers for post-procedure cardiomyocyte necrosis. CK-MB elevation, which occurs in approximately 25% of patients, and troponin T release, which occurs in up to 50% of patients,1 have been shown to be predictors of poor late outcome.2,3
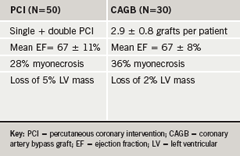
Magnetic resonance imaging (MRI) has been used to assess myocardial damage. In a recent study, Selvanayagam and co-workers4 correlated post-PCI troponin elevations with the volume of myocardial tissue destruction using delayed-enhancement MRI. This was a low-risk group of 50 patients in whom a single- or double-vessel PCI was planned with a mean left ventricular (LV) ejection fraction of 67 ± 11%. Some 28% of the patients had evidence of procedure-related myocardial necrosis resulting in a loss of 5.0 ± 4.8% of total ventricular mass (table 1). There was no statistically significant adverse effect on global LV function. Myocardial injury occurred in one or both of two distinct sites:
- An area of the apical myocardium apparently related to embolisation of solid matter during left anterior descending (LAD) coronary artery balloon dilatation plus stenting.
- The mid-ventricular myocardium adjacent to the new stent.
In an attempt to minimise PCI-induced myocyte damage, mechanical devices including balloons distal to a central aspiration device, and a polyurethane filter bag contained on a radio-opaque loop to trap embolic debris, have been introduced. In a randomised trial of 500 patients,5 the device entrapped embolic debris in 73% of cases, but there was no information about any reduction in cardiomyocyte necrosis. This may be because these devices do not eliminate side branch occlusion and have no effect on PCI-induced endothelial dysfunction.
Myocardial damage induced by surgery
The objective of every operation should be a technically perfect result without producing myocardial damage. From the practical viewpoint the prerequisites for optimal protection are uniform and adequate distribution of a protective agent, excellent visualisation of the operative field, a simple technique that does not distract the surgeon, and optimal protection of the brain and kidneys. The consequences of inadequate myocardial protection range from low cardiac output states leading to multi-organ failure, increased mortality and prolonged hospital stay; to subtle degrees of damage causing delayed myocardial fibrosis. In patients with pre-operative impaired LV function, the margins of functional reserve are narrow. Recently much debate has occurred about the relative merits of on-pump and off-pump surgery and whether the avoidance of a period of extracorporeal perfusion is beneficial to the myocardium. This debate remains unresolved, but in expert hands equivalent results can be obtained by both techniques.
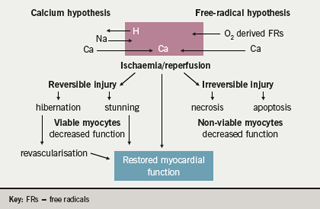
Myocardial stunning and cardiomyocyte necrosis associated with ischaemia/reperfusion injury results from the attenuation or cessation of coronary blood flow, such that oxygen delivery to the myocardium is insufficient to meet basal myocardial requirements to preserve cellular membrane stability and viability. Recovery involves the resumption of normal oxidative metabolism and the restoration of myocardial energy reserves, the reversal of ischaemia-induced swelling and loss of membrane ion gradients, and repair of damaged cell organelles, such as the mitochondria and the sarcoplasmic reticulum (figure 1). There are two major hypotheses; increases in intracellular calcium and the accumulation of reactive oxygen species (ROS) causing the sarcolemmal peroxidation of the cell phospholipid layer, leading to the loss of cellular integrity and facilitating calcium entry. After the aortic cross-clamp is removed, the cardiomyocyte may function normally, be stunned, or become dysfunctional from either necrosis or apoptosis.
Post-operative myocardial stunning
There is plenty of clinical evidence to indicate that all patients undergoing coronary artery bypass graft (CABG) experience varying degrees of myocardial stunning. This is defined as a form of post-ischaemic dysfunction where there is normal myocardial perfusion, preserved contractile reserve, and delayed but full recovery of function. This occasionally requires inotropic support, which after hours or days resolves and leaves behind no objective evidence of myocardial infarction.6 However, there are significant disadvantages to the use of inotropes. Experimental studies have shown clearly that increasing doses of catecholamines will cause cardiomyocyte necrosis as the myocardial oxygen consumption exceeds the heart’s capacity to increase coronary blood flow. There is recent evidence that therapeutic levels of inotropic support in the post-ischaemic heart increases intracellular calcium and subsequent apoptosis resulting in cell death.7 This is probably critical in the post coronary artery surgical patient if there are areas of the heart that have not been adequately revascularised.
In a study comparing PCI and CABG outcomes, there was a greater incidence of CK-MB leak from CABG patients than PCI patients.8 However, when the criterion for significant myocardial injury was changed to 10-times normal, there was no difference and the CABG patients had a significant, but small, increase in three-year survival. In the Arterial Revascularisation Therapies Study (ARTS) there was a direct relationship between CK-MB and long-term outcomes.9 At one year the worst adverse outcomes as defined by the incidence of major adverse clinical events (MACE) occurred in patients with CK-MB levels greater than five-times normal. In a more recent study from the UK using troponin levels and delayed-enhancement MRI, Selvanayagam and co-workers10 reported a 36% incidence of myocyte necrosis and a 2% loss of LV mass.
The principles of myocardial protection
The basic principles of myocardial protection are:
- rapid cardiac arrest, since the myocardial oxygen stores are depleted within six seconds as oxidative metabolism switches from aerobic to anaerobic metabolism
- hypothermia to decrease myocardial oxygen consumption and prevent the depletion of high energy phosphate
- the avoidance of myocardial oedema.
Most surgeons use blood cardioplegia to provide additional substrate oxygen, improved buffering and anti-oxidants; and they employ a combination of antegrade and retrograde delivery systems. Despite numerous reports, there have been no definitive prospective studies that narrow the techniques enough to allow universal adoption of one particular technique.
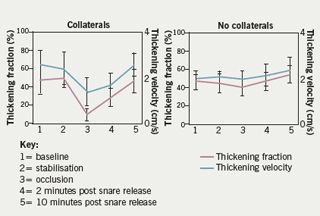
The avoidance of cardiopulmonary bypass by the use of off-pump techniques has not resulted in a clear benefit in terms of reduced mortality or reduced incidence of peri-operative infarction. However, most observers have reported a significant reduction in troponin T release. Although there is enormous enthusiasm for off-pump coronary artery surgery in some quarters, the overall uptake has been about 20% across the board. Using high-fidelity LV pressure recordings combined with 2-D echo recordings from peri-operative transoesophageal echocardiography, we have shown remarkable tolerance of the myocardium to occlusion of the LAD artery for periods of up to 15 minutes (figure 2). In the presence of collaterals from a dominant right coronary artery, there is very little change in myocardial contractility as revealed by analysis of anterior wall thickening. In the absence of collaterals, normal wall thickening is restored within 10 minutes.11
It has been known for some time that short bursts of ischaemia followed by reperfusion protect the myocardium against subsequent longer ischaemic insults, as evidenced by reduction of cardiac troponin T release. This phenomenon, called ischaemic pre-conditioning, can protect the human heart from ischaemic injury during coronary bypass surgery but is not practised routinely because repeated clamping of the aorta is likely to induce cerebral emboli.12 Pharmacological pre-conditioning may also confer benefit but this remains unproven.
Conclusion
Over the last 50 years in coronary artery surgery, intra-operative myocardial damage from inadequate protection has decreased progressively, despite the demographic changes that have brought older and sicker patients forward for myocardial revascularisation. Nevertheless, both CABG and PCI are associated with a small but definite incidence of myocardial injury. Myocardial protection strategies are less developed in PCI and the need for repeat revascularisation procedures is more frequent than after surgery. We must rise to the challenge by finding ways and means of providing adequate revascularisation while inducing the least amount of cardiomyocyte injury for our patients.
Conflict of interest
None declared.
Key messages
- Heart muscle may be damaged during revascularisation
- Both percutaneous intervention and surgery can result in damage
- New methods are being developed to minimise myocardial cell injury
References
- Gomes WJ, Buffolo E. Coronary stenting and inflammation: implications for further surgical and medical treatment. Ann Thorac Surg 2006;81:1918–25.
- Kini AS, Lee P, Marmur JD et al. Correlation of postpercutaneous coronary intervention creatinine kinase-MB and Troponin l elevation in predicting mid-term mortality. Am J Cardiol 2004;93:18–23.
- Stone GW, Mehran R, Dangas G, Lansky AJ, Kornowski R, Leon MB. Differential impact on survival of electrocardiographic Q-wave versus enzymatic myocardial infarction after percutaneous intervention: a device-specific analysis of 7147 patients. Circulation 2001;104:642–7.
- Selvanayagam JB, Porto I, Channon K et al. Troponin elevation after percutaneous coronary intervention directly represents the extent of irreversible myocardial injury: insights from cardiovascular magnetic resonance imaging. Circulation 2005;111:1027–32.
- Stone GW, Webb J, Cox DA. Distal microcirculatory protection during percutaneous coronary intervention in acute ST-segment elevation myocardial infarction. JAMA 2005;293:1063–72.
- Kloner RA, Przklenk K, Kay GL. Clinical evidence for stunned myocardium after coronary artery bypass surgery. J Cardiac Surg 1994;9(suppl):397–402.
- Stamm C, Friehs I, Cowan DB et al. Dopamine treatment of postischemic contractile dysfunction rapidly induces calcium-dependent pro-apoptotic signalling. Circulation 2002;106(12 suppl 1):I290–8.
- Brener SJ, Lytle BW, Schneider JP, Ellis SG, Topol EJ. Association between CK-MB elevation after percutaneous or surgical revascularisation and three year mortality. J Am Coll Cardiol2002;40:1961–7.
- Costa MA, Carere RG, Lichtenstein SV et al. Incidence, predictors and significance of abnormal cardiac enzyme rise in patients treated with bypass surgery in the arterial revascularisation therapies study (ARTS). Circulation 2001;104: 2689–93.
- Selvanayagam JB, Pigott D, Balacumaraswami L, Petersen SE, Neubauer S, Taggart DP. Relationship of irreversible myocardial injury to Troponin l and creatinine-kinase-MB elevation after coronary artery bypass surgery: insights from cardiovascular magnetic resonance imaging. J Am Coll Cardiol 2005;45:629–31.
- Koh TW, Carr-White GS, DeSouza AC, Ferdinand FC, Pepper JR, Gibson DG. Effect of coronary occlusion on left ventricular function with and without collateral supply during beating heart coronary artery surgery. Heart 1999;81:285–91.
- Teoh LKK, Grant R, Hulf JA, Pugsley WB, Yellon DM. The effect of preconditioning (ischemic and pharmacological) on myocardial necrosis following coronary artery surgery. Cardiovasc Res2002;53:175–80.

