Glucagon-like peptide-1 (GLP-1) receptor agonists are an injectable glucose-lowering therapy used in the treatment of type 2 diabetes mellitus (T2DM). Alongside dipeptidyl peptidase-4 (DPP-4) inhibitors, they exert their effect by augmenting the incretin pathway. GLP-1 receptor agonists offer reductions in glycosylated haemoglobin (HbA1c) and weight alongside a low risk of hypoglycaemia. The cardiovascular safety of GLP-1 receptor agonists in patients at elevated cardiovascular risk has been examined in large double-blind, placebo-controlled cardiovascular safety trials. Liraglutide demonstrated superiority over placebo with reduced major adverse cardiovascular events (MACE) in the treatment group. Exenatide, lixisenatide and semaglutide have shown non-inferiority versus placebo for MACE. These trials demonstrated no association between GLP-1 agonist therapy and hospitalisation for heart failure.
Pharmacology
Glucagon-like peptide-1 (GLP-1) is a peptide hormone released from the intestinal L-cells in response to oral glucose ingestion. Alongside glucose-dependent insulinotropic polypeptide (GIP), it is one of two key incretin hormones, which potentiate glucose-dependent insulin release.1 Additional favourable effects of GLP-1 include suppression of glucagon secretion, slowing of gastric emptying and increased satiety. GLP-1 receptor agonists replicate the physiological role of endogenous GLP-1. They have changes to the amino acid sequence that make them resistant to breakdown by the enzyme dipeptidyl peptidase-4 (DPP-4) plus some have structural properties allowing them a more prolonged effect. At present, there are five GLP-1 receptor agonists licensed for use in the UK (table 1).
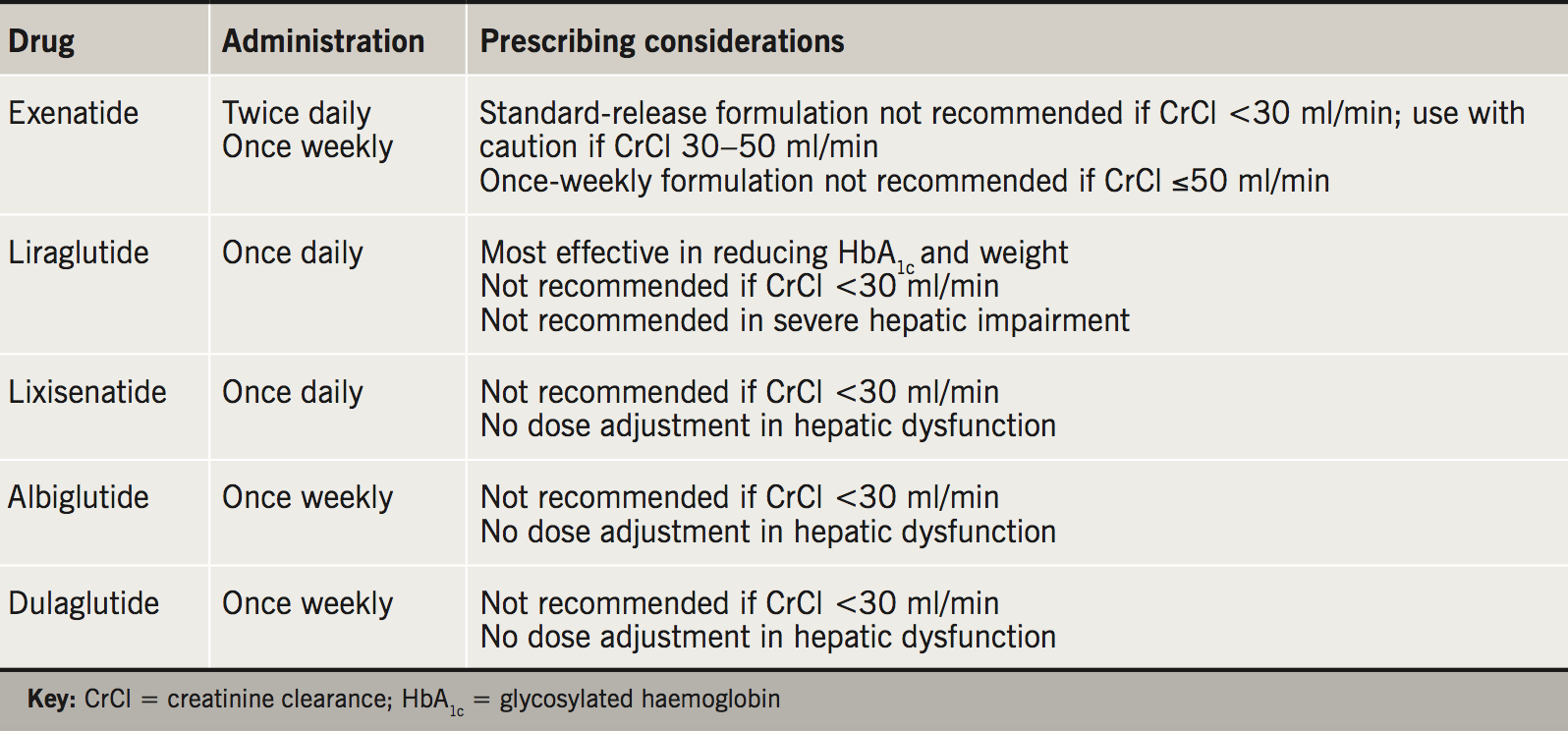
GLP-1 receptor agonists are administered as a subcutaneous preparation. They offer a glycosylated haemoglobin (HbA1c) reduction of approximately 1% (11 mmol/mol) and carry a low risk of hypoglycaemia.2 Their major advantage is weight loss of 2 to 4 kg, on average.3 The most common side effect is gastrointestinal upset, which leads to discontinuation of therapy in around 10% of patients. There is insufficient evidence to confirm a suspected association between the use of GLP-1 receptor agonists and pancreatitis or pancreatic cancer.4,5
The Scottish Intercollegiate Guidelines Network (SIGN) and National Institute for Health and Care Excellence (NICE) recommend GLP-1 receptor agonists as a third-line therapy for obese patients with type 2 diabetes mellitus (T2DM).6,7 They are recommended, in addition to metformin and sulfonylurea, for patients with body mass index (BMI) ≥30 kg/m2 (SIGN) or ≥35 kg/m2 (NICE). Additionally, NICE acknowledge patients with a BMI below 35 kg/m2 may benefit if they have obesity-related comorbidities, or if insulin would have significant occupational implications. NICE recommend the treatment is only continued beyond six months if there has been a reduction in HbA1c of 1% (11 mmol/mol) and weight loss of 3% body weight.7
ELIXA
ELIXA (Evaluation of Cardiovascular Outcomes in Patients With Type 2 Diabetes After Acute Coronary Syndrome During Treatment With Lixisenatide) assessed the safety and efficacy of lixisenatide versus placebo in 6,068 patients with T2DM and recent acute coronary syndrome (ACS).8 Participants were diagnosed with ACS, defined as myocardial infarction (ST-elevation or non-ST-elevation) or hospitalisation for unstable angina, in the 180 days preceding randomisation. The study randomised participants from 49 countries in a 1:1 ratio to receive lixisenatide (10 µg daily ± titration to 20 µg daily) for a median follow-up period of 25 months. The mean baseline HbA1c was 7.7%. Exclusion criteria included coronary artery bypass graft surgery performed for the qualifying event, or a planned coronary revascularisation procedure within 90 days of screening. Baseline characteristics included heart failure in 22% of patients and previous ACS prior to the index event in 22%.
The primary outcome of major adverse cardiovascular events (MACE), comprising nonfatal myocardial infarction, nonfatal stroke, hospitalisation for unstable angina or cardiovascular death, occurred in 406 (13.4%) patients in the lixisenatide group and 399 (13.2%) patients in the placebo group (hazard ratio [HR] 1.02; 95% confidence interval [CI] 0.89 to 1.17; p<0.001 for non-inferiority, p=0.81 for superiority). There were no significant between-group differences in rates of heart failure hospitalisation (4.2% vs. 4%; HR 0.96; 95% CI 0.75 to 1.23; p=0.75), or for a secondary end point comprising the primary outcome, heart failure hospitalisation and coronary revascularisation.
LEADER
LEADER (Liraglutide Effect and Action in Diabetes: Evaluation of Cardiovascular Outcome Results) was a large cardiovascular outcome trial (CVOT) that assessed the cardiovascular safety of liraglutide compared with placebo.9 The study recruited 9,340 patients with T2DM (mean baseline HbA1c 8.7%) from 410 centres in 32 countries. Patients were randomised to receive liraglutide (1.8 mg once daily or maximum tolerated dose) or placebo in a 1:1 ratio with median follow-up of 3.8 years. Participants were aged ≥50 years with one or more established cardiovascular diagnosis (coronary artery disease, cerebrovascular disease, peripheral vascular disease, chronic kidney disease stage III or greater, or New York Heart Association [NYHA] class II or III chronic heart failure) or aged ≥60 years with at least one cardiovascular risk factor (microalbuminuria or proteinuria, hypertension and left ventricular hypertrophy, left ventricular systolic or diastolic dysfunction, or an ankle–brachial pressure index <0.9). Baseline characteristics included established cardiovascular disease in 81% of patients and heart failure in 14% of patients.
The primary MACE end point (comprising cardiovascular death, nonfatal myocardial infarction and nonfatal stroke) occurred in significantly fewer patients in the liraglutide group than in the placebo group (13% vs. 14.9%; HR 0.87; 95% CI 0.78 to 0.97; p<0.001 for non-inferiority, p=0.01 for superiority). This was driven by a significant reduction in cardiovascular death (4.7% vs. 6%; HR 0.78; 95% CI 0.66 to 0.93; p=0.007) with no significant reduction seen in the other components of the primary end point (nonfatal myocardial infarction and nonfatal stroke). Additionally, death from any cause was significantly reduced in the liraglutide group compared with the placebo group (8.2% vs. 9.6%; HR 0.85; 95% CI 0.74 to 0.97; p=0.02). There was no significant difference in the rate of heart failure hospitalisation between groups.
SUSTAIN-6
SUSTAIN-6 (Trial to Evaluate Cardiovascular and Other Long-term Outcomes With Semaglutide in Subjects With Type 2 Diabetes) assessed the cardiovascular safety of semaglutide in a large, randomised, placebo-controlled trial.10 This drug is not currently licensed and the trial was performed as a pre-licensing safety study. There were 3,297 patients with T2DM randomised to receive once-weekly semaglutide (0.5 mg or 1 mg) or placebo in a 1:1:1 ratio for 104 weeks. Patients were aged ≥50 years with established cardiovascular disease (coronary artery disease, cerebrovascular disease or peripheral vascular disease), chronic heart failure (NYHA class II or III) or chronic kidney disease (stage III or greater). Patients aged ≥60 years with one cardiovascular risk factor (persistent microalbuminuria [30 to 299 mg/g or proteinuria], hypertension and left ventricular hypertrophy, left ventricular systolic or diastolic dysfunction or ankle–brachial pressure index <0.9) were also included. Baseline characteristics included previous myocardial infarction in 32.5% of patients and heart failure in 23.6% of patients.
The primary MACE outcome, a composite of cardiovascular death, nonfatal myocardial infarction and nonfatal stroke, occurred in 108 (6.6%) patients treated with semaglutide compared with 146 (8.9%) patients treated with placebo (HR 0.74; 95% CI 0.58 to 0.95; p<0.001 for non-inferiority, p=0.02 for superiority). An expanded composite outcome comprising revascularisation (coronary or peripheral) and hospitalisation for unstable angina or heart failure, in addition to the primary outcome criteria, occurred in 199 patients receiving semaglutide and 264 patients receiving placebo (HR 0.74; 95% CI 0.62 to 0.89; p=0.03). The risk of nonfatal stroke was significantly reduced in the semaglutide group compared with placebo (1.6% vs. 2.7%; HR 0.61; 95% CI 0.38 to 0.99; p=0.04). There was no significant between-group difference in nonfatal myocardial infarction, cardiovascular death or hospitalisation for heart failure. As superiority was not a pre-specified outcome, only non-inferiority can be concluded from the aforementioned results.
EXSCEL
EXSCEL (Exenatide Study of Cardiovascular Event Lowering Trial) is the largest and most recently published of the GLP-1 receptor agonist CVOTs.11 The results were presented on 14 September 2017 at the European Association for the Study of Diabetes annual meeting, and published simultaneously online. The study randomised 14,752 patients to receive extended-release exenatide 2 mg or placebo once weekly for a median of 3.2 years follow-up. Of the participants, 73.1% had existing cardiovascular disease and 16.2% had a history of heart failure.
The primary outcome, a MACE composite of cardiovascular death, nonfatal myocardial infarction and nonfatal stroke, occurred in similar numbers of patients in the exenatide and placebo groups (11.4% vs. 12.2%; HR 0.91; 95% CI 0.83 to 1.00). Rates of heart failure hospitalisation were also similar between groups (3% vs. 3.1%).
The EXSCEL trial was designed with elements of a ‘pragmatic’ trial and subjects were seen every six months by the investigators. A major deficiency of the trial is a very high rate of discontinuation (45% in the placebo group, 43% in the exenatide group). As a consequence, there was a lower than expected exposure to the intervention. There was a reduction in all-cause mortality as a secondary outcome, with a nominal p value of 0.016. It is uncertain what the overall results would have been if more subjects had persisted with the allocated intervention. Thus, non-inferiority has been demonstrated but any claim for superiority should be considered inconclusive.
Discussion
The GLP-1 receptor agonist liraglutide has demonstrated superiority over placebo in reducing a primary composite MACE end point through its impact on reduced cardiovascular deaths. Similar effects were seen in SUSTAIN-6 but not ELIXA or EXSCEL. Lixisenatide and exenatide have amino acid structures that differ considerably from native GLP-1, whereas liraglutide has a structure that is closer to native GLP-1. At this point in time it is unknown whether the differing results are explained by differences in the drugs, differences in the subjects that were included in the studies, or differences in study design and conduct.
In animal models, GLP-1 receptors have been identified in widespread sites including the pancreatic islet cells, liver, brain, kidneys, heart and vascular endothelium. Evidence from animal studies suggests that GLP-1 receptor agonists act directly on these target organs, exerting an insulin-independent effect. Multiple favourable effects of GLP-1 receptor stimulation have been demonstrated in animal models, including improved endothelial function, sodium excretion, recovery from ischaemic injury and myocardial function.12 Similar observations have been made in small, open-label phase 3 clinical trials examining intravenous GLP-1 infusion in patients with and without diabetes mellitus.12
In the LEADER trial, treatment with liraglutide was associated with benefits in several cardiovascular variables.9 After three years of follow-up, mean weight loss was 2.3 kg greater (95% CI 2.0 to 2.5 kg) and mean systolic blood pressure was 1.2 mmHg lower (95% CI 0.5 to 1.9 mmHg) in the liraglutide group than in the placebo group. In addition, the incidence of renal microvascular events (defined as new macroalbuminuria or doubling of serum creatinine and estimated glomerular filtration rate [eGFR] ≤45 ml/min/1.73m2, need for renal replacement therapy or death from renal disease) was reduced in the liraglutide group (1.5 vs. 1.9 events per 100 patient-years of observation; HR 0.78; 95% CI 0.67 to 0.92; p=0.003). However, liraglutide was also associated with small but significant increases in mean resting heart rate (3.0 beats per minute; 95% CI 2.5 to 3.4) and diastolic blood pressure (mean increase 0.6 mmHg; 95% CI 0.2 to 1.0). While the cardiovascular benefit of liraglutide may be accrued from small positive effects in various metabolic parameters, further study is required. Large cardiovascular outcome trials involving albiglutide and dulaglutide are ongoing and will influence future guidance on GLP-1 receptor agonist use (table 2).

Key messages
- Glucagon-like peptide-1 (GLP-1) receptor agonists are an injectable therapy licensed for use in obese patients with type 2 diabetes mellitus
- Liraglutide was associated with a significantly reduced risk of major adverse cardiovascular events (MACE) compared with placebo in a large cardiovascular outcome trial involving patients with established cardiovascular disease (CVD)
- Three other GLP-1 receptor agonists were associated with no increase in MACE when compared with placebo in randomised-controlled trials: exenatide (in patients with and without CVD), semaglutide (in patients with CVD) and lixisenatide (in patients with CVD and recent acute coronary syndrome)
- There has been no association between GLP-1 receptor agonists and heart failure hospitalisation observed in these trials
Conflict of interest
MF has received payment for lectures and advisory boards from Astra Zeneca, Eli Lilly, GSK, Novo Nordisk, and Sanofi. GM has received payment for lectures and advisory boards from Astra Zeneca, Eli Lilly, Novo Nordisk and Sanofi. EJ: none declared.
Editors’ note
This article is the fourth in the series ‘drugs for diabetes’. Previous articles have covered dipeptidyl peptidase-4 (DPP-4) inhibitors (doi:10.5837/bjc.2017.001), SGLT2 inhibitors (doi:10.5837/bjc.2017.010) and glitazones (thiazolidinediones) (doi:10.5837/bjc.2017.018). Subsequent articles in the series cover Older antidiabetic drugs (doi: 10.5837/bjc.2018.007) and glucose-lowering drugs for patients with cardiac disease (doi:10.5837/bjc.2018.016).
References
1. Holst JJ, Ørskov C. The incretin approach for diabetes treatment. Diabetes 2004;53:S197–S204. https://doi.org/10.2337/diabetes.53.suppl_3.S197
2. Amori RE, Lau J, Pittas AG. Efficacy and safety of incretin therapy in type 2 diabetes. JAMA 2007;298:194–206. https://doi.org/10.1001/jama.298.2.194
3. Vilsboll T, Christensen M, Junker AE, Knop FK, Gluud LL. Effects of glucagon-like peptide-1 receptor agonists on weight loss: systematic review and meta-analyses of randomised controlled trials. BMJ 2012;344:d7771. https://doi.org/10.1136/bmj.d7771
4. Chalmer T, Almdal TP, Vilsbøll T, Knop FK. Adverse drug reactions associated with the use of liraglutide in patients with type 2 diabetes – focus on pancreatitis and pancreas cancer. Expert Opin Drug Saf 2015;14:171–80. https://doi.org/10.1517/14740338.2015.975205
5. Jensen TM, Saha K, Steinberg WM. Is there a link between liraglutide and pancreatitis? A post hoc review of pooled and patient-level data from completed liraglutide type 2 diabetes clinical trials. Diabetes Care 2015;38:1058–66. https://doi.org/10.2337/dc13-1210
6. Scottish Intercollegiate Guidelines Network (SIGN). Management of diabetes. Guideline 116. Edinburgh: SIGN, 2010. Available from: http://www.sign.ac.uk/sign-116-management-of-diabetes.html
7. National Institute for Health and Care Excellence. Type 2 diabetes in adults: management. NICE guideline 28. London: NICE, 2015. Available from: https://www.nice.org.uk/guidance/ng28
8. Pfeffer MA, Claggett B, Diaz R et al. Lixisenatide in patients with type 2 diabetes and acute coronary syndrome. N Engl J Med 2015;373:2247–57. https://doi.org/10.1056/NEJMoa1509225
9. Marso SP, Daniels GH, Brown-Frandsen K et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2016;375:311–22. https://doi.org/10.1056/NEJMoa1603827
10. Marso SP, Bain SC, Consoli A et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med 2016;375:1834–44. https://doi.org/10.1056/NEJMoa1607141
11. Holman RR, Bethel MA, Mentz RJ et al. EXSCEL Study Group. Effects of once-weekly exenatide on cardiovascular outcomes in type 2 diabetes. N Engl J Med 2017;377:1228–39. https://doi.org/10.1056/NEJMoa1612917
12. Okerson T, Chilton RJ. The cardiovascular effects of GLP-1 receptor agonists. Cardiovasc Ther 2012;30:e146–e155. https://doi.org/10.1111/j.1755-5922.2010.00256.x

