Control of hypercholesterolaemia is an effective way of reducing cardiovascular events in patients at elevated risk of cardiovascular disease. Repeat cholesterol measurement is a good practice, but is rarely performed, with clinicians often adopting a ‘fire and forget’ policy.
A retrospective audit at two cardiac centres in the West Midlands was performed to evaluate performance in lipid measurement following initiation of lipid therapy, and to evaluate whether all patients with an indication for a proprotein convertase subtisilin/kexin 9 inhibitor (PCSK9i) following acute coronary syndrome (ACS) were identified according to National Institute for Health and Care Excellence (NICE) recommendations.
We identified 167/7,048 patients with a total cholesterol of ≥7.2 mmol/L, of which 33 patients had previous vascular disease affecting more than one territory (polyvascular disease). Total cholesterol threshold ≥7.2 mmol/L was chosen in order to achieve a sufficient number of samples for the audit to be completed successfully. Low-density lipoprotein (LDL) level was performed in 93 patients on admission and repeated within three months in 20 patients (13%). Overall, 48 patients (30%) had any repeat LDL recorded. Of this group, 10 patients met the NICE criteria for PCSK9i.
Failure to repeat LDL levels in patients following hospital discharge with a diagnosis of ACS, is likely to explain the small number of patients identified as being eligible for PCSK9i. Underreporting of comorbid vascular disease also prevents the identification of patients at very high risk. We advocate re-establishing the practice of routine repeat fasting lipids measurement following discharge from hospital in order to correctly identify patients who may be eligible for optimal lipid-lowering therapy.
Introduction
Control of hypercholesterolaemia has been established as a highly effective first-line management in acute coronary syndrome (ACS) patients.1,2 Data from a meta-analysis of randomised-controlled trials demonstrated that for each 1 mmol/L reduction in low-density lipoprotein (LDL)-cholesterol (LDL-C), statins reduced major cardiovascular (CV) events (myocardial infarction [MI], CV death, any stroke or coronary revascularisation) by an estimated 22%.3 National Institute for Health and Care Excellence (NICE) and international guidelines recommend fasting lipid profiles before, and within three months following, the initiation of high-intensity statin therapy as good practice in monitoring the response to lipid-lowering therapy.4,5
PCSK9 (proprotein convertase subtilisin/kexin 9) inhibitor (PCSK9i) is a monoclonal antibody targeting PCSK9 protein, which is involved in the control of the LDL-receptor. Randomised-controlled trials have demonstrated significant reductions in LDL-C of at least 50%, which has translated into major reductions in the composite primary outcomes of CV death, MI, stroke, hospitalisation for unstable angina, or coronary revascularisation.6,7
Table 1. National Institute for Health and Care Excellence (NICE) low-density lipoprotein cholesterol (LDL-C) threshold value for primary non-familial hypercholesterolaemia or mixed dyslipidaemia with cardiovascular disease (CVD), above which proprotein convertase subtisilin/kexin 9 (PCSK9) inhibitors are recommended
| CVD risk | LDL-C level |
|---|---|
| High risk (any history of ACS, CAD, PVD, ischaemic stroke, arterial revascularisation procedure) | >4.0 mmol/L |
| Very high risk (polyvascular disease/recurrent cardiovascular events) | >3.5 mmol/L |
| Key: ACS = acute coronary syndrome; CAD = coronary artery disease; PVD = peripheral vascular disease | |
NICE technology appraisals (TA 393 and 394) recommend PCSK9i therapy (alirocumab or evolocumab) as secondary prevention for ACS patients with persistently elevated LDL-C level following high-intensity statin (table 1).8,9 PCSK9i is not recommended as primary prevention in primary non-familial hypercholesterolaemia or mixed dyslipidaemia.
NICE also acknowledge that a different threshold is needed in primary heterozygous familial hypercholesterolaemia. In this population, PCSK9i is recommended only if LDL-C level is persistently above 5.0 mmol/L as primary prevention, or above 3.5 mmol/L as secondary prevention. There is no further differentiation between high-risk or very-high-risk groups in primary heterozygous familial hypercholesterolaemia with CV disease.
We assessed compliance to NICE guidelines for the management of hyperlipidaemia following ACS at two cardiac centres; Worcester Acute Hospital NHS Trust and Royal Stoke University Hospital.
Method
We performed a retrospective audit at Worcester Acute Hospital NHS Trust and Royal Stoke University Hospital. Our sample population was ACS patients recorded in the local Myocardial Ischaemia National Audit Project (MINAP) registries, between 1 January 2010 and 31 December 2017 at Worcester, and between 1 January 2010 and 31 December 2014 at Stoke, with elevated total cholesterol levels (total cholesterol ≥7.2 mmol/L).
The standards against which we audited were:
- NICE clinical guideline 181 recommendation
- In ACS patients, lipid sample on admission and about three months after initiation of high-dose statin treatment.
- NICE TA 393 and 394 recommendation
- In patients with primary non-familial hypercholesterolaemia or mixed dyslipidaemias with CV disease at high and very high risk of CV disease, treatment with PCSK9i is recommended in the context of persistently elevated LDL-C level (>4.0 mmol/L or >3.5 mmol/L, if high or very high risk, respectively).
Biochemical databases and medical notes were reviewed to collect baseline and follow-up data, and data on lipid profile LDL-C at initiation and at follow-up.
Results
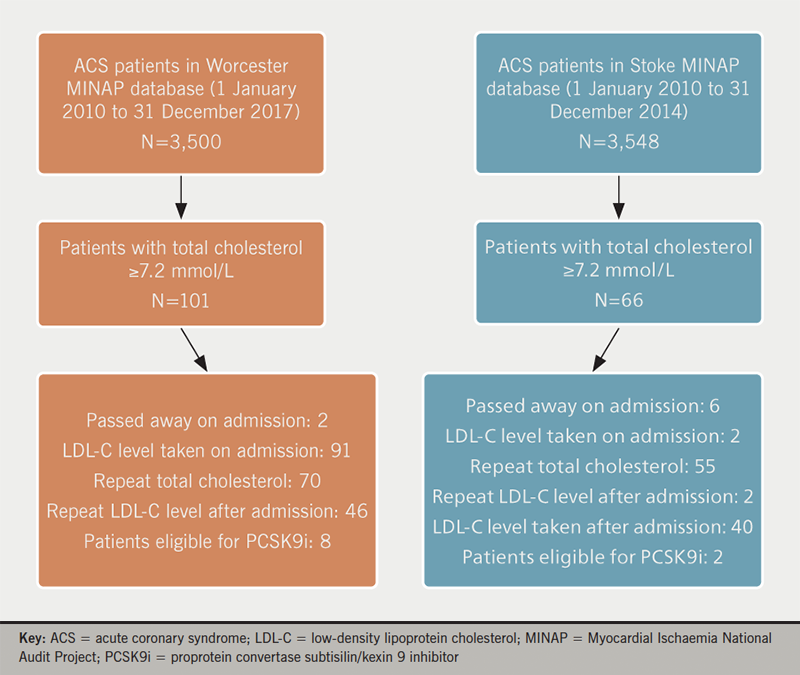
Table 2. Patient demographics, distribution of ‘high’ and ‘very high’ risk group, and lipid-lowering therapy on discharge
| Patient demographics | |
|---|---|
| Patients with total cholesterol ≥7.2 mmol/L, n | 167 |
| Male, n (%) | 95 (57%) |
| Mean age, years | 62 |
| Died on admission, n | 8 |
| Mean total cholesterol level | 7.8 mmol/L |
| Mean LDL-C level | 4.88 mmol/L |
| Distribution of ‘high’ risk and ‘very high’ risk group (NICE TA 393 and 394 definition) | |
| N (%) | |
| ‘High’ risk CVD group | 131 (82%) |
| ‘Very high’ risk CVD group | 28 (18%) |
| MI | 16 (10%) |
| MI + CVA | 2 (1%) |
| CVA | 7 (5%) |
| PAD | 3 (2%) |
| Patients died on admission | 8 |
| Lipid-lowering therapy on discharge | |
| N (%) | |
| No therapy | 10 (6%) |
| Statin monotherapy | 139 (87%) |
| Atorvastatin 80 mg | 119 |
| Atorvastatin 40 mg | 6 |
| Atorvastatin 20 mg | 3 |
| Atorvastatin 10 mg | 2 |
| Pravastatin 20 mg | 1 |
| Simvastatin 40 mg | 8 |
| Ezetimibe monotherapy | 3 (2%) |
| Statin and ezetimibe therapy | 5 (3%) |
| Atorvastatin 80 mg + ezetimibe | 4 |
| Rosuvastatin 20 mg + ezetimibe | 1 |
| Rosuvastatin 40 mg + fibrate | 1 (1%) |
| Ezetimibe + fibrate | 1 (1%) |
| Key: CVA = cerebrovascular accident; CVD = cardiovascular disease; LDL-C = low-density lipoprotein cholesterol; MI = myocardial infarction; NICE TA = National Institute for Health and Care Excellence Technology Appraisal; PAD = peripheral artery disease | |
In total, 7,048 ACS patients were identified on the MINAP database for the audit period. Of these, 167 patients had a total cholesterol ≥7.2 mmol/L (2.4%); 101 from Worcester and 66 from Stoke. Figure 1 shows the flow diagram of the audit and table 2 summarises the patient demographics, distribution of ‘high’ risk and ‘very high’ risk CV disease category, as well as distribution of lipid-lowering therapy on discharge.
All patients recorded in our sample population were at ‘high risk’ CV disease category (NICE TA 393 and 394 definition), of whom 28 patients (18%) were ‘very high risk’, as they had documented polyvascular disease.
There were 139 (87%) patients on statin monotherapy, five patients on a combination of high-dose statin and ezetimibe, three patients on ezetimibe monotherapy, one patient each on statin + fibrate and ezetimibe + fibrate therapy. Overall, 10 patients were not on any lipid-lowering therapy.
All patients had a cholesterol level taken on admission. Total cholesterol level was repeated in 125 out of 159 patients (79%). LDL-C level was taken in 93 patients on admission (58%) and was repeated within three months post-discharge in 20 patients (13%). Overall, repeat LDL-C level was recorded in 88 patients (55%) during the study period. The mean time interval for repeat LDL-C measurement was 6.89 months.
Following commencement of lipid-lowering therapy, there was a 37% and 44% reduction in total cholesterol and LDL-C level, respectively. NICE recommends >40% reduction in total cholesterol as the indication of a successful response to lipid-lowering therapy (table 3).
In this study, we identified 10 patients who met the criteria set out by NICE TA 393 and TA 394 for PCSK9i. This represents 0.14% of total cohorts.
Discussion
This retrospective audit has demonstrated reasonable compliance to NICE clinical guidance 181 recommendation with respect to lipid monitoring following an ACS event (79% compliance rate) and a small number of eligible patients who met the criteria for PCSK9i (TA 393 and 394 recommendations).
There are a number of potential explanations for the low compliance to NICE TA 393 and TA 394.
Table 3. Reduction in cholesterol and low-density lipoprotein (LDL) level following lipid-lowering therapy
| Pre-treatment, mmol/L | Post-treatment, mmol/L | Mean reduction | |
|---|---|---|---|
| Mean LDL | 4.88 ± 1.34 | 2.73 ± 1.37 | 44% |
| Mean cholesterol | 7.87 ± 0.68 | 4.92 ± 1.68 | 37.3% |
First, LDL-C is inconsistently measured during hospital admission (58% in this study) but is seldom measured as part of follow-up in the community. The most likely explanation is that LDL-C measurement requires a fasting blood sample, and following the recommendation by NICE clinical guidance 181, most general practitioners monitor follow-up lipid samples by measuring total cholesterol, non-high-density lipoprotein (HDL)-cholesterol and HDL cholesterol in a non-fasted sample and not LDL-C. This is important, as failure to measure LDL-C will prevent the identification of patients who are eligible for PCSK9i, which relies on LDL-C level for eligibility.
In this study, only 55% of patients had LDL-C repeated at anytime during follow-up and, therefore, the pool of patients who may be eligible for PCSK9i will be reduced by half. There were no significant differences in practice between the two sites in this study.
To mitigate against this, in Worcester, we have adopted a protocol whereby our cardiac rehabilitation team monitor lipid profile (including LDL-C) in patients who enrol in the programme, and the primary care physicians monitor those who declined the cardiac rehabilitation programme, in particular. These patients may have been followed-up locally by their primary physicians, whose biochemistry results, such as lipid profile, could be shared on local electronic biochemistry results. However, whether there is a clear pathway with respect to management and escalation remains unclear. Communication between cardiology team and primary care physicians would be crucial in identifying patients with suspected familial dyslipidaemia, with respect to closer monitoring and referral to a local lipid clinic or a familial hypercholesterolaemia service, such as West Midlands Familial Hypercholesterolaemia Service, for further assessment.
Second, we believe that there is underreporting of polyvascular disease in our sample population, and this is important as it leads to suboptimal identification of patients at ‘very high risk’, who could potentially benefit from PCSK9i therapy. We recorded 28 patients who had polyvascular disease, which represents 18% of the sample population. There were 10% of MI, 5% of cerebrovascular accident (CVA), 2% of peripheral artery disease (PAD), and 1% of MI + CVA. Bhatt et al. evaluated prior polyvascular disease as a risk factor for adverse ischaemic outcomes in ACS.10 In their sample population of 95,749 patients, the proportion of MI, CVA, and PAD was 43%, 10%, and 12%, respectively. One possible explanation is the MINAP registry is usually recorded by administrative staff with no medical experience. Therefore, clear documentation on discharge summaries to fill in the registry can help with identification of very-high-risk patients.
Third, part of better lipid management is to explore the reason for unsuccessful response to therapy. We should explore the possibility of statin intolerance or non-adherence to therapy. Table 3 shows that there were 10 patients discharged with no lipid-lowering therapy following ACS events. There is very limited documentation on why patients were discharged with no lipid-lowering therapy. There were three patients who reported insensitivity or allergy to statins. Out of these 10 patients, two of them are within the ‘very-high-risk’ group. Non-adherence could be due to fear of intolerance or adverse effects, lack of awareness or misperception, or poor follow-up.11 Without better control of hyperlipidaemia, these patients are up to 40% more likely to have a further cardiovascular event.12
Lastly, in this study, 75% of patients received high-dose statin, atorvastatin 80 mg on discharge, and a further 5% patients were on statin + ezetimibe, or ezetimibe alone. We observed a reduction in total cholesterol of 37% and a reduction in LDL-C of 44%. The NICE guideline recommends reduction in total cholesterol >40% as an indicator of successful therapy, while European Society of Cardiology (ESC) guidelines recommend reduction in LDL-C ≥50%, as a class IA recommendation, both in ‘high-risk’ and ‘very-high-risk’ groups. We acknowledge there is no mechanism in place at our centres to assess the changes made to statin therapy during the follow-up period. This highlights that we still need to adopt better lipid management strategy in these patients.
A meta-analysis was carried out to assess LDL-C, non-HDL-cholesterol, and apolipoprotein B levels among patients treated with statins.13 The study explored eight trials and evaluated the relative strength of the association of LDL-C, non-HDL-cholesterol, and apolipoprotein B with risk of cardiovascular events among patients treated with statins. The study observed, among patients on statin treatment, on-treatment levels of LDL-C, non-HDL-cholesterol and apolipoprotein B were each associated with risk of future major cardiovascular events. The study concluded the strength of this association was greater for non-HDL-cholesterol than LDL-C and apolipoprotein B, respectively. However, for the purpose of this audit, we follow the current recommendations from NICE to use LDL-C as the biomarker for considering PCSK9i. The ESC recommends LDL-C level as the primary lipid analysis for screening, diagnosis and management (class IC), whereas non-HDL-cholesterol is recommended for risk assessment, particularly in patients with high tryglycerides, diabetes, obesity or very low LDL-C level (class IC).
Conclusion
This study focused on the current practice of hyperlipidaemia management in ACS patients at two cardiac centres in the UK. We showed reasonable adherence to NICE recommendations with respect to repeat cholesterol measurement, but LDL-C level was measured infrequently. This resulted in a small number of patients identified as eligible for PCSK9 inhibitors. This trend seems to be shared with other centres.14
We emphasise the role of the multi-disciplinary team in ensuring better management of hyperlipidaemia following ACS, improvements in documentation with respect to polyvascular disease for better identification of patients in the ‘very-high-risk’ group, and education to general practitioners about the value of repeat LDL-C measurement in their patients on secondary prevention lipid therapy.
Key messages
- Control of hypercholesterolaemia is an effective way of reducing cardiovascular events in patients at elevated risk of cardiovascular disease. Repeat cholesterol measurement is good practice, but is rarely performed, with clinicians often adopting a ‘fire and forget’ policy to lipid management
- Following National Institute of Health and Care Excellence (NICE) approval of proprotein convertase subtisilin/kexin 9 inhibitor (PCSK9i), there is a need to identify patients who are eligible for this treatment
- Failure to repeat low-density lipoprotein (LDL)-cholesterol levels in patients following hospital discharge with a diagnosis of acute coronary syndrome (ACS), is likely to explain the small number of patients identified as being eligible for PCSK9i
- We advocate ending the ‘fire and forget’ policy of lipid-monitoring and re-establishing the practice of routine repeat fasting lipids measurement following discharge from hospital in order to correctly identify all patients who may be eligible for optimal lipid-lowering therapy
Conflicts of interest
None declared.
Funding
None.
Study approval
Ethical approval was not required to carry out this local clinical audit. Patient confidentiality was maintained throughout the undertaking of this audit.
References
1. Cannon CP, Braunwald E, McCabe CH et al. Intensive versus moderate lipid lowering with statins after acute coronary syndrome. N Engl J Med 2004;350:1495–504. https://doi.org/10.1056/NEJMoa040583
2. Heart Protection Study Collaborative Group. MRC/BHF Heart Protection Study of cholesterol lowering with simvastatin in 20 536 high-risk individuals: a randomised placebo-controlled trial. Lancet 2002;360:7–22. https://doi.org/10.1016/S0140-6736(02)09327-3
3. Cholesterol Treatment Trialists Collaboration; Baigent C, Blackwell L, Emberson J et al. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet 2010;376:1670–81. https://doi.org/10.1016/S0140-6736(10)61350-5
4. National Institute for Health and Care Excellence. Cardiovascular disease: risk assessment and reduction, including lipid modification. CG181. London: NICE, 2014. Available from: https://www.nice.org.uk/guidance/cg181
5. Mach F, Baigent C, Catapano AL et al.; ESC Scientific Document Group. 2019 ESC/EAS guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J 2020;41:111–88. https://doi.org/10.1093/eurheartj/ehz455
6. Sabatine MS, Giugliano RP, Keech AC et al.; FOURIER Steering Committee and Investigators. Evolocumab and clinical outcomes in patients with cardiovascular disease. N Engl J Med 2017;376:1713–22. https://doi.org/10.1056/NEJMoa1615664
7. Schwartz GG, Steg PG, Szarek M et al.; ODYSSEY OUTCOMES Committees and Investigators. Alirocumab and cardiovascular outcomes after acute coronary syndrome. N Engl J Med 2018;379:2097–107. https://doi.org/10.1056/NEJMoa1801174
8. National Institute for Health and Care Excellence. Alirocumab for treating primary hypercholesterolaemia and mixed dyslipidaemia. TA393. London: NICE, 2016. Available from: https://www.nice.org.uk/guidance/ta393
9. National Institute for Health and Care Excellence. Evolocumab for treating primary hypercholesterolaemia and mixed dyslipidaemia. TA394. London: NICE, 2016. Available from: https://www.nice.org.uk/guidance/ta394
10. Bhatt DL, Peterson ED, Harrison RA et al. Prior polyvascular disease: risk factor for adverse ischaemic outcomes in acute coronary syndromes. Eur Heart J 2009;30:1195–202. https://doi.org/10.1093/eurheartj/ehp099
11. Banach M, Stulc T, Dent R et al. Statin non-adherence and residual cardiovascular risk: there is need for substantial improvement. Int J Cardiol 2016;225:184–96. https://doi.org/10.1016/j.ijcard.2016.09.075
12. Serban MC, Colantonio LD, Manthripragada AD et al. Statin intolerance and risk of coronary heart events and all-cause mortality following myocardial infarction. J Am Coll Cardiol 2017;69:1386–95. https://doi.org/10.1016/j.jacc.2016.12.036
13. Boekholdt SM, Arsenault BJ, Mora S et al. Association of LDL cholesterol, non-HDL cholesterol, and apolipoprotein B levels with risk of cardiovascular events among patients treated with statins: a meta analysis. JAMA 2012;307:1302–09. https://doi.org/10.1001/jama.2012.366
14. Elamin AFM, Grafton-Clarke C, Chen KW et al. Potential use of PCSK9 inhibitors as a secondary preventative measure for cardiovascular disease following acute coronary syndrome: a UK real-world study. Postgrad Med J 2019;95:61–6. https://doi.org/10.1136/postgradmedj-2018-136171

