Hypoglycaemia is defined as a lower than normal level of blood glucose, and in patients on glucose-lowering therapy, defined as glucose levels less than 4 mmol/L. In the UK, it is usually classified as ‘mild’, if the episode is self-treated, or ‘severe’, if the individual requires third-party assistance. However, the American Diabetes Association definition of hypoglycaemia is different.1 They classify hypoglycaemia into five categories. These are shown in table 1.
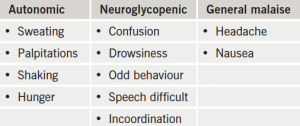
 Symptoms with which patients present vary widely, and include autonomic symptoms, such as sweating, shaking, palpitations, and hunger, or neuroglycopenic symptoms due to cerebral glucose deprivation, such as drowsiness, confusion, odd behaviour and speech disturbances. One of the most commonly used measures of the severity of hypoglycaemia, the Edinburgh Hypoglycaemia Scale, is shown in table 2. Sometimes, patients do not have the classical autonomic or neuroglycopenic symptoms related to hypoglycaemia. To a diabetologist, this hypoglycaemic unawareness is worrying. This is because, while the patient loses their warning symptoms, neurocognitive function remains compromised and their decision-making ability is impaired. It is known that it is most frequently due to recurrent episodes of hypoglycaemia. Almost 19.5% of patients with type 1 diabetes can have an impaired awareness of hypoglycaemia.3 Hypoglycaemic unawareness is more common in the elderly, and in patients with a longer duration of diabetes, and is associated with an almost six-fold increase in the risk of severe hypoglycaemia.4
Symptoms with which patients present vary widely, and include autonomic symptoms, such as sweating, shaking, palpitations, and hunger, or neuroglycopenic symptoms due to cerebral glucose deprivation, such as drowsiness, confusion, odd behaviour and speech disturbances. One of the most commonly used measures of the severity of hypoglycaemia, the Edinburgh Hypoglycaemia Scale, is shown in table 2. Sometimes, patients do not have the classical autonomic or neuroglycopenic symptoms related to hypoglycaemia. To a diabetologist, this hypoglycaemic unawareness is worrying. This is because, while the patient loses their warning symptoms, neurocognitive function remains compromised and their decision-making ability is impaired. It is known that it is most frequently due to recurrent episodes of hypoglycaemia. Almost 19.5% of patients with type 1 diabetes can have an impaired awareness of hypoglycaemia.3 Hypoglycaemic unawareness is more common in the elderly, and in patients with a longer duration of diabetes, and is associated with an almost six-fold increase in the risk of severe hypoglycaemia.4
The balance with glycaemic control

Hypoglycaemia is an under-recognised complication of diabetes management. Hypoglycaemia, and the fear of it, are the major limiting factors in intensifying glycaemic control by titration of insulin and other hypoglycaemic agents in management of type 1 and type 2 diabetes mellitus. Unfortunately, hypoglycaemia is far more common than is usually appreciated by medical staff. Self-reported incidence of severe hypoglycaemia in patients with type 1 diabetes were as high as 3.2 episodes per subject year in patients with long duration of diabetes, and the incidence of mild hypoglycaemia was up to 36 episodes per subject year.5 This could mean an average of two episodes of mild hypoglycaemia per week. In patients with type 2 diabetes on sulfonylureas or insulin for less than two years, the incidences of hypoglycaemia were comparable, at 0.1 and 0.2 episodes per subject year, and the incidence of mild hypoglycaemia was four episodes per subject year.6 Although these numbers seem small, it should be noted that patients with type 2 diabetes on insulin have a much higher rate of hospital admissions related to severe hypoglycaemia compared with patients with type 1 diabetes, probably reflecting patient comorbidities.7
The prevalence of diabetes mellitus is reaching epidemic proportions, with an estimated five million people being diagnosed with the condition by 2025 in the UK.8 Previous work has shown that a large proportion of patients with acute or chronic coronary artery disease have abnormal glucose regulation, with between 50% and 60% of those presenting with acute coronary syndrome (ACS) and hyperglycaemia being previously undiagnosed.9 The link between diabetes mellitus and cardiovascular disease is also well established. In relation to hospital admission, there is a greater than 75% increased risk of angina, a 55% increased risk of myocardial infarction, a 73% increased risk of heart failure and a 34% increased risk of stroke among people with both types of diabetes.10
The Diabetes Control and Complications Trial and the UK Prospective Diabetes Study, showed conclusively that good glycaemic control decreased long-term complications in patients with newly diagnosed type 2 and type 1 diabetes, respectively.11,12 These trials set the goal for clinicians to try and achieve normoglycaemia in patients with diabetes, with aggressive targets. However, in both studies, there was an increase in hypoglycaemia rates with a lowering of glycosylated haemoglobin (HbA1c). This was, of course, offset by the major reductions in the risk of developing micro- and macrovascular disease. More recent work looking at further, aggressive lowering of HbA1c in patients with long-standing type 2 diabetes have shown that intensive treatment is associated with much higher incidence of hypoglycaemia.13,14 Indeed, the finding of higher mortality in the intensive-therapy group in the ACCORD (Action to Control Cardiovascular Risk in Diabetes) trial led to a discontinuation of intensive therapy in this study.13 The largest single category of deaths in the ACCORD trial was termed ‘unexpected/presumed cardiovascular disease’. While this group had a statistically greater number of severe hypoglycaemic events (p=0.04), they also had a statistically significant decrease in non-fatal myocardial infarctions (p=0.004).13 The other study also showed similar results, with a definite increase in the number of sudden deaths, but this was not statistically significant.14 Thus, intensification of glycaemic control has been shown to be of benefit in people within the first 7–10 years of diagnosis, but after that, abrupt tightening is associated with increased harm.
Impact on the heart
Acute hypoglycaemia provokes a surge of counter regulatory hormones, in particular glucagon and adrenaline, and to a lesser extent growth hormone, vasopressin, noradrenaline and cortisol. This marked sympathetic response leads to haemodynamic changes with the priorities being the maintenance of cerebral perfusion, and splanchnic perfusion to maintain gluconeogenesis. This haemodynamic change causes an increase in cardiac stress – an increase in heart rate and systolic blood pressure, decrease in peripheral arterial resistance and widening pulse pressure. It also increases myocardial contractility and stroke volume. This increase in cardiac stress probably has little impact in someone with normal cardiovascular reserve. However, it can have significant impact on patients who have pre-existing ischaemic heart disease. Furthermore, hypoglycaemia also stimulates an increase in inflammatory cytokines, such as C-reactive protein, interleukin (IL)-1 and IL-6, tumour necrosis factor α and endothelin 1.15 This results in endothelial injury and changes in the coagulation cascade increasing the risk of cardiovascular events. Hypoglycaemia also stimulates platelet aggregation and platelet activation.16,17 Together, these changes could explain the anecdotal case reports of hypoglycaemia precipitating angina and ACS.
Experimental hypoglycaemia causes ST-wave changes, as well as lengthening of the QT interval and cardiac repolarisation.18 This leads to a decrease in T-wave amplitude and an increase in length of the T-wave, and is reflected by an increase in QTc interval, and could potentially trigger more dangerous tachyarrhythmias, such as ventricular tachycardia and atrial fibrillation, or even sinus bradycardia and asystole.
This effect is augmented in someone with pre-existing prolongation of the QTc interval, such as patients with autonomic neuropathy or genetic predisposition. In addition, many patients with end-organ damage, such as peripheral neuropathy, may be on drugs for their pain that are also known to prolong the QT interval. For example, patients with foot ulcers have been shown to have a prolonged QTc interval and that this is associated with premature mortality.19 While many drugs may cause this prolongation, it may also be a sign of underlying ischaemic heart disease. Acute changes in the QTc interval are thought to be consequent to the hypokalaemia caused by the abundant catecholamine release induced by the hypoglycaemic episode. In an experimental setting, these effects have been found to be attenuated by potassium replacement and beta blockers. A recent study looking at electrocardiogram (ECG) changes during hypoglycaemia in patients with type 2 diabetes, showed an increased incidence of arrhythmias, bradycardia, atrial and ventricular ectopics, especially during nocturnal hypoglycaemia.20
There are further layers of complexity. Several agents used to treat diabetes do not lower blood glucose – they stop glucose levels from rising. A recent study showed a high incidence of asymptomatic episodes of hypoglycaemia with blood glucose levels of <3.1 mmol/L in patients receiving treatment with insulin and/or sulfonylureas – agents that lower blood glucose. However, severe hypoglycaemia did not develop in any of the control subjects on metformin and/or dipeptidyl peptidase-4 (DPP4) inhibitors – drugs that stop glucose levels from rising. Patients in whom blood glucose levels dropped to <3.1 mmol/L had a higher number of ventricular arrhythmias.21 An analysis of over 250,000 patients showed that the risk of death of someone on a sulfonylurea was more than double that of someone on metformin.22
The ‘Dead in Bed’ syndrome generated a lot of interest in the 1980s and 1990s. Several theories were postulated to try to explain the mysterious and disturbing deaths of otherwise healthy young patients with type 1 diabetes.23 These young people had retired to bed under normal circumstances and were found dead in an undisturbed bed the next morning. Circumstantial evidence pointed to hypoglycaemia, or an event related to it, to be responsible.24 These authors also describe the new evidence to support that repeated hypoglycaemia stimulates orexin neurons in the hypothalamus, causing defective awakening and hypotonia of the upper airway muscles. They postulate that these abnormalities lead to an increase in sympathetic tone, variations in intrathoracic pressure, cardiac arrhythmias and depression of ventilation.24 It is thought that these factors lead to cardio-respiratory arrest and sudden death during sleep. Patients with diabetes have been found to be at least three to four times higher risk for a sudden death compared with a non-diabetic population.25
Conclusion
In summary, hypoglycaemia is common in individuals on glucose-lowering drugs and should be considered a cardiovascular risk factor in itself. In addition to diabetes being a risk factor for premature cardiovascular mortality, hypoglycaemia is also associated with an increased risk of cardiovascular death. Cardiologists need to work in collaboration with their diabetes colleagues to gain better awareness of the condition and what needs to be done to minimise its frequency and impact.
Funding
All of the authors are full-time employees of the UK National Health Service.
Conflict of interest
None declared.
References
1. Seaquist ER, Anderson J, Childs B et al. Hypoglycemia and diabetes: a report of a workgroup of the American Diabetes Association and The Endocrine Society. Diabetes Care 2013;36:1384–95. http://dx.doi.org/10.2337/dc12-2480
2. Deary IJ, Hepburn DA, MacLeod KM, Frier BM. Partitioning the symptoms of hypoglycaemia using multi-sample confirmatory factor analysis. Diabetologia 1993;36:771–7. http://dx.doi.org/10.1007/BF00401150
3. Geddes J, Schopman JE, Zammitt NN, Frier BM. Prevalence of impaired awareness of hypoglycaemia in adults with type 1 diabetes. Diabet Med 2008;25:501–04. http://dx.doi.org/10.1111/j.1464-5491.2008.02413.x
4. Gold AE, MacLeod KM, Frier BM. Frequency of severe hypoglycemia in patients with type I diabetes with impaired awareness of hypoglycemia. Diabetes Care 1994;17:697–703. http://dx.doi.org/10.2337/diacare.17.7.697
5. UK Hypoglycaemia Study Group. Risk of hypoglycaemia in types 1 and 2 diabetes: effects of treatment modalities and their duration. Diabetologia 2007;50:1140–7. http://dx.doi.org/10.1007/s00125-007-0599-y
6. Amiel SA, Dixon T, Mann R, Jameson K. Hypoglycaemia in type 2 diabetes. Diabet Med 2008;25:245–54. http://dx.doi.org/10.1111/j.1464-5491.2007.02341.x
7. Stanisstreet D, Walden E, Jones C et al. The hospital management of hypoglycaemia in adults with diabetes mellitus. London: Joint British Diabetes Societies Inpatient Care Group for NHS Diabetes, 2010. Available from: http://www.diabetologists-abcd.org.uk/subsite/JBDS_IP_Hypo_Adults_Revised.pdf
8. International Diabetes Federation. IDF diabetes atlas, 6th edition. Brussels, Belgium: IDF, 2013. Available from: http://www.idf.org/diabetesatlas
9. Schiele F, Bueno H, Hochadel M et al. Admission hyperglycemia improves the GRACE risk score for prediction of in-hospital mortality: insights from the Euro Heart Survey ACS III. J Am Coll Cardiol 2014;63:A47. http://dx.doi.org/10.1016/S0735-1097(14)60047-4
10. Diabetes UK. Diabetes: facts and stats. Version 3. March 2014. Available from: http://www.diabetes.org.uk/Documents/About%20Us/Statistics/Diabetes-key-stats-guidelines-April2014.pdf
11. The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 1993;329:977–86. http://dx.doi.org/10.1056/NEJM199309303291401
12. UK Prospective Diabetes Study Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 1998;352:837–53. http://dx.doi.org/10.1016/S0140-6736(98)07019-6
13. ACCORD study group. Long-term effects of intensive glucose lowering on cardiovascular outcomes. N Engl J Med 2011;364:818–28. http://dx.doi.org/10.1056/NEJMoa1006524
14. Duckworth W, Abraira C, Moritz T et al. Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med 2009;360:129–39. http://dx.doi.org/10.1056/NEJMoa0808431
15. Lalic NM. The case for: hypoglycemia is of cardiovascular importance. Diabetes Care 2013;36:S264–S266. http://dx.doi.org/10.2337/dcS13-2016
16. Desouza CV, Bolli GB, Fonseca V. Hypoglycemia, diabetes, and cardiovascular events. Diabetes Care 2010;33:1389–94. http://dx.doi.org/10.2337/dc09-2082
17. Hanefeld M, Duetting E, Bramlage P. Cardiac implications of hypoglycaemia in patients with diabetes – a systematic review. Cardiovasc Diabetol 2013;12:135. http://dx.doi.org/10.1186/1475-2840-12-135
18. Frier BM, Schernthaner G, Heller SR. Hypoglycemia and cardiovascular risks. Diabetes Care 2011;34:S132–S137. http://dx.doi.org/10.2337/dc11-s220
19. Fagher K, Londahl M. The impact of metabolic control and QTc prolongation on all-cause mortality in patients with type 2 diabetes and foot ulcers. Diabetologia 2013;56:1140–7. http://dx.doi.org/10.1007/s00125-013-2860-x
20. Chow E, Bernjak A, Williams S et al. Hypoglycemia in patients with type 2 diabetes and cardiovascular risk. Diabetes 2014;63:1738–47. http://dx.doi.org/10.2337/db13-0468
21. Stahn A, Pistrosch F, Ganz X et al. Relationship between hypoglycemic episodes and ventricular arrhythmias in patients with type 2 diabetes and cardiovascular diseases: silent hypoglycemias and silent arrhythmias. Diabetes Care 2014;37:516–20. http://dx.doi.org/10.2337/dc13-0600
22. Roumie CL, Greevy RA, Grijalva CG et al. Association between intensification of metformin treatment with insulin vs sulfonylureas and cardiovascular events and all-cause mortality among patients with diabetes. JAMA 2014;311:2288–96. http://dx.doi.org/10.1001/jama.2014.4312
23. Heller SR. Abnormalities of the electrocardiogram during hypoglycaemia: the cause of the dead in bed syndrome? Int J Clin Pract 2002;129:27–32.
24. Parekh B. The mechanism of dead-in-bed syndrome and other sudden unexplained nocturnal deaths. Curr Diabetes Rev 2010;5:210–15. http://dx.doi.org/10.2174/157339909789804387
25. Wright RJ, Frier BM. Vascular disease and diabetes: is hypoglycaemia an aggravating factor? Diabetes Metab Res Rev 2008;24:353–63. http://dx.doi.org/10.1002/dmrr.865

