Cardiac sarcoid remains a notoriously difficult to diagnose condition and arrhythmias remain an important initial presentation. It is amenable to treatment therefore it is important to make an early diagnosis to reduce morbidity and mortality.
Case report
A 51-year-old Asian woman presented with intermittent presyncope and profound breathlessness. She had no significant past medical history of note and was not receiving any regular medication. A resting 12-lead electrocardiogram (ECG) revealed a second-degree atrioventricular block. She subsequently underwent insertion of a dual-chamber permanent pacemaker. Further investigations at that time revealed unobstructed coronary arteries on angiography and normal ventricular function on transthoracic echocardiography.
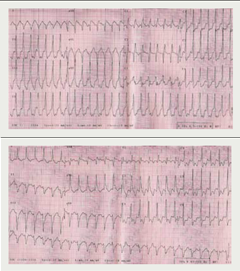
Her symptoms initially improved but relapsed one year later with breathlessness, presyncope but now intermittent rapid palpitation. A 24-hour Holter monitor revealed symptomatic rapid polymorphic ventricular tachycardia (figure 1). She consequently had her pacemaker upgraded to a biventricular internal defibrillator.
Despite biventricular pacing and adequate heart rate control with anti-arrhythmic medication, she continued to remain significantly breathless on minimal exertion prompting further investigation including pulmonary function tests, which revealed normal lung volumes and gas exchange.
A plain chest radiograph revealed diffuse interstitial infiltration. Upon thoracic high-resolution computed tomography there were typical abnormalities consistent with pulmonary sarcoid disease with enlarged paratracheal, subcarinal and aortic pulmonary lymphadenopathy
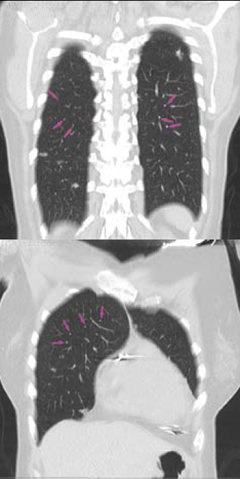
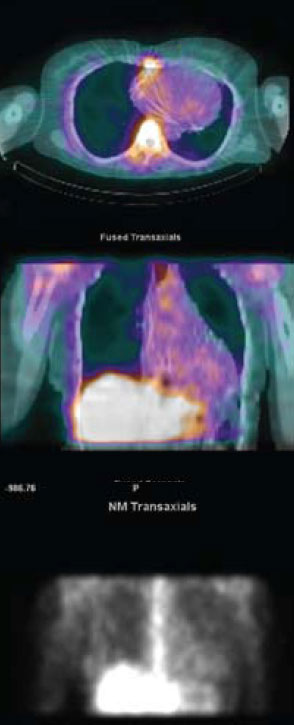
(figure 2). Cardiac involvement was confirmed on myocardial scintigraphy with gallium demonstrating abnormal myocardial tracer uptake
(figure 3). Steroid therapy was commenced with pulsed methylprednisolone alongside further anti-arrhythmic medication with a significant improvement in symptoms.
Discussion
All patients presenting with cardiac arrhythmias should be investigated in order to identify any underlying cardiac disease. Cardiac sarcoid may be the initial clinical manifestation of systemic sarcoidosis and a tachy- or bradyarrhythmia may be the first indication of its presence.
Cardiac sarcoid is more common than previously recognised, presenting not infrequently with sudden cardiac death. It is less likely to be diagnosed ante-mortem because less than 10% of patients have cardiovascular symptoms.1,2 It is, therefore, important to recognise overt and non-specific symptoms such as chest pain, breathlessness, palpitations and dizziness, and investigate appropriately.
Arrhythmias are an important clinical presentation in patients who have sarcoid disease with cardiac involvement, and virtually every type of arrhythmia has been documented.2,3 Fleming reviewed 138 fatalities out of 300 patients in whom a diagnosis of cardiac sarcoid had been made in life or at necropsy, and found that, at presentation, 73% had ventricular or supraventricular arrhythmias, 26% had complete heart block and 61% had left or right bundle branch blocks. Sudden death had occurred in 77 cases, and in 49 of these cardiac sarcoidosis had not been diagnosed previously.2 Uusimaa et al. described nine patients with cardiac sarcoid whose initial presentation was of ventricular tachyarrhythmia without any detectable systemic findings.4
It is important to note that not all patients who have documented arrhythmias may present with symptoms as Mikhail et al. revealed in a study of 147 consecutive patients diagnosed with sarcoidosis. In these patients, 9.5% presented with ECG abnormalities, however, 72% of these patients had no cardiac symptoms.1 While 41 of 80 patients published by Stein et al. with cardiac sarcoid had conduction and repolarisation abnormalities, none had clinical symptoms of cardiac disease.5
Clearly, the initial absence of systemic symptoms or signs of sarcoidosis posed a diagnostic conundrum in our patient. Furthermore, insertion of a pacemaker in the absence of a firm diagnosis effectively eliminated the use of cardiac magnetic resonance (CMR) at her next presentation.
CMR remains a useful diagnostic and monitoring tool in evaluating patients with cardiac sarcoid as demonstrated by Matsuki and Matsuo.6 The improvement or stability on follow-up CMR images post treatment has been correlated to clinical features and found useful as shown by Vignaux et al., but the significance may have to be tested in a larger trial.7
Gadolinium enhancement improves accuracy and localisation of granulomatous lesions and can be used as a guide to obtaining endomyocardial biopsy specimens. However, the proportion of patients with evidence of granulomatous cardiac lesions remains small. Hagemann and Wurm, in a review of 702 patients, reported that, while 5% had clinical evidence of cardiac sarcoidosis, only 15% had cardiac lesions at necropsy.8 Scadding and Mitchell reported a far worse percentage with four of the 500 patients reviewed having such evidence.9
Radionuclide tests with thallium and gallium have been widely used to demonstrate infiltration of myocardial tissue. The theory of reverse distribution of thallium on exercise or infusion of dipyridamole or adenosine has also been used to great effect in aiding diagnosis, and in the presence of normal coronary arteries this has a higher sensitivity. However, a normal scan does not exclude the presence of myocardial involvement nor does a positive scan add prognostic value.10
The goals of treatment for cardiac sarcoid are to reduce the extent of the disease in myocardial tissue, control of heart failure and establish rhythm management. Corticosteroids have played an important role in treatment of sarcoid disease, however, evidence for use remains anecdotal. In individual cases and small series, they have been shown to reverse mechanical and electrical abnormalities, however, prolonged use has also been reported to lead to increased risks of ventricular aneurysms.11,12 Immunosuppressants and cytotoxic agents have also been used as steroid-sparing agents.13
Anti-arrhythmic drugs reduce the risk of sudden cardiac death by abolishing fatal arrhythmias. This treatment alone has been insufficient in most reports because the majority of these arrhythmias are ventricular in origin, often incessant and drug refractory.1 Device therapy with pacemakers and automated cardiac defibrillators should thus be employed as there is also evidence that prognosis is improved.14 Catheter ablation procedures for refractory lethal arrhythmias may sometimes be necessary as an adjunct.15
In patients with heart failure, conventional pharmacotherapy with beta blockers, angiotensin-converting enzyme (ACE) inhibitors and diuretics remain the mainstay of treatment. Some patients may require heart transplantation where extensive myocardial involvement may be unresponsive to the aforementioned therapies. These patients often do well with a low recurrence of disease in the allograft.16
Overall, the diagnostic and treatment modalities of cardiac sarcoid in the last 10 years remain similar and no current literature has followed up patients who have been subjected to a standardised management strategy.18,19 The available data on prognosis are, thus, variable, with some reports quoting five-year survival rates ranging between 2 and 40%.2,12,17
The authors conclude that cardiac sarcoid remains notoriously difficult to diagnose and we recommend a high index of suspicion in order to achieve this. Prompt diagnosis, expert assessment and early aggressive treatment may improve prognosis and, as such, it is important to investigate extensively for structural heart disease in patients who present with unexplained arrhythmias.
Acknowledgements
We would like to thank Dr Kshama Wechalekar and Dr Michael Rubens for providing the radiology images. We would also like to acknowledge the contribution of Dr Paul Oldershaw in reading through this manuscript.
Conflict of interest
None declared.
References
- Mikhail JR, Mitchell DN, Bull KP. Abnormal electrocardiographic findings in sarcoidosis. In: Kiwai, Hosoda Y, eds. Proceedings of 6th international conference on sarcoidosis. Tokyo: University of Tokyo Press, 1974:365–72.
- Fleming HA. Death from sarcoid heart disease. United Kingdom series 1971–1986. 300 cases with 138 deaths. In: Grassi C, Rizzato G, Pozzi E, eds. Sarcoidosis and other granulomatous disorders. 11th world congress. Amsterdam: Elsevier, 1988:19.
- Uusimaa P, Ylitalo K, Anttonen O et al. Ventricular tachyarrhythmia as a primary presentation of sarcoidosis. Europace 2008;10:760–6.
- Suzuki T, Tsugiyasu K, Kubata S et al. Holter monitoring as a noninvasive indicator of cardiac involvement in sarcoidosis. Chest 1994;106:1021–4.
- Stein E, Jackler I, Stimmel B, Stein W, Siltzbach LE. Asymptomatic electrocardiographic alterations in sarcoidosis. Am Heart J 1973;86:474–7.
- Matsuki M, Matsuo M. MR findings of myocardial sarcoidosis. Clin Radiol 2000;55:323–5.
- Vignaux C, Dhote R, Duboc D et al. Clinical significance of myocardial magnetic resonance abnormalities in patients with sarcoidosis: a 1-year follow-up study. Chest 2002;122:1895–901.
- Hagemann GJ, Wurm K. The clinical, electrocardiographic and pathological features of cardiac sarcoidosis. In: Jones Williams W, Davies BH, eds. Sarcoidosis and other granulomatous diseases. 8th international conference. Cardiff: Alpha Omega, 1980:601.
- Scadding JG, Mitchell DN. The heart. In: Sarcoidosis. 2nd ed. London: Chapman and Hall, 1985:329–48.
- Okayama K, Kurata C, Tawarahara K. Diagnostic and prognostic value of myocardial scintigraphy with thallium 201 and gallium 67 in cardiac sarcoidosis. Chest 1995;107:330–4.
- Shammas RL, Movahed A. Successful treatment of myocardial sarcoidosis with steroids. Sarcoidosis 1994;11:37–9.
- Roberts WC, McAllister HA Jr, Ferrans VJ. Sarcoidosis of the heart: a clinicopathologic study of 35 necropsy patients (group 1) and review of 78 previously described necropsy patients (group 11). Am J Med 1977;63:86–108.
- Baughman RP, Ohmichi M, Lower EE. Combination therapy for sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis 2001;18:133–7.
- Winters SL, Cohen M, Greenberg S et al. Sustained ventricular tachycardia associated with sarcoidosis: assessment of the underlying cardiac anatomy and the prospective utility of programmed ventricular stimulation, drug therapy and an implantable antitachycardia device. J Am Coll Cardiol 1991;18:937–43.
- Koplan BA, Soejima K, Baughman K, Epstein LM, Stevenson WG. Refractory ventricular tachycardia secondary to cardiac sarcoid: electrophysiologic characteristics, mapping, and ablation. Heart Rhythm 2006;3:924–929 B.
- Valantine HA, Tazelaar HD, Macoviak J et al. Cardiac sarcoidosis: response to steroids and transplantation. J Heart Transplant 1987;6:244–50.
- Yazaki Y, Isobe M, Hiroe M et al. Prognostic determinants of long-term survival in Japanese patients with cardiac sarcoidosis treated with prednisone. Am J Cardiol 2001;88:1006–10.
- Baughman R. Therapeutic options for sarcoidosis: new and old. Curr Opin Pulm Med 2002;8:464–9.
- Mitchell DN, du Bois RM, Oldershaw PJ. Cardiac sarcoidosis—a potentially fatal condition that needs expert assessment. BMJ 1997;21:314–20.
