Identifying targets in the thrombosis pathway Figure 1 summarises the central role of platelets in the genesis of thrombosis.1 The platelet is initially activated in response to shear stress, events … Continue reading Aspirin – scope and limitations
Identifying targets in the thrombosis pathway
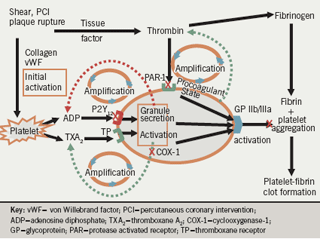
Figure 1 summarises the central role of platelets in the genesis of thrombosis.1 The platelet is initially activated in response to shear stress, events such as percutaneous coronary intervention (PCI) or plaque rupture, and the release of local agonists and exposure of the subendothelial components to flowing blood. Tissue factor ‘lights the fire’ by producing minute quantities of thrombin which then amplify the process. Binding of platelets to collagen and von Willebrand factor also leads to platelet activation; agents that block the interaction between platelets and these factors are now undergoing investigation.
Platelet activation and aggregation are amplified by adenosine diphosphate (ADP) and thromboxane A2 (TXA2), which act as secondary agonists on adjacent platelets. Thrombin has a pivotal role in platelet activation via PAR (protease activated receptor)-1 and PAR-4 receptors; drugs that block its interaction with PAR-1 receptors are now under investigation. The PAR-1 – thrombin interaction is largely unchecked by current oral antiplatelet therapy. Only parenteral glycoprotein (GP) IIb/IIIa blockers potently and uniformly inhibit aggregation induced by thrombin. ADP, thromboxanes and thrombin (generated on the surface of platelets) all amplify the process of thrombosis. The final pathway leading to platelet aggregation is the interaction between the activated conformation of the GP IIb/IIIa receptor on the platelet membrane and the dimeric fibrinogen molecule and von Willebrand factor.
Thrombin converts fibrinogen to fibrin. Finally, the fibrin – platelet aggregate stabilises the clot and may lead to occlusion of the vessel. It is the combination of the vulnerable substrate within the vessel wall and the vulnerable, pro-thrombotic blood that is essential for the clinical occurrence of an occlusive thrombotic event.
Limitations of aspirin
Management guidelines agree closely on the role of aspirin in secondary prevention and have similar recommendations on its use as primary prevention, with some variation in risk thresholds at which treatment is indicated. Aspirin may also be recommended for atrial fibrillation when an oral anticoagulant is unsuitable. The American Heart Association recommends aspirin for primary prevention of ischaemic stroke in women (but not men) with sufficiently increased risk.
However, the ‘one size fits all’ and ‘one mechanism fits all’ approach may reflect the limitations of current evidence from clinical trials and our lack of understanding of the complete mechanisms of action of aspirin, respectively. The evidence for use of low-dose aspirin comes not from robust prospective data but primarily from meta-analyses and retrospective analyses. There is currently a huge controversy surrounding its role as primary prevention in patients with diabetes. More studies are needed, particularly to address the heterogeneity in thrombotic risk in this population. Underutilisation and low compliance with treatment with aspirin remain significant challenges.
Resistance measurement lacks specificity
Aspirin resistance should be defined in terms of insufficient pharmacological inhibition of platelet cyclooxygenase-1 (COX-1)-dependent thromboxane synthesis. In the presence of aspirin, platelets utilise the COX-2 pathway to synthesise thromboxane and through this mechanism the antithrombotic efficacy of aspirin may be reduced. The Aspirin-induced platelet effect (ASPECT) study clearly showed that the detection of resistance depends on the method used: COX-1-specific methods utilising platelet-rich plasma and arachidonic acid as the agonist indicate a lower incidence of resistance than methods using whole blood,2,3 possibly due to the presence of unblocked COX-2. There was also a clear dose-dependent effect on platelet function in non-COX-1 specific assays such as collagen-induced aggregation, the platelet function analyser and urinary 11-dehydro-thromboxane B2.2

However, aspirin also exerts antithrombotic effects via non-COX mechanisms (figure 2). It has dose-dependent effects on collagen-, shear-, and ADP-induced aggregation (incompletely) and on thrombin generation, and it acetylates prothrombin, antithrombin, fibrinogen and factor XIII. It has been proposed that aspirin alters the function of the GP IIb/IIIa receptor and it affects clot permeability.2,4,5
The importance of compliance was illustrated by Tantry.6 In healthy volunteers, aspirin completely blocked arachidonic acid (AA)-induced platelet aggregation in platelet-rich plasma measured by turbidometric aggregometry and in whole blood measured by thromboelastography (TEG); complete inhibition was also evident among those patients who are fully compliant with treatment, both before and after percutaneous coronary intervention (PCI). AA-induced platelet aggregation was not inhibited in non-compliant patients before PCI; when these patients received a single 325 mg dose of aspirin after PCI, aggregation was completely inhibited 24 hours later.
Aspirin and diabetes
Platelets in patients with diabetes have increased reactivity as well as increased turnover. There is, however, a high level of uncertainty about the balance of benefit and risk of primary prevention with aspirin in this population and management guidelines have made conflicting recommendations.7
A recent meta-analysis found that the benefits of primary prevention with aspirin were similar for patients with or without diabetes8 and the Prevention of progression of arterial disease and diabetes (POPADAD) trial showed no benefit from aspirin in patients with diabetes and peripheral arterial disease.9 What could explain this? The ASPECT study compared platelet function in diabetic and non-diabetic patients and found a stronger dose-dependency among patients with diabetes, suggesting that these patients may need higher doses of aspirin.2 It is also possible that glycaemic control affects platelet function: platelet aggregation induced by ADP is higher in patients with glycosylated haemoglobin (HbA1C) levels >7% compared with <7%, showing that poor glycaemic control is associated with higher platelet reactivity.10
Bleeding risk and higher doses
Evidence that higher doses of aspirin monotherapy (≥200 mg/day) are associated with an increased risk of bleeding compared with lower doses is unconvincing. Evidence for an increased risk was suggested in the presence of dual antiplatelet therapy from a subanalysis of the PCI-CURE study, which found a significant increase in risk of major bleeding among patients who received doses of ≥200 mg/day compared with those who received lower doses.11 The Clopidogrel for high atherothrombotic risk and ischaemia stabilisation, management and avoidance (CHARISMA) trial suggested that aspirin at doses of 100 mg/day or above may increase risk in patients taking dual therapy with clopidogrel.12 However, data from a true prospective study of aspirin dose suggest otherwise. The Clopidogrel optimal dose usage to reduce recurrent events/optimal antiplatelet strategy for intervention (CURRENT-OASIS 7) trial included 25,087 patients randomised in a 2 x 2 design to receive two dose regimens of clopidogrel with high-dose (300–325 mg/day) or low-dose (75–100 mg/day) aspirin; the primary endpoint was the incidence of ischaemic events after 30 days.13 There was no difference between aspirin doses for overall efficacy or bleeding risk, whereas bleeding risk was greater with high-dose clopidogrel.
Conflict of interest
PAG has a consulting agreement with Bayer and is paid by Bayer to be the Chairman of the adjudication committee of a major clinical trial funded by Bayer.
References
- Gurbel PA, Becker RC, Mann KG et al. Platelet function monitoring in patients with coronary artery disease. J Am Coll Cardiol 2007;50:1822–34.
- Gurbel PA, Bliden KP, DiChiara J et al. Evaluation of dose-related effects of aspirin on platelet function: results from the Aspirin-Induced Platelet Effect (ASPECT) study. Circulation 2007;115:3156–64.
- Lordkipanidzé M, Pharand C, Schampaert E et al. A comparison of six major platelet function tests to determine the prevalence of aspirin resistance in patients with stable coronary artery disease. Eur Heart J 2007;28:1702–08.
- Szczeklik A, Krzanowski M, Góra P et al. Antiplatelet drugs and generation of thrombin in clotting blood. Blood 1992;80:2006–11.
- Undas A, Brummel-Ziedins KE, Mann KG. Antithrombotic properties of aspirin and resistance to aspirin: beyond strictly antiplatelet actions. Blood 2007;109:2285–92.
- Tantry US, Bliden KP, Gurbel PA. Overestimation of platelet aspirin resistance detection by thrombelastograph platelet mapping and validation by conventional aggregometry using arachidonic acid stimulation. J Am Coll Cardiol 2005;46:1705–09.
- Nicolucci A, De Berardis G, Sacco M et al. AHA/ADA vs. ESC/EASD recommendations on aspirin as a primary prevention strategy in people with diabetes: how the same data generate divergent conclusions. Eur Heart J 2007;28:1925–7.
- Calvin AD, Aggarwal NR, Murad MH et al. Aspirin for the primary prevention of cardiovascular events: a systematic review and meta-analysis comparing patients with and without diabetes. Diabetes Care 2009 Sep 9; published online ahead of print.
- Belch J, MacCuish A, Campbell I et al. The prevention of progression of arterial disease and diabetes (POPADAD) trial: factorial randomised placebo controlled trial of aspirin and antioxidants in patients with diabetes and asymptomatic peripheral arterial disease. BMJ 2008;337:a1840. doi: 10.1136/bmj.a1840.
- Singla A, Antonino MJ, Bliden KP, Tantry US, Gurbel PA. The relation between platelet reactivity and glycemic control in diabetic patients with cardiovascular disease on maintenance aspirin and clopidogrel therapy. Am Heart J 2009;158:784.e1–6.
- Jolly SS, Pogue J, Haladyn K et al. Effects of aspirin dose on ischaemic events and bleeding after percutaneous coronary intervention: insights from the PCI-CURE study. Eur Heart J 2009;30:900–07.
- Steinhubl SR, Bhatt DL, Brennan DM et al; CHARISMA Investigators. Aspirin to prevent cardiovascular disease: the association of aspirin dose and clopidogrel with thrombosis and bleeding. Ann Intern Med 2009;150:379–86.
- Mehta SR. A randomized comparison of a clopidogrel high loading and maintenance dose regimen versus standard dose and high versus low dose aspirin in 25,000 patients with acute coronary syndromes: Results of the CURRENT OASIS 7 Trial. European Society of Cardiology Congress. Barcelona. 29 Aug – 02 Sep 2009.

