Much of the European Society of Cardiology (ESC) 2014 Congress programme was devoted to important clinical issues such as management of arrhythmia, and stroke prevention in non-valvular atrial fibrillation (NVAF). This was supplemented with updates from major AF Registry data, which chart the rates of AF, along with trends in adopting novel oral anticoagulants (NOACs), and the incidence of DC cardioversion and radiofrequency ablation throughout Europe. There was also much anticipation surrounding the results from the randomised clinical studies looking at rivaroxaban in cardioversion, and amiodarone in AF ablation, both included in ESC hotline sessions.
X-VERT: rivaroxaban▼ an alternative to VKA in cardioversion for AF

Oral anticoagulant therapy with rivaroxaban is an alternative to vitamin K antagonists (VKAs) in patients with AF who are undergoing elective cardioversion according to the results of the X-VERT study.1 In addition, rivaroxaban may potentially have one important advantage over VKAs, with a shorter time to cardioversion, the study suggests.
Professor Riccardo Cappato (University of Milan, Italy), the co-principal investigator who presented the results, said X-VERT had shown that time to cardioversion was shorter with rivaroxaban compared to VKA in both study arms, with it being significantly shorter in the delayed strategy arm of the study.
X-VERT (eXplore the efficacy and safety of once-daily oral riVaroxaban for the prevention of caRdiovascular events in patients with nonvalvular aTrial fibrillation scheduled for cardioversion) is the first prospective, randomised trial to compare the safety and efficacy of rivaroxaban compared to VKA therapy in cardioversion.
It was hailed as a “landmark trial in the development of NOACs” by discussant Professor Christoph Bode (Univeristy of Freiburg, Germany) who said there had previously only been post-hoc data for the use of NOACs and VKAs in cardioversion.
Carried out in 141 centres and 16 countries on patients with AF scheduled to undergo either electrical (97.6%) or pharmacological (2.4%) cardioversion, X-VERT randomised 1,002 patients to oral rivaroxaban 20 mg once daily and 502 patients to VKA (warfarin or another VKA at the investigator’s discretion, based on local standard of care).
Using established guidelines, patients were assigned to either early (58%) or delayed (42%) cardioversion. In the delayed group, cardioversion was allowed if at least three consecutive weeks of adequate anticoagulation was documented prior to cardioversion. VKA anticoagulation was considered adequate if the international normalised ratio (INR) was maintained in the range of 2.0–3.0 for that time period, while anticoagulation with rivaroxaban was considered adequate by drug compliance of at least 80% for that time period.
Cardioversion in the early group was performed within a target range of one to five days after randomisation, while patients in the delayed group had cardioversion performed between 21 and 25 days after randomisation. Follow-up was for 30 days.
The study found that compared to patients taking a VKA, patients treated with rivaroxaban had a similar of risk of the primary composite outcome (stroke or transient ischaemic attack, peripheral embolism, myocardial infarction, and cardiovascular death), which occurred in 0.5% of the rivaroxaban group versus 1.02% of the VKA group, which was not statistically significant.
In the early cardioversion group, the primary composite outcome occurred in 0.71% of rivaroxaban-treated patients and 1.08% of VKA-treated patients, whereas in the delayed cardioversion group it occurred in 0.24% and 0.93% patients in the rivaroxaban and VKA groups, respectively.
There was no difference in major bleeding between the rivaroxaban and VKA groups (0.61% versus 0.80%, respectively; RR 0.76; CI 0.21-2.67), and also no clinically important differences in either the overall cumulative incidence of adverse events and serious adverse events by treatment assignment or by cardioversion strategy.
The study did, however, show that time to cardioversion was significantly shorter with the delayed strategy when patients were treated with rivaroxaban compared to the VKA-treated patients (median 22 versus 30 days, respectively, p < 0.001). A result, Professor Cappato said, that was due to the “inability to achieve adequate anticoagulation prior to cardioversion in the VKA group”.
There were 95 VKA-treated patients with inadequate anticoagulation in the delayed strategy arm compared to only one patient in the rivaroxaban group. Time to cardioversion was similar with the early strategy using rivaroxaban compared with VKA (median 1 day).
The study drew some criticism for its small size from discussant Professor Steve Nissan (Cleveland Clinic, Ohio, US), who said this made the findings inconclusive. Professor Cappato disagreed saying it gave “clinically meaningful information”. He did, however, say that the practical advantage of rivaroxaban over VKAs, demonstrated by the short time to cardioversion should be interpreted with caution since time to cardioversion was not a prespecificed outcome of the study.
AMIO CAT: amiodarone shows short-term benefits in AF ablation
AMIO CAT, the first double-blind randomised clinical trial to evaluate a short-term course of the antiarrhythmic, amiodarone, in AF ablation showed that early recurrence of arrhythmia could be effectively reduced with this medication within the first three months after ablation. The benefit does not persist beyond this.2
“Although amiodarone did not affect recurrence at six months, our study shows that short-term use of this medication after ablation is still a relevant strategy because of its beneficial effects during the first three months,” said investigator Dr Stine Darkner (Heart Centre, Rigshospitalet, Copenhagen, Denmark).
The study included 212 patients undergoing radiofrequency ablation for the treatment of either paroxysmal or persistent AF who were randomised to receive eight weeks of either amiodarone (n=108) or placebo (n=104) starting immediately after their procedure.
The primary end point of the study was AF lasting more than 30 seconds after the “blanking period” – a three-month period in which AF episodes can occur as part of the healing process and are generally not counted in final study results.
At six months, the study showed no significant difference in AF recurrence between the treated and placebo groups (39% vs. 48%, p=0.18). But during the blanking period, amiodarone reduced the number of AF recurrences compared to placebo (34% vs. 53%, p=0.006), and more than halved arrhythmia-related hospitalisation (p=0.006) and cardioversion rates (p=0.0004).
Looking separately at patients who entered the study with either paroxysmal (n=107) or persistent (n=105) AF, the analysis showed that “it seems that effect was largely driven by the group of patients with persistent AF,” noted Dr Darkner.
There was no statistically significant difference in the number of serious adverse events between the treated and placebo groups. Even though significantly more patients in the amiodarone group experienced transient adverse effects, these patients did not report reduced quality of life compared to those treated with placebo.
“The adverse effects of amiodarone therapy are well known and not unexpected,” said Dr Darkner. “The similarities in quality of life, despite the higher number of adverse events in the amiodarone group may be due to the reduced AF-related hospitalisations and cardioversions,” she suggested.
“Current guidelines for AF ablation do not give specific recommendations with respect to early antiarrhythmic drug therapy after ablation, but our data suggests that short term prophylactic treatment with amiodarone should be considered, particularly for patients with persistent AF”, Dr Darkner concluded.
ENGAGE AF-TIMI 48: edoxaban dose reduction shows preserved efficacy
More evidence on stroke prevention in AF has emerged from subgroup analyses of major outcome studies, ENGAGE AF-TIMI 483 and ROCKET AF,4 which were presented at the congress.
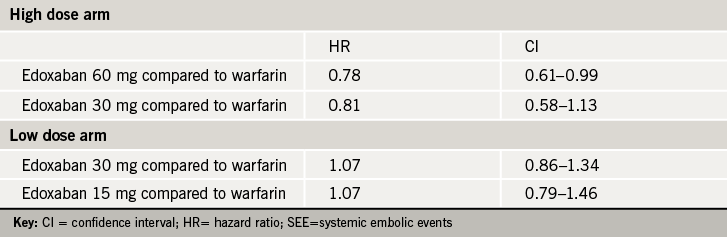
Subgroup analysis of the ENGAGE AF-TIMI 48 study showed that dose reduction with edoxaban preserved efficacy with even greater relative reductions in bleeding compared with warfarin in the treatment of patients with NVAF.
Explaining the background to the analysis, Dr Christian T Ruff (Brigham and Women’s Hospital, Boston, US) said there has been emerging concern that the fixed dosing regimen of the NOACs may not “fit all” and that to optimise the risk/benefit of NOACs, it may be necessary to measure drug concentration and/or anticoagulant activity. This led the TIMI study group to look at the relationship between edoxaban dose, concentration, anti-factor Xa activity and outcomes in the ENGAGE AF-TIMI 48 trial.
In the phase 3 study, some 24.5% of patients met the clinical criteria for an edoxaban dose reduction. These were high-risk patients with clinical factors which increased the risk of bleeding due to higher drug exposure, such as those with a creatinine clearance of 30-50 ml/min, weight ≤ 60 kg, and/or concomitant use of certain P-glycoprotein inhibitors (verapamil, quinidine), Such patients randomised to edoxaban were given a 50% dose reduction, from 60 mg to 30 mg in the high-dose arm of the study, and from 30 mg to 15 mg in the low-dose arm of the study.
The subgroup analysis found that this 50% dose reduction of edoxaban led to mean edoxaban exposure being reduced by 29% and 35%, and anti-factor Xa activity being reduced by 20% and 25%, in the high-dose and low-dose regimens, respectively, compared to the population who were not dose reduced.
Compared to warfarin, this dose reduction of edoxaban preserved relative efficacy in prevention of stroke and systemic embolic events, and provided even greater safety with respect to major bleeds and intracranial haemmorhage (see tables 1 and 2).
Digoxin may increase adverse events in AF treatment
 Digoxin may increase the risk of death in AF patients by approximately 20%, according to results from a subgroup analysis of the ROCKET AF trial4 presented by Dr Manesh Patel (Duke University, Durham, North Carolina, US). The findings suggest that caution may be needed when using digoxin in complex AF patients.
Digoxin may increase the risk of death in AF patients by approximately 20%, according to results from a subgroup analysis of the ROCKET AF trial4 presented by Dr Manesh Patel (Duke University, Durham, North Carolina, US). The findings suggest that caution may be needed when using digoxin in complex AF patients.
Dr Patel said, “In this sub-analysis of the ROCKET AF trial we found that AF patients who took digoxin had an increased risk of adverse cardiovascular events, including vascular death or sudden death”.
The ROCKET AF trial was a double-blind trial conducted in 14,171 patients with NVAF who were at increased risk of stroke. The study took place at 178 sites in 45 countries. Rivaroxaban was non-inferior to warfarin for the prevention of stroke or systemic embolism.
In the current analysis, the researchers examined the risks of all-cause mortality and cardiovascular mortality in 5,239 patients taking digoxin at the start of the study, compared to 8,932 patients who were not taking digoxin. The researchers found that use of digoxin was associated with a 22% increased risk of death due to any cause and a 22% increased risk of death due to a cardiovascular event.
The use of digoxin in AF patients has not been rigorously studied in randomised trials and, Dr Patel said, “our analysis represents an important observation ..digoxin could potentially be harmful and we need randomised trials to determine whether it can be used patients with atrial fibrillation”.
Our findings suggest that doctors should proceed with caution when using this drug for complex AF patients”, he concluded.
Permanent AF has double stroke risk of PAF
Much was also learned at the congress about the prescribing of anticoagulants and the natural history of AF.
Permanent AF, for example, has been found to double the risk of stroke compared to paroxysmal AF (PAF) according to findings presented by Dr Thomas Vanassche (McMaster University, Hamilton, Canada).5
The finding comes from an analysis of pooled data on 6,563 aspirin treated patients who were enrolled in the ACTIVE-A (clopidogrel) and AVERROES (apixaban▼) clinical trials. Both studies included an aspirin control arm, with no patients receiving anticoagulants. “Although it may seem intuitive that the continuous presence of AF carries a higher stroke risk than short, intermittent episodes, this has been an area of controversy” as patients in permanent AF are often younger and at lower stroke risk than those with PAF, said Dr Vanassche. Also, some studies have not reported a significant difference. This analysis showed an adjudicated yearly event rate of 4.2% versus 2.1% for permanent AF versus PAF, respectively.
The message is that it’s very important to acknowledge that, regardless of its form and presentation, AF increases risk of stroke and “if risk is sufficiently high, they (patients) should be treated with anticoagulants,” he concluded.
High AF mortality despite treatment
Even when anticoagulants are deployed, mortality and morbidity of AF patients remains high after one-year’s treatment, according to Professor Greg Lip (University of Birmingham, UK), who outlined data from the The Atrial Fibrillation General Pilot (part of the ESC’s, EURObservational Research Programme [EORP]).6 This reflects the impact of European AF guidelinesof 2010 and 2012 and includes over 3,000 patients from nine countries.
The one-year follow-up results reveal that more than 80% of AF patients who are at risk of embolic stroke (according to the CHA2DS2-VASc score) receive appropriate anticoagulation. But one-year mortality is still high (approximately 6%), of which most are cardiovascular deaths. Approximately 20% of patients taking anticoagulants required a change in treatment at one year. Some patients taking warfarin switched to a non-vitamin K antagonist oral anticoagulant (NOAC), while some initially on a NOAC changed to a different antithrombotic drug. A small minority of patients were started on aspirin despite the guidelines and little evidence for the efficacy and safety of aspirin.
Professor Lip said “The results show that many European cardiologists prescribe oral anticoagulation for their patients with AF and this is encouraging. But we shouldn’t rest on our laurels given that these patients still experience high mortality and morbidity at one year. And we need more information on why so many patients are changing the type of anticoagulation they use.”
Inappropriate anticoagulation in low-risk patients
While awareness of anticoagulation is increasing, inappropriate anticoagulation in some low-risk patients has been revealed by the GARFIELD-AF registry. It has highlighted that some truly low-risk patients with NVAF continue to receive antithrombotic therapy.7
Data from GARFIELD-AF have shown that of those patients who were assessed using a scoring system, a total of 6.6% and 3.6% had CHADS2 and CHA2DS2-VASc scores of 0, respectively, and are therefore classified as truly low risk. The CHADS2 scoring system had been used to assess 12,195 patients and the CHA2DS2-VASc system to assess 12,184 patients in the registry.
Of these truly low-risk patients, approximately 75% received antithrombotic or antiplatelet therapy, although they are considered to be a low risk of stroke in current European Society of Cardiology guidelines.
Data have also shown a trend towards differences in predictive accuracy for stroke/SE between the two scoring systems for low risk patients although no definite conclusions can be drawn because of the small numbers. While the unadjusted one-year incidence of all-cause death, stroke/SE and major bleeding increased substantially when risk scores were ≥1, the one-year incidence of stroke/SE was lower in those with CHA2DS2-VASc of 0 (0.7%) versus those with a CHADS2 score of 0 (1.2%).
Worse outcome with poor control
Results presented from the GARFIELD-AF registry show that an increased risk of severe adverse outcomes is associated with suboptimal INR (international normalised ratio) control.8 In the first cohort of patients in the registry with NVAF taking a vitamin K antagonist (VKA) at one-year follow-up (n = 6,297), INR results were available for 5,107 VKA-treated patients.
Unadjusted rates of stroke/systemic embolism (SE), major bleeding and all-cause death were higher in poorly controlled patients (defined as a frequency in range [FIR] of less than 70%) than those who were well controlled (defined as an FIR of more than 70%).
Overall GARFIELD-AF has now enrolled over 36,000 prospective patients with AF in four cohorts in 30 countries and 739 sites.
Patient satisfaction and switching
Data from another registry, PREFER in AF, is shedding light on some of the reasons why patients are changing the type of anticoagulation they use, with treatment satisfaction being one of the main reasons for patients being switched from a VKA to a NOAC.
Patients switching were more likely to have mobility problems than those stably treated with a VKA (13.3% versus 7.3%), and more likely to be having difficulties in dose adjustments, extreme discomfort with bruising or pain, dissatisfaction with their treatment and some anxiety or depression when being treated with a VKA.9
The PREFER in AF registry has enrolled 7,243 patients from 461 sites with follow-up data at one year collected from 6,412 patients. Its data tallies with that of EORP (see above) showing that at one-year follow-up, the majority of patients with NVAF (80%) were on oral anticoagulants and, of these, the percentage being on NOACs was increasing, doubling in the year from 6.1% to 12.6%, with use of vitamin K antagonist and antiplatelet agents reducing in the same period.
Global differences
Substantial inter-country differences were noted in the PREFER in AF registry with a higher uptake of NOACs for the treatment of AF in Austria, France, Germany, Spain and Switzerland than in the UK and Italy.10
More differences in atrial fibrillation management were shown in a global online survey carried out on 1,100 cardiologists from seven countries (Brazil, France, Germany, Japan, Spain UK and USA). The survey was carried out by Daiichi Sankyo and the Heart Rhythm Society.
Some 84% of cardiologists overall indicated a majority of their patients with non-valvular AF received an oral anticoagulant, with the lowest use (79.4%) indicated by UK cardiologists compared to 86.6% by cardiologists in Japan.
The survey also highlighted that 58% of cardiologists think that there is no such thing as a “typical” NVAF patient, with the majority (88%) agreeing that the diversity of the condition makes it important to focus on individual co-morbidities and patient characteristics to provide appropriate disease management. The survey showed, on average, NVAF patients have about three co-morbid conditions.
Almost all the cardiologists surveyed (98%) believed that NVAF patients may experience a delay in diagnosis primarily because they are asymptomatic, but also due to low awareness among primary care physicians (40%) and the general public (36%) of the condition.
Similar results were seen in a French awareness campaign of AF in general practice, PROFIL FA.11 This has shown that early diagnosis is often difficult and in up to 20% of AF-related strokes, AF is discovered too late at the time of the stroke. The campaign – carried out on 603 physicians and 4,592 patients, also highlighted that an irregular pulse was linked to a diagnosis of AF in one one out of two cases, although ESC 2012 recommendation say an irregular pulse should be systematically sought in all patients over 65 years.
More reports from the ESC
If you want to find out more from the ESC, read our report at https://bjcardio.co.uk/2014/10/news-from-the-esc-congress-2014/, and watch our podcast where Professor Keith Fox (University of Edinburgh, and Chairman of the ESC Programme Committee) talks to Dr Afzal Sohaib (Imperial College London, and President of the British Junior Cardiologists’ Association) and discusses the relevance of the following studies in everyday practice:
- in heart failure – PARADIGM-HF and CONFIRM-HF
- in coronary artery disease SIGNIFY, ATLANTIC, CULPRIT, MITOCARE, COMPLETE and TASTE
- in arrhythmia X-VERT and STICS
- and in lipid lowering ODYSSEY and SOLID-TIMI 52.
References
1. Cappato R. Presentation 4945/4946 (X-VERT study) in Hot Line V session coronary artery disease and atrial fibrillation, 2nd September 2014, European Society of Cardiology Congress 2014, Barcelona, Spain, 30th August–3rd September 2014.
2. Darkner S. Presentation 4947/4948 (AMIO CAT study) in Hot Line V session coronary artery disease and atrial fibrillation, September 2nd 2014, European Society of Cardiology Congress 2014, Barcelona, Spain, 30th August–3rd September 2014.
3. Gugliano R, Ruff CT. Presentation of the ENGAGE AF-TIMI 48 subgroup analysis. Press conference, 2nd September 2014, European Society of Cardiology Congress 2014, Barcelona, Spain, 30th August–3rd September 2014.
4. Patel MR. Abstract 4873 (subgroup analysis of ROCKET-AF study) in session Atrial fibrillation: how to improve prognosis, 2nd September 2014, European Society of Cardiology Congress 2014, Barcelona, Spain, 30th August–3rd September 2014.
5. Vanassche T. Presentation 4182 (analysis of 6,563 aspirin-treated patients in ACTIVE or AVERROES) in State of the Art and Featured Research session – new insights into prevention of thromboembolic events in atrial fibrillation, 2nd September 2014, European Society of Cardiology Congress 2014, Barcelona, Spain, 30th August–3rd September 2014.
6. Lip G. Presentation 902 (One-year follow-up of the EURObservational Research Programme) in Registry Hot Line: atrial fibrillation and myocardial infarction, 31st August 2014, European Society of Cardiology Congress 2014, Barcelona, Spain, 30th August–3rd September 2014. (see press release: http://www.escardio.org/about/press/press-releases/esc14-barcelona/Pages/atrial-fibrillation-registry.aspx).
7. Bassand J-P, Goldhaber SZ, Camm AJ et al. Poster presentation 6246 in Anticoagulants and stroke in atrial fibrillation area, 2nd September 2014, European Society of Cardiology Congress 2014, Barcelona, Spain, 30th August–3rd September 2014.
8. Haas S, Goto S, Fitzmaurice D et al. Poster presentation 6256 in Stroke and anticoagulants in atrial fibrillation area, 2nd September 2014, Euorpean Society of Cardiology Congress 2014, Barcelona, Spain, 30th August–3rd September 2014.
9 De Caterina R, Bruggenjurgen B, Darius H et al. Poster presentation 6250 in Anticoagulants and stroke in atrial fibrillation area, 2nd September 2014, European Society of Cardiology Congress 2014, Barcelona, Spain, 30th August–3rd September 2014.
10. Calkins H. Presentation of global survey at press conference 31st August 2014, European Society of Cardiology Congress 2014, Barcelona, Spain, 30th August–3rd September 2014.
11. Davy JM. Presentation of PROFIL FA study, Abstract 4876 in session Atrial fibrillation: how to improve prognosis, 2nd September 2014, European Society of Cardiology Congress 2014, Barcelona, Spain, 30th August–3rd September 2014.



