Papillary fibroelastomas (PFE) are rare benign cardiac tumours mainly originating on aortic and mitral valvular surfaces. Management is individualised, but most recommend surgical excision due to thromboembolic risk. We report a 75-year-old man with symptomatic severe aortic stenosis compounded by PFE. Redo sternotomy aortic valve replacement was deferred in favour of the trans-apical (TAVR) approach. This report highlights, for the first time, the application of TAVR as a strategy for aortic valve stenosis and PFE to mitigate risk posed by injury to patent internal mammary arterial graft in close proximity to the manubrium, and complications due to the patient’s multiple comorbidities.
Introduction
Papillary fibroelastoma (PFE) are rare intra-cardiac tumours and manifest with varied clinical presentation. This report highlights the application of transcatheter aortic valve replacement (TAVR) in patients with severe aortic stenosis, compounded by PFE, as a strategy to mitigate the high risk of redo surgery in patients with multiple comorbidities.
Case presentation
We report a 75-year-old man who presented with multiple syncopal episodes, who had undergone previous coronary artery bypass grafting (CABG). Both left internal mammary arterial (LIMA) graft to left anterior descending (LAD) artery and saphenous vein graft to diagonal territory were patent. There was a chronic occlusion of a vein graft to the right coronary artery (RCA).
Risk factors included hypertension, hyperlipidaemia, severe peripheral vascular disease, and remote smoking history. The patient had a heavily calcified ascending aorta and bilateral iliac artery stenting. Carotid duplex and blood tests were unremarkable.
On transthoracic echocardiography (TTE) a severely stenotic aortic valve (mean gradient 48 mmHg; Vmax 4.4 m/s; aortic valve area by continuity equation 0.7 cm2) with a likely PFE attached to the left coronary cusp seen. There was moderate left ventricular (LV) systolic dysfunction (LV ejection fraction 40%) and dilatation. Computed tomography (CT) and transoesophageal echocardiography confirmed a 7 × 4 × 6 mm non-calcified mass on the left coronary cusp (figures 1A and 1B).
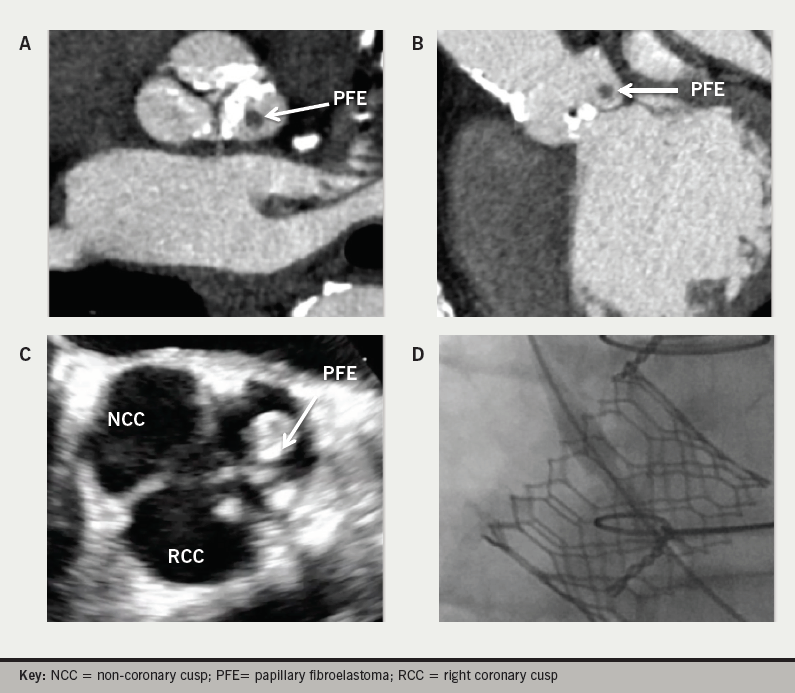
Multi-disciplinary discussion favoured a TAVR over redo sternotomy for open surgical aortic valve replacement (SAVR), due, principally, to the risk of a LIMA graft abutting the manubrium and calcifications of the ascending aorta. Trans-apical access was necessary as the bilaterally stented iliacs made the trans-femoral route unsuitable (≤3 mm) and circumferential calcification of the infra-renal abdominal aorta precluded trans-caval access. The concerns for TAVR were the unknown risk of PFE embolisation, PFE occlusion of a coronary ostium during valve deployment, and the long-term consequences or growth of a PFE ‘trapped’ in the aortic annulus/TAVR stent complex. By computer modelling we were confident that the PFE would not cause issue with coronary ostium patency. We were uncertain about risk of embolisation, as no literature on this combination of issues in TAVR was available, but felt this potential complication would be unlikely. With a tumour doubling time of 3.6 years, the slow growing natural history of PFE made it unlikely to impact on the life-expectancy of this patient.1
Intra-operative trans-oesophageal echocardiography (TOE) confirmed the diagnosis (figure 1C) and a Sapien 3 (29 mm) was implanted, without incident, trans-apically through an antero-lateral thoracotomy (figure 1D). TOE confirmed a well-seated valve, unchanged LV function and no aortic regurgitation or mobile mass. Complete atrio-venticular block developed in the first instance protected by epicardial pacing wires apically and a permanent pacing system was inserted on post-operative day one. Extubation in the operating room was without neurological sequelae and haemodynamics were stable on transfer to the intensive care unit.
The patient made an uneventful recovery and was discharged on the sixth post-operative day. TTE confirmed a well-seated Sapien 3 valve with no intra- or para-valvular regurgitation (LV ejection fraction 45%).
Discussion
This case report is the first to show that in the event conventional surgery risk is prohibitive then TAVR, in symptomatic severe aortic stenosis compounded by PFE, would be a potential option.
PFE are small, rare and benign cardiac tumours mainly originating on aortic and mitral valvular surfaces. Clinical presentation varies from asymptomatic, to flow obstruction, to severe thromboembolic events.
Surgical resection, on cardiopulmonary bypass, is indicated for symptomatic patients. A single-centre experience of resection of aortic valve PFE (n=21), by a shave technique, was safe with no observed 30-day mortality and no tumour recurrence (mean follow-up 17 ± 14 months).2
Consideration, by the ‘heart team’, of the risk posed by multiple comorbidities and potential injury to a patent LIMA favoured TAVR over SAVR. The information for decision-making was limited, as no evidence for PFE embolisation stroke risk during TAVR deployment existed. However, we were quite confident that the PFE would remain firmly attached to the aortic valve leaflet during Sapien 3 deployment and would not interfere with coronary ostium perfusion. We do recognise that the long-term consequences of PFE growth are unknown in this situation, but we felt it was a low risk for any future problems. Follow-up of the patient will be required to determine the fate of the tumour.
Conclusion
This report highlights, for the first time, the safe application of TAVR in severe aortic stenosis compounded by PFE as a strategy to mitigate the high risk of redo surgery in those with multiple comorbidities.
Conflicts of interest
None declared.
Funding
None.
Patient consent
Informed consent was obtained for the production of this manuscript and the images included. The authors confirm patient anonymity has been maintained throughout. Formal patient consent has been retained by the authors to ensure confidentiality.
References
1. Ayabe S, Hara K, Yamazaki I, Toda E, Tamura T. Slow growing cardiac tumor: a case of fibroelastoma. J Cardiol 2000;36:129–32.
2. Ikegami H, Andrei AC, Li Z, McCarthy PM, Malaisrie SC. Papillary fibroelastoma of the aortic valve: analysis of 21 cases, including a presentation with cardiac arrest. Tex Heart Inst J 2015;42:131–5. https://doi.org/10.14503/THIJ-14-4262
