The National Institute for Health and Care Excellence (NICE) and NHS England have shown a commitment to embedding shared decision-making (SDM) in clinical practice and developing decision aids based on clinical guidelines. Healthcare policy makers are keen to enhance the engagement of patients in SDM in the belief that it improves the benefits accrued from healthcare interventions. This may be important for interventions such as implantable cardioverter-defibrillator (ICD) implantation, where cost-effectiveness is under scrutiny. NHS England invited the ICD implanters in the north of England to participate in a regional commissioning quality incentive (CQUIN) project to improve decision-making around a primary prevention ICD implant. A collaborative project included the development of a specific SDM tool, the first of its kind in the UK, followed by training and education of the clinical teams. The project illustrates that this approach is practical and deliverable and could be applied and used in other regions, and considered in additional clinical areas.
Introduction
Most clinicians support the idea of person-centred care as a model of best practice, yet we know from published research and National Health Service (NHS) patient surveys that people still want to be more involved in decisions about their healthcare.1,2 In shared decision-making (SDM), healthcare professionals and individuals work together to select tests, treatments, management or support packages based on evidence and the individual’s informed preferences.3 This evidence should be the best available assessment of the likely benefits, risks, and outcomes of the options, with the individual’s values and preferences being central to the decision.
NICE (National Institute for Health and Care Excellence) leads a SDM Collaborative, involving multiple members, including NHS England to encourage SDM implementation throughout England.4 Their ambition is to make SDM the standard of care through clinical education, by making effective patient-decision aids readily available, and by raising patients’ expectations of having an active role in determining the most appropriate care for them. NICE is committed to developing decision aids based on clinical guidelines, and NHS England is committed to embedding SDM in its strategic and practical developments.5
SDM in primary prevention implantable cardioverter-defibrillator (ICD) implant
Randomised-controlled trials have demonstrated that implantable cardioverter-defibrillators (ICDs) reduce mortality in appropriately selected patients with severe left ventricular (LV) systolic dysfunction without a history of aborted sudden cardiac death (SCD).6 Offering primary prevention ICD therapy is recommended in national and societal guidance7 to an increasing patient population as indications expand.8 However, ICDs are associated with risks, including short-term procedural complications, reduced quality of life and increased hospitalisation rates.9 Unsurprisingly, patient preferences around ICD therapy vary.10 Patients who are older, have multiple comorbidities, or live with a higher symptom burden, may see a trade-off between SCD versus quality of life. Furthermore, there are issues in introducing the possibility of deactivation at a later date.11
Healthcare policy makers are keen to enhance the engagement of patients in SDM in the belief that it improves the benefits accrued from healthcare interventions. This may be important for interventions, such as ICD implantation, where cost-effectiveness is under scrutiny. The US Medicaid services have acknowledged this,12 and their reimbursement framework now mandates that all patients under consideration for primary prevention ICD implantation must have a SDM encounter using an evidence-based tool.13
NHS England offers financial incentives (Commissioning Quality Incentive or CQUIN schemes) for quality improvement to hospital providers of specialised services. In 2017, the NHS England team commissioning specialised services in the North East of England invited providers of ICD device implantation services to participate in a CQUIN scheme to improve decision-making around primary prevention ICD implantation.
Regional collaborative project to improve SDM
A collaborative strategy was developed by the three ICD implanting centres in the North of England (Northumbria Healthcare NHS Foundation Trust [NHCT], Newcastle upon Tyne Hospitals NHS Foundation Trust [NUTH], and South Tees Hospitals NHS Foundation Trust [ST]). The project included the development of a specific SDM tool, the first of its kind in the UK, followed by training and education of the clinical teams.
Critical to the success of implementing a regional CQUIN indicator is coordination of the initiative. In the case of the SDM CQUIN, this was led by the Clinical Governance and Risk Department at NUTH. The Quality Development Manager devised a project action plan, which enabled the three trusts to work to a common objective/outcome, acknowledging that their specific action plans would need to be adapted locally to allow for local variation in patient groups, facilities and procedures. The clinical governance teams worked cooperatively to organise and share the CQUIN submissions and data collection for training, staff surveys and patient experience data, although each trust was required to submit its own individual CQUIN submissions.
As a group, the clinical project leads met with the Quality Development Manager from NUTH on four occasions. The two initial meetings were also attended by Richard Thomson, Professor of Epidemiology and Public Health for his SDM expertise (with subsequent email input) and one of the local specialist commissioners. At this meeting the following were discussed:
- Selection of an appropriate SDM tool
- Local adaptation and development of the tool
- Training – who, when and how, including train-the-trainer sessions
- Evaluation of implementation
- Enhanced information delivery – what to offer, when and how to make sure patients receive it
- Action Plan update
- Previously agreed items dovetailed into NUTH initial CQUIN action plan
- How does the action plan look now? What is the impact of the amendments?
A further three meetings were undertaken. These were designed to discuss progress with action plans, share results and outcomes, and share experience of implementation.
Training and education
The project leads from each implanting site all underwent specific SDM micro-skills training, as developed through the UK MAGIC (Making Good decisions In Collaboration) implementation programme.14 The larger trusts (NUTH and ST) adopted a ‘train-the-trainers’ model with team members disseminating the teaching to the wider clinical team, including medical and surgical staff, cardiac physiologists and specialist nurses. In NHCT, all individuals making ICD implant decisions attended a local training course.
Development of a decision aid
Patient-decision aids are tools designed to help patients participate in decision-making about their healthcare choices. They provide information on the available options and help patients clarify and communicate the personal value they associate with different features of the options. Patient-decision aids do not advise people to choose one option over another, nor are they meant to replace practitioner consultation. Instead, they prepare patients to make informed, values-based decisions with their practitioner. A Cochrane review of 105 trials of decision aids identified that compared with usual care across a wide variety of decision contexts, people exposed to decision aids felt more knowledgeable, better informed, and clearer about their values, and had a more active role in decision-making with more accurate risk perceptions.13 There is growing evidence that decision aids may improve value-congruent choices. There are no observed adverse effects on health outcomes or satisfaction.
The International Patient Decision Aid Standards collaboration (IPDAS)15 is an organisation that has established a set of quality standards for patient-decision aids. They define the core components of SDM tools – to provide information about options, present probabilities (in an unbiased and, importantly, in an understandable format), provide methods for clarifying values and structured guidance for deliberation and communication.
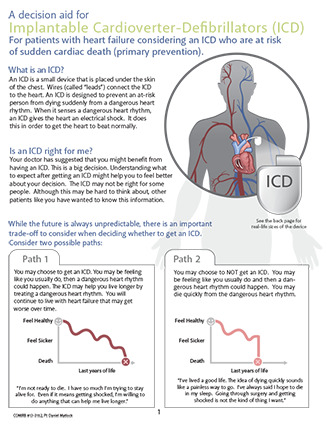
As a team, we developed a regional SDM tool that was consistent with this framework.16 We collaborated with Dr Dan Matlock, University of Denver, US who developed one of the ICD decision-making aids recommended for use in the US for insurance coverage and payment for any patient receiving an ICD. Our regional tool is an adaptation of this ‘ICD infographic’,17 which we modified and tailored to patients in the UK (click on image to view this supplementary material).
During the development of the tool we carefully considered which data to present, since each decision represents a judgement, which is open to challenge. It is known that the benefits and risks of ICD implantation differ between ischaemic and non-ischaemic aetiologies, and also by gender and age. They also change over time as new therapies are adopted and new risk stratification is developed. Every SDM tool is a compromise and is designed to provide a framework to facilitate management discussions with patients rather than provide a rigid algorithm. We also recognise that this tool may not cover all scenarios for ICD implantation, such as secondary prevention and aetiology other than heart failure.
Evaluation, outcomes and limitations
As part of the project we were asked by the specialised commissioners to gather information to evaluate the impact of this project. We gathered survey data from the cardiology staff and from patients undergoing discussions regarding ICD implantation before and after the project. The questionnaires used were standardised across the three sites and were adapted from those used in the MAGIC programme drawing upon validated questionnaires in SDM.14
Staff members, including cardiology consultants, speciality trainees, arrhythmia nurses and cardiac physiologists, were sent an online survey both before and after the project. We received 48 responses across the three trusts prior to the initiation of this project. This included 22 cardiology consultants, 10 speciality trainees and 16 allied professionals. Our re-evaluation after the project received 50 responses (23 consultants, 10 speciality trainees and 17 allied professionals). At baseline, 90% (43/48) of healthcare professionals stated that involving patients in decisions about their care was definitely important, which increased to 96% (48/50) after project completion (the remainder felt it was probably important at both points surveyed). However, only 88% (42/48 and 44/50) felt they routinely involved patients in making decisions about their treatment and care both before and after the project. Prior to the project, 69% (33/48) felt that providing patients with decision aids was definitely a good idea, increasing to 86% after the project (43/50) with the remainder stating it was “probably a good idea” at both time points. At baseline, 35% (17/48) of healthcare professionals felt they had all the skills and 65% (31/48) felt they had some of the skills they needed to involve patients in decisions about their treatment and care. The balance of this changed with rates of 46% (23/50) and 54% (27/50), respectively, after the project, reflecting an increased number of individuals feeling adequately skilled. Professionals reporting routine use of a SDM tool increased from 6% (29/48) prior, to 24% (12/50) after completion. Those sometimes using a SDM tool increased from 42% (20/48) to 44% (22/50) and those never using a SDM tool fell from 46% (22/48) to 28% (14/50). At baseline, 33% (16/48) reported having had previous information or training in SDM, which increased to 64% (32/50) after the project.
The surveys were emailed to a wide distribution list across multiple sites and, as such, a response rate was not available, and there may have been a degree of responder bias (although we would expect that to have been similar at both time points). There was also some change in the personnel within each unit during the project and we are, therefore, not evaluating the same group before and after our intervention, which may have influenced the data. However, the results do suggest a cultural shift. The data suggest a wider appreciation and understanding of the principles of SDM, and it is now discussed and applied more frequently, which is a significant achievement of the project.
Patients were asked about their experience in the consultation with respect to the decision-making process. The feedback was very good in the pre-project results, and patients generally rated their consultation highly, including awareness and involvement in the process. This may reflect well-recognised problems with ceiling effects in patient-completed SDM questionnaires.18 The nature of the project prevented within-patient comparisons and the second round of patient surveys at the end of the project did not show any marked change in the scores. The response rate was low at all centres and the sample population varied between sites. In addition, patients may not have been aware of their communication preferences and may not have had enough experience of consultations to offer a meaningful assessment. Ideally, a patient would have two consultations, with and without a SDM approach, and evaluate which they preferred, but this is challenging to do, has its own limitations and is beyond the scope of this project.
The project also has an inherent selection bias, which may limit its applicability. The three cardiologists who led the project were self-selected because of their interest in SDM and complex device therapy, so may not be representative of clinicians across secondary and tertiary care. Furthermore, we also only looked at the final step of a complex clinical heart failure pathway in terms of device therapy.
In summary
There has been a cultural change within the local cardiology teams in their attitudes to SDM alongside this project. Additional benefits from this include the widespread discussion of SDM across the region in many forums, including device multi-disciplinary team (MDTs) meetings, valve MDTs, and revascularisation MDTs. It cannot be overstated how significant this shift in attitudes and behaviours has been. The project has effectively introduced the concept of SDM to a wide audience and local SDM leads have been identified. In delivering this CQUIN, we have trained large numbers of staff in the principles and micro-skills of SDM, applicable across clinical practice, not just in ICD implantation.
The project looks at the role of SDM in a device pathway. However, the only point that has been addressed is the final point of the pathway, i.e. when a patient visits a specialist to discuss ICD therapy. Often patients will have had numerous discussions with healthcare professionals during this journey and it is not possible to capture how many, if any, of those consultations have followed a SDM model. As such, patients frequently attend the implanting centre with preconceived ideas and expectations about the care they are going to be offered. It can, thus, prove challenging to adequately explore all options. If the project were to be repeated in other areas, such as revascularisation or atrial fibrillation, as has been suggested by some, we would recommend that SDM be implemented at all stages from primary care to secondary care through to tertiary care.
This CQUIN project has taken a significant amount of time to complete, and that should not be underestimated in planning similar projects. The formal meetings alone required seven hours of senior clinician and management time plus up to eight hours travel each to attend these. The time taken for training the senior individuals within the project is in excess of 46 person-hours. In addition, the local trainers then delivered up to three local four-hour sessions to the remaining clinical body. The complexity of the CQUIN data returns was challenging and time-consuming for the clinical leads, and required continuous and firm support from Clinical Governance and administrative teams in relation to data collection, analysis and submission to commissioner teams. This varied between trusts, and it is important to recognise the significance of this process if this type of quality improvement project is going to be widely successful. The work was undertaken by clinicians with an interest in communication, in addition to their clinical work, and rolling this out will require local device lead clinicians to champion local adoption.
This project was only deliverable because of the links forged in the strong regional cardiac rhythm management (CRM) group of the strategic clinical network. This network has continued despite funding challenges and has clearly demonstrated its value in improving direct patient care. Our history of collaboration across trusts within the network, and the good relationships between clinicians, underpinned the success of the project. Given the NHS aims of increased integration between healthcare providers, we hope that there may be renewed recognition of the importance of supporting regional cardiac network groups to drive quality improvement across a region.
What next?
The work on this CQUIN started in 2017. This has been a standing agenda item on our regional CRM network meetings of all clinical leads, which has raised awareness among all trusts within the network. An educational session covering SDM has been included in the regional cardiology clinical advisory group education day on behalf of the North East Cardiovascular Network. The meeting was attended by approximately 100 delegates from a wide range of cardiac service providers. In addition, our work has been recognised nationally and we have been approached to share our learning with other regions and specialities, and the NICE SDM collaborative. We are keen to share our experiences and the tool we created to enable other centres to follow this model of care.
Key messages
- NHS England and the National Institute for Health and Care Excellence (NICE) are committed to increasing implementation of shared decision-making in patient care
- Implantable cardioverter-defibrillator (ICD) implanting centres in the North East of England collaborated to deliver a joint project to improve shared decision-making in this setting
- A specific shared decision-making tool has been developed and widely used, in addition to specific training of all staff
Conflicts of interest
None declared.
Acknowledgements
Fiona Suttie, Craig Dobson, and Dr Dan Matlock.
Funding
None.
Study approval
No ethical approval required.
References
1. NHS Patient Survey Programme. 2015 adult inpatient survey. Statistical release. Newcastle upon Tyne: Care Quality Commission, 2016. Available from: http://www.cqc.org.uk/sites/default/files/20150822_ip15_statistical_release_corrected.pdf
2. Care Quality Commission. Better care in my hands: a review of how people are involved in their care. Newcastle upon Tyne: Care Quality Commission, 2017. Available from: https://www.cqc.org.uk/publications/themed-work/better-care-my-hands-review-how-people-are-involved-their-care
3. National Institute for Health and Care Excellence (NICE)/NHS England. Shared decision making collaborative: a consensus statement. London: NICE, 2016. Available from: https://www.nice.org.uk/Media/Default/About/what-we-do/SDM-consensus-statement.pdf
4. National Institute for Health and Care Excellence. Shared decision making. Available at: https://www.nice.org.uk/about/what-we-do/our-programmes/nice-guidance/nice-guidelines/shared-decision-making
5. National Institute for Health and Care Excellence. Shared decision making collaborative – an action plan. London: NICE, 2016. Available from: https://www.nice.org.uk/Media/Default/About/what-we-do/shared-decision-making-collaborative-action-plan.pdf
6. Bardy GH, Lee KL, Mark DB et al.; Sudden Cardiac Death in Heart Failure Trial (SCD-HeFT) Investigators. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure. N Engl J Med 2005;352:225–37. https://doi.org/10.1056/NEJMoa043399
7. Hunt SA, Abraham WT, Chin MH et al. American College of Cardiology, American Heart Association Task Force on Practice Guidelines, American College of Chest Physicians, International Society for Heart and Lung Transplantation, Heart Rhythm Society: ACC/AHA 2005 guideline update for the diagnosis and management of chronic heart failure in the adult. Circulation 2005;112:e154–e235. https://doi.org/10.1161/CIRCULATIONAHA.105.167586
8. Goldberger Z, Lampert R. Implantable cardioverter-defibrillators: expanding indications and technologies. JAMA 2006;295:809–18. https://doi.org/10.1001/jama.295.7.809
9. Goldenberg I, Moss AJ, Hall WJ et al.; Multicenter Automatic Defibrillator Implantation Trial (MADIT) II Investigators. Causes and consequences of heart failure after prophylactic implantation of a defibrillator in the multicenter automatic defibrillator implantation trial II. Circulation 2006;113:2810–17. https://doi.org/10.1161/CIRCULATIONAHA.105.577262
10. Noyes K, Corona E, Zwanziger J et al.; Multicenter Automatic Defibrillator Implantation Trial II. Health-related quality of life consequences of implantable cardioverter defibrillators: results from MADIT II. Med Care 2007;45:377–85. https://doi.org/10.1097/01.mlr.0000257142.12600.c1
11. Standing H, Exley C, Flynn D et al. A qualitative study of decision-making about the implantation of cardioverter defibrillators and deactivation during end-of-life care. Health Serv Deliv Res 2016;4(32). https://doi.org/10.3310/hsdr04320
12. Centers for Medicare and Medicaid Services. Decision Memo for Implantable Cardioverter Defibrillators (CAG-00157R4). Baltimore, MD: CMS, 2018. Available from: https://www.cms.gov/medicare-coverage-database/details/nca-decision-memo.aspx?NCAId=288
13. Stacey D, Légaré F, Lewis K et al. Decision aids for people facing health treatment or screening decisions. Cochrane Database Syst Rev 2017;4(4):CD001431. https://doi.org/10.1002/14651858.CD001431.pub5
14. Joseph-Williams N, Lloyd A, Edwards E et al. Implementing shared decision making in the NHS: lessons from the MAGIC programme. BMJ 2017;357:j1744. https://doi.org/10.1136/bmj.j1744
15. International Patient Decision Aid Standards (IPDAS). Homepage. Available at: http://ipdas.ohri.ca/
16. Matlock DD, Spatz ES. Design and testing of tools for shared decision making. Circ Cardiovasc Qual Outcomes 2014;7:487–92. https://doi.org/10.1161/CIRCOUTCOMES.113.000289
17. Matlock DD, Vigil D, Jenkins A et al. A decision aid for implantable cardioverter-defibrillators (ICD) for patients with heart failure considering an ICD who are at risk for sudden cardiac death (primary prevention). University of Colorado, 2014. Available from: https://patientdecisionaid.org/wp-content/uploads/2017/01/ICD-Infographic-5.23.16.pdf
18. Scholl I, Koelewijn-van Loon M, Sepucha K et al. Measurement of shared decision making – a review of instruments. Z Evid Fortbild Qual Gesundhwes 2011;105:313–24. https://doi.org/10.1016/j.zefq.2011.04.012