Pitfalls in the interpretation of cardiopulmonary exercise testing data
Dear Sirs
Cardiopulmonary exercise testing (CPET) is non-invasive, safe,1 and reproducible.2 CPETs provide objective information on exercise capacity. Peak oxygen uptake (VO2 peak) – the primary parameter when investigating a decrease in exercise tolerance – is affected by many cardiac and non-cardiac diseases and is a validated independent predictor for hospitalisation and death.3–5 The oxygen uptake efficiency slope (OUES) is another valuable measurement to assess exercise tolerance and is valid even when patients are not able to exercise till their maximum.
In daily practice, CPET data can be difficult to interpret. Several different formulae are available to predict normal exercise tolerance. These formulae provide a variety of results as they use different input functions and mathematical assumptions. The 2003 American Thoracic Society/American College of Chest Physicians (ATS/ACCP) statement6 acknowledges the absence of optimal CPET reference values and recommends that each exercise laboratory select an appropriate set of reference values, but failed to state how to make this selection. Different statements have recommended the use of different formulae. The validity of reference ranges has been noted to be uncertain in older patients, tall patients, in over- or underweight patients, and in women.7 All formulae use similar demographic factors and make adjustment in the same direction. When applied to a typical cardiac patient, however, the formulas give markedly different results.
Applying formulae to clinical patients
Seven different formulae for VO2 peak (Wasserman,7 Neder,8 Hansen,6 Jones,9 Koch10) and for OUES (Hollenberg,11 Buys12) were applied to 111 patients with systolic heart failure (n=33), patients with paroxysmal atrial fibrillation (n=69) and healthy individuals (n=9), who were participating in previous studies undertaken at our institution. When the formulae were applied to our patient population, a large variety in predicted values was observed. One patient in this study, for example, achieved a VO2 peak of 290% predicted in the Jones formula, but only 72% predicted by the Koch formula. All formulae, however, except the Wasserman’s, returned either a maximal or minimal value for at least one patient, highlighting that the extent of discrepancies applies to all formulas.
The ATS/ACCP statement suggests a threshold of >84% for normal predicted VO2 peak,6 In 44 out of 111 (40%) patients, one formula suggested predicted peak exercise capacity was over 100%, whilst another suggested it was below the 84% threshold predicted. This could have dramatic effect when interpreting CPET results as patients could be wrongly labelled as having a normal or abnormal VO2 peak. Figure 1 shows output of all seven formulae displayed for each individual. These are ordered in age order to emphasise that there is variation observed across age.
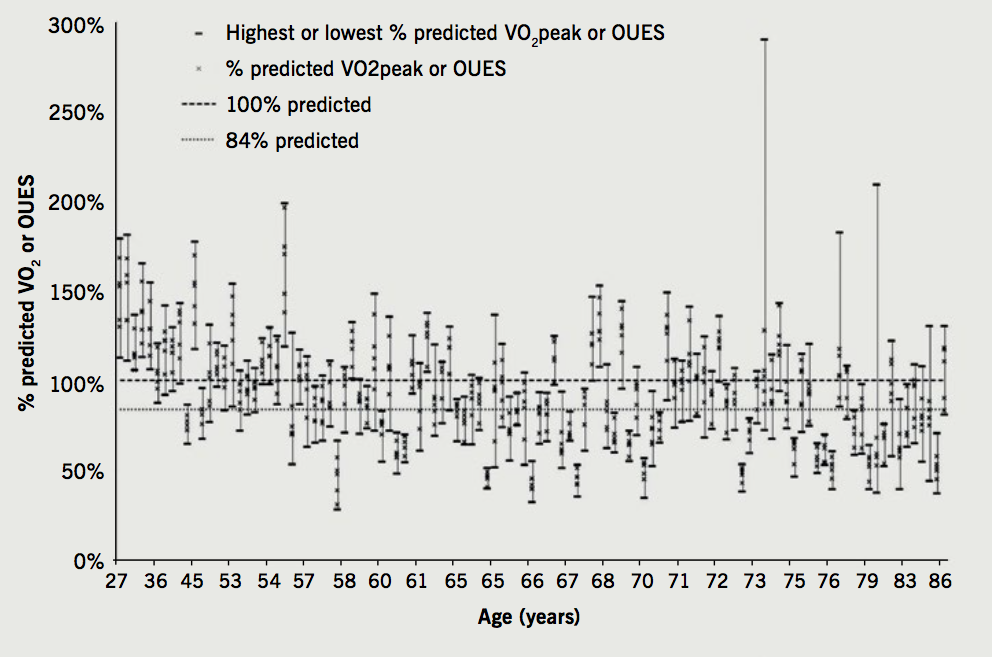
The ATS/ACCP statement6 noted several methodological limitations in studies reporting reference ranges. These included small sample size, retrospective study design, lack of randomisation and inclusion of smokers. Furthermore, the commonly-used Wasserman formula is a modified form of the Hansen formula, but the rationale and evidence basis for the modification has not yet been published or peer-reviewed. Moreover, studies13 are still emerging computing new predicting equations to better fit certain populations, demonstrating the need for an improved validated predicting equation. The authors of the Koch formula have noted systematic differences between their estimates and others’, particularly in older patients.14 The Buys formula for OUES was based on volunteers aged 20 to 60, and so may not be applicable to the patients within this analysis. A large discrepancy was observed between the Buys and the Hollenberg formula for OUES.
Implications for daily clinical practice
Across various populations, from normal controls to those with advanced heart failure, different references ranges for peak exercise capacity can give markedly differing results. These discrepancies between formulae may have significant and important implications for the interpretation of CPET reports in the clinical setting and in comparing studies published in the literature. Until more research is done to clarify the formulae that best predicts clinical outcome in these populations, physicians should consider percentage predicted VO2 and OUES parameters in their clinical decision-making with caution. In particular, percentage predicted from other equipment or between institutions needs to be treated with extreme caution. Proper reference ranges with consistent methodology should be developed which will permit the accurate, reliable reproducible use of CPET in daily cardiac assessment.
Acknowledgement
We would like to acknowledge Mrs Sarah Anderson BSc (Hons) in the data collection process for both studies conducted in our trust.
Conflict of interest
None declared.
References
1. Skalski J, Allison TG, Miller TD. The safety of cardiopulmonary exercise testing in a population with high-risk cardiovascular diseases. Circulation 2012;126:2465–72. http://doi.org/10.1161/CIRCULATIONAHA.112.110460
2. Barron A, Dhutia N, Mayet J, Hughes AD, Francis DP, Wensel R. Test-retest repeatability of cardiopulmonary exercise test variables in patients with cardiac or respiratory disease. Eur J Prev Cardiol 2014;21:445–53. http://doi.org/10.1177/2047487313518474
3. Weisman I, Zeballos R. An integrated approach to the interpretation of cardiopulmonary exercise testing. Clin Chest Med 1994;15:421–45.
4. Coeckelberghs E, Buys R, Goetschalckx K, Cornelissen VA, Vanhees L. Prognostic value of the oxygen uptake efficiency slope and other exercise variables in patients with coronary artery disease. Eur J Prev Cardiol 2016;23:237–44. http://doi.org/10.1177/2047487315569410
5. Piepoli M, Corra U, Agostini P. Statement on cardiopulmonary exercise testing in chronic heart failure due to left ventricular dysfunction: recommendations for performance and interpretation Part III: Interpretation of cardiopulmonary exercise testing in chronic heart failure and future applications: Task Force of the Italian Working Group on Cardiac Rehabilitation and Prevention (Gruppo Italiano di Cardiologia Riabilitativa e Prevenzione, GICR), endorsed by Working Group on Cardiac Rehabilitation and Exercise Physiology of the European Society of Cardiology. Eur J Cardiovasc Prev Rehabil 2006;13:485–94.
6. American Thoracic Society/American College of Chest Physicians. Statement on cardiopulmonary exercise testing. Am J Respir Crit Care Med 2003;167:211–77. http://doi.org/10.1164/rccm.167.2.211
7. Wasserman K (editor). Principles of exercise testing and interpretation: including pathophysiology and clinical applications, 4th ed. Philadelphia: Lippincott Williams & Wilkins; 2005.
8. Neder J, Nery L, Castelo A, et al. Prediction of metabolic and cardiopulmonary responses to maximum cycle ergometry: a randomised study. Eur Respir J 1999;14:1304–13.
9. Jones NL, Makrides L, Hitchcock C, Chypchar T, McCartney N. Normal standards for an incremental progressive cycle ergometer test. Am Rev Respir Dis 1985;131:700–8.
10. Koch B, Schaper C, Ittermann T, et al. Reference values for cardiopulmonary exercise testing in healthy volunteers: the SHIP study. Eur Respir J 2008;33:389–97. http://doi.org/10.1183/09031936.00074208
11. Hollenberg M, Tager IB. Oxygen uptake efficiency slope: an index of exercise performance and cardiopulmonary reserve requiring only submaximal exercise. J Am Coll Cardiol 2000;36:194–201.
12. Buys R, Coeckelberghs E, Vanhees L, Cornelissen VA. The oxygen uptake efficiency slope in 1411 Caucasian healthy men and women aged 20-60 years: reference values. Eur J Prev Cardiol 2015;22:356-63. http://doi.org/10.1177/2047487314547658
13. Paap D, Takken T. Reference values for cardiopulmonary exercise testing in healthy adults: a systematic review. Expert Rev Cardiovasc Ther 2014;12:1439–53. http://doi.org/10.1586/14779072.2014.985657
14. Koch B, Schäper C, Völzke H, Ewert R, Gläser S. From the authors. Eur Respir J 2009;34:287–8.

