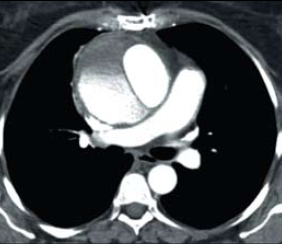
A 57-year-old woman presented with a six-week history of non-productive cough associated with sharp chest pain. Her past medical history included a metallic aortic valve replacement for aortic regurgitation, hypertension and hypercholesterolaemia. The patient had a blood pressure of 97/60 mmHg and was afebrile. On examination, the metallic valve was audible with no added heart sounds. Examination of other systems was normal. Electrocardiogram (ECG) showed a normal sinus rhythm with no ischaemic changes. Admission blood tests showed an elevated white cell count (11.4 x 109 per litre) and a C-reactive protein of 225.8 mg/L. Her chest radiograph demonstrated a widened mediastinum, evidence of previous cardiac surgery and a metallic valve (figure 1). Transthoracic echocardiography showed a dilated aortic root measuring 62 mm at the level of the sinotubular junction. In addition, a thrombus was visualised in the ascending aorta with a dissection flap, which was confirmed by computed tomography (CT) scan (figure 2). The patient was transferred to a cardiothoracic unit to undergo surgical repair of the dissection and replacement of the metallic valve. The dissection was shown to arise from the suture line of the previous valve replacement.
Discussion
Ortner’s syndrome, or cardiovocal syndrome, is a clinical condition associated with left recurrent laryngeal nerve palsy due to cardiovascular disease. The palsy arises from compression of the recurrent laryngeal nerve as it passes between the arch of the aorta and the pulmonary artery.1,2 The syndrome was first described in 1897 by Norbert Ortner, an Austrian physician who ascribed hoarseness of voice with left recurrent laryngeal nerve palsy secondary to a dilated left atrium in three patients with mitral valve stenosis.3 It has now been estimated that the incidence of Ortner’s syndrome in mitral stenosis ranges from 0.6 to 5%.4
Following the first description of Ortner’s syndrome, case reports have described other cardiovascular associations, including left atrial enlargement due to mitral regurgitation or atrial myxoma, severe pulmonary hypertension and congenital heart disease.5-8 In addition, aneurysms of the aorta and pulmonary artery causing Ortner’s syndrome have been reported and can present with dissection,9,10 as described here. However, cough is an unusual initial presentation of Ortner’s syndrome. Most case reports describe hoarseness of voice as the presenting symptom. Left laryngeal nerve palsy and paralysis of the left vocal cord can also present with dysphagia and shortness of breath during speech.11 Furthermore, effective cough cannot be mounted and patients are at risk of aspiration. Finally, our case describes chest pain symptoms not classically associated with aortic dissection. It has been reported that aortic dissection in patients with previous cardiac surgery does not always present with central tearing chest pain, and this is thought to be due to denervation of cardiac sympathetic supply as a result of the previous operation.12 This case highlights how a common symptom such as cough can be due to unsuspecting pathology and knowledge of the anatomy can assist in diagnosis.
Conflict of interest
None declared
References
- Fetterolf G, Norris G. The anatomical explanation of paralysis of left recurrent laryngeal nerve found in certain cases of mitral stenosis. Am J Med Sci 1911;141:625–38. http://dx.doi.org/10.1097/00000441-191105000-00001
- Ari R, Harvey WP, Hufnagel CA. Etiology of hoarseness associated with mitral stenosis: improvement following mitral surgery. Am Heart J 1955;50:153–60. http://dx.doi.org/10.1016/0002-8703(55)90261-1
- Ortner N. Recurrenslahmung bei mitral stenose. Wien Klin Wochenschr 1897;10:753–5.
- Solanki SV, Yajnik VH. Ortner’s syndrome. Indian Heart J 1972;24:43–6.
- Fennessy BG, Sheahan P, McShane D. Cardiovascular hoarseness: an unusual presentation to otolaryngologists. J Laryngol Otol 2008;122:327–8. http://dx.doi.org/10.1017/S0022215107008110
- Gulel O, Koprulu D, Kucuksu Z et al. Images in cardiovascular medicine. Cardiovocal syndrome associated with huge left atrium. Circulation 2007;115:e318–e319.
- Ishimoto S, Ito K, Toyama M et al. Vocal cord paralysis after surgery for thoracic aortic aneurysm. Chest 2002;121:1911–15. http://dx.doi.org/10.1378/chest.121.6.1911
- Kishan CV, Wongpraparut N, Adeleke K et al. Ortner’s syndrome in association with mitral valve prolapse. Clin Cardiol 2000;23:295–7. http://dx.doi.org/10.1002/clc.4960230416
- Kamp O, van Rossum AC, Torenbeek R. Transesophageal echocardiography and magnetic resonance imaging for the assessment of saccular aneurysm of the transverse thoracic aorta. Int J Cardiol 1991;33:330–3. http://dx.doi.org/10.1016/0167-5273(91)90367-X
- Harano M, Tanemoto K, Kuinose M et al. A case of chronic traumatic dissecting aneurysm of the thoracic aorta. Kyobu Geka 1994;47:1023–5.
- Mulpuru SK, Vasavada BC, Punukollu GK et al. Cardiovocal syndrome: a systematic review. Heart Lung Circ 2008;17:1–4. http://dx.doi.org/10.1016/j.hlc.2007.04.007
- Collins JS, Evangelista A, Nienaber CA et al. Differences in clinical presentation, management, and outcomes of acute type a aortic dissection in patients with and without previous cardiac surgery. Circulation 2004;110(11 suppl 1):II, 237–42. http://dx.doi.org/10.1161/01.CIR.0000138219.67028.2a


