A 56-year-old male with no previous significant medical history initially presented to his general practitioner for a routine health check prior to starting a new occupation. An electrocardiogram (ECG) was taken that was found to be unusual and, hence, a referral to cardiology outpatients was made. Initial recommendation was made for ablation therapy based on the finding of Wolff-Parkinson-White (WPW) syndrome. After a successful procedure, the patient developed significant palpitations with haemodynamic compromise that required emergency direct current (DC) cardioversion. Subsequent re-investigation found a previously unmasked uncommon form of accessory tachyarrhythmia. This case report highlights the finding of Mahaim pathway in a patient initially treated for WPW syndrome.
Introduction
The term pre-excitation was originally used to describe premature activation of the ventricles in Wolff-Parkinson-White (WPW) syndrome. However, this term has now been broadened to include a number of other conditions in which ventricular activation occurs via other anomalous pathways. It is important to define the underlying electrical mechanism when patients present with arrhythmias, as this can affect the management of the patient. Terms such as supraventricular tachycardia (SVT) should be replaced with more precise diagnoses, such as atrioventricular (AV) nodal re-entry tachycardia (AVNRT). Patients with the uncommon finding of a Mahaim pathway often have other accessory pathways in parallel (40%). This adds to the diagnostic dilemma, and it is not uncommon to unearth the underlying Mahaim tachycardia after initial surgical or catheter ablation of the accessory pathway has taken place. The following case report highlights the diagnostic challenges we face in the sub-speciality of electrophysiology.
Case report
A previously healthy, asymptomatic 56-year-old male was referred to cardiology outpatients following the incidental finding of a type B WPW pre-excitation on a routine 12-lead electrocardiogram (ECG) (figure 1). Transthoracic echocardiography was performed and showed no significant valvular abnormalities, but an exercise tolerance test demonstrated no loss of the accessory pathway conduction, despite achieving maximum heart rate. Subsequently, in view of the possibility of developing atrial fibrillation with a rapid ventricular response, he was referred for a radiofrequency ablation of a presumed right-sided accessory pathway.
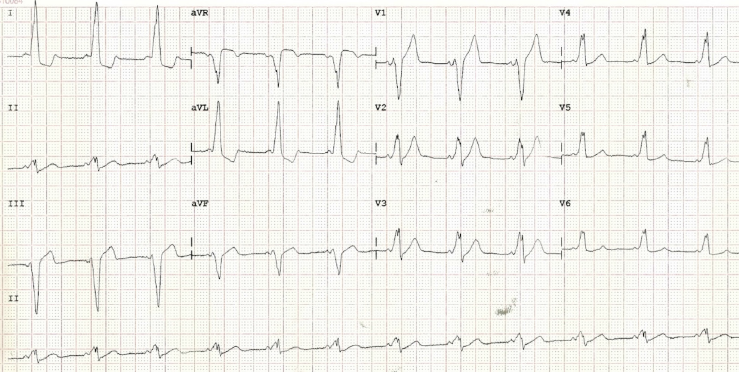
Diagnostic electrophysiology (EP) studies revealed a readily inducible antidromic AVNRT conducted through an accessory pathway located at the five o’clock position on the tricuspid annulus. This was successfully ablated with use of a Celsius Thermacool D curve, and the patient was discharged. An ECG, three months post-ablation, demonstrated normal sinus rhythm without any evidence of pre-excitation.
Eight months following the ablation procedure, the same patient presented to a district general hospital emergency department complaining of palpitations and breathlessness. His ECG, on this admission, revealed a broad complex tachycardia with a ventricular rate of 170 bpm (figure 2).
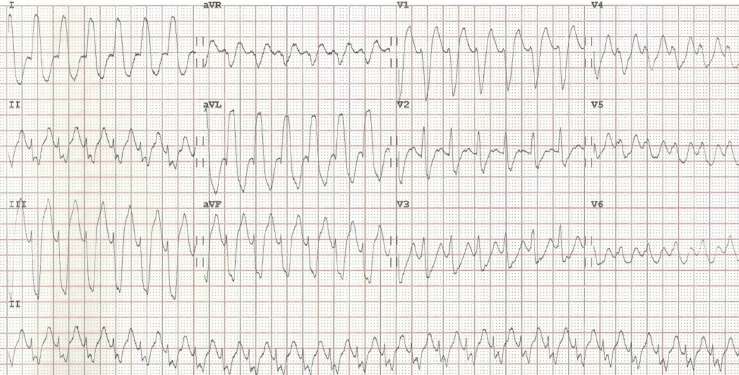
Clinically, there was evidence of haemodynamic compromise, and so he was direct current (DC) cardioverted back to sinus rhythm. His post-cardioversion ECG did not reveal a recurrence of the accessory pathway, raising the suspicion of a possible concealed pathway (figure 3).
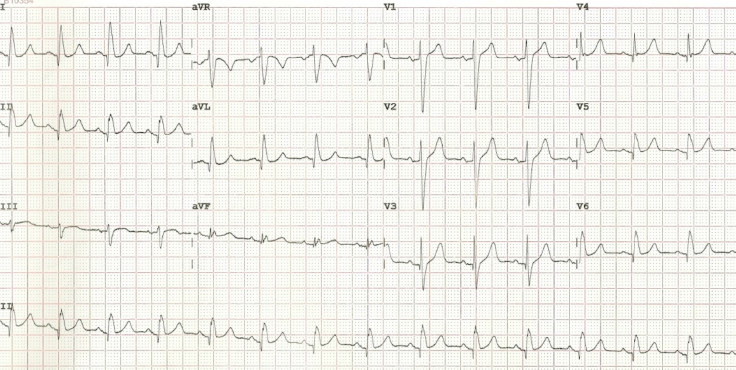
He subsequently underwent a second diagnostic EP study, which revealed an inducible tachycardia with left bundle branch block morphology. Initial mapping for a Kent potential was unsuccessful, therefore, atrial entrainment from close to the tricuspid valve annulus was used to approximately localise the pathway. This revealed an accessory pathway, which had not been seen at the initial ablation procedure, located at the seven o’clock position on the tricuspid annulus, which demonstrated antegrade decremental conduction. This was recognised as representing a Mahaim fibre, which was subsequently mapped from its atrial insertion and successfully ablated. The patient was followed-up six months following the ablation procedure, where he reported no further episodes of palpitations or breathlessness.
Discussion
The term ‘Mahaim fibre’ was first conceived in 1937 by Mahaim and Bennet to describe multiple muscular connections in the normal heart that joined the AV bundle to the crest of the ventricular septum. Such accessory pathways originated from the His bundle and inserted into the distal right bundle branch (fasciculoventricular fibres).1 This definition was later expanded to include accessory pathways of conducting tissue extending from the AV node to the right ventricular myocardium (nodoventricular fibres).
However, subsequent studies have demonstrated that these more commonly originate from the free wall of the right atrium, than the AV node itself, and insert into or close to the right bundle branch.2 These two principle classes of Mahaim fibre differ anatomically, but share common electrophysiological properties in displaying purely antegrade decremental conduction and widened QRS width with left bundle branch morphology at higher pacing rates. The exact prevalence of Mahaim fibre is not known, but the consensus in the literature is that it is relatively uncommon, representing less than 3% of all mapped AV accessory pathways.3
The resting 12-lead ECG in a patient with a Mahaim pathway usually demonstrates normal sinus rhythm without any evidence of pre-excitation, unless there is a co-existent accessory AV pathway. This is thought to occur in as many as 40% of patients, and can often conceal a Mahaim fibre, which may only become apparent following radiofrequency ablation of the alternative pathway.4,5 The most common presenting arrhythmia in patients with a Mahaim fibre is a re-entrant tachycardia, with the Mahaim fibre acting as the antegrade limb and the AV node as the retrograde limb of the circuit. Typically, the surface ECG reveals a left bundle branch pattern and left axis deviation. Management is either pharmacological with class Ia or Ic anti-arrhythmics and/or beta blockers, or, more commonly, a curative approach through radiofrequency ablation of the Mahaim fibre.3
Conclusion
From our case report it is evident that Mahaim fibre tachycardias form a distinct subgroup of pre-excitation syndromes (approximately 3%). This should be suspected if the ECG in tachycardia reveals a left bundle branch block morphology with left axis deviation. In some patients, this may only manifest following ablation of a co-existing accessory pathway.
Acknowledgement
The authors would like to thank Dr Waktare, Consultant Cardiologist and Cardiac Electrophysiologist, for the provision of the electrophysiology study and radiofrequency ablation procedure reports.
Conflict of interest
None declared.
References
1. Klein LS, Hackett FS, Zipes DP, Miles WM. Radiofrequency ablation of Mahaim fibers at the tricuspid annulus. Circulation 1993;87:738–47. http://dx.doi.org/10.1161/01.CIR.87.3.738
2. Tchou P, Lehmann MH, Jazeyari M, Akhtar M. Atriofasicular connection or a nodoventricular mahaim fiber? Electrophysiological elucidation of the pathway and associated re-entrant circuit. Circulation 1988;77:837–48. http://dx.doi.org/10.1161/01.CIR.77.4.837
3. Aliot E, de Chillou C, Revault d’Allones G, Mabo P, Sadoul N. Mahaim tachycardias. Eur Heart J 1998;19(suppl E):E25–E31, E52–E53.
4. Gallagher JJ, Smith WM, Kasell JH et al. Role of Mahaim fibers in cardiac arrhythmias in man. Circulation 1981;64:176–89. http://dx.doi.org/10.1161/01.CIR.64.1.176
5. Tonkin AM, Dugan FA, Svenson RH et al. Coexistence of functional Kent and Mahaim-type tracts in the pre-excitation syndrome. Demonstration by catheter techniques and epicardial mapping. Circulation 1975;52:193–200. http://dx.doi.org/10.1161/01.CIR.52.2.193

