Structural heart disease may be missed using clinical examination alone and limited echocardiograms or ‘quick-scans’ may be a way to improve rates of detection. To evaluate the finding of clinically unexpected abnormalities using ‘quick-scans’, scans were performed in a general practice by a level 7 sonographer using a GE V scan system. Indications were: murmur; potentially cardiac symptom (e.g. chest pain or breathlessness); cardiac history in the GP notes (e.g. myocardial infarction); atrial fibrillation; chronic obstructive pulmonary disease with disproportionate dyspnoea; age ≥75 years. Standard transthoracic echocardiograms were performed if clinically indicated or if the ‘quick-scan’ detected a significant abnormality.
There were 163 ‘quick scans’ indicated, which were normal in 80 (49%), mildly abnormal in 67 (41%) and significantly abnormal in 16 (10%). Abnormalities were moderate left ventricular (LV) systolic dysfunction, moderate mitral regurgitation, moderate-to-severe aortic stenosis and mild aortic dilatation. Within the 90 patients without agreed indications the ‘quick-scans’ were normal in 64 (71%) and mildly abnormal in 26 (29%) while none were significantly abnormal.
In conclusion, ‘quick scans’ can detect clinically unexpected pathology. These results are consistent with a global move to use the hand-held ultrasound machine as an extension of the clinical examination.
Introduction
Heart failure and valve disease are common. The population prevalence of each is 2–3% and this rises to more than 10% aged ≥75 years.1,2 Echocardiography is a key diagnostic investigation but it is a relatively scarce resource and is also underutilised.3 It has been suggested that access can be improved by targeting high-risk individuals using limited ‘point-of-care’ studies also called ‘quick-scans’.4 There has been an increase in the application of ‘quick-scans’ with the development of hand-held machines, which allow near-patient testing in the community or on ward-rounds, in clinics or in emergency departments.5,6 However, experience with ‘quick-scans’ in the community using hand-held ultrasound devices is limited. We, therefore, set up a pilot clinical service.
Methods
Setting
The new service was offered by newsletter and direct email to all GP practices in Lambeth and Southwark, of which, two expressed interest. For the first, a regular time slot for ‘quick-scans’ was staffed every week, but only 13 patients were referred in seven weeks and these were judged insufficient for analysis. The second practice had a list of 12,565 patients, did not have booked visits and saw typically 100 patients daily. This practice referred 253 patients on two sessions a week over 26 weeks.
Quick scan
We used a V scan (GE Medical Systems), which has a display unit 135 × 73 × 28 mm in size and weighs 390 g. ‘Quick-scans’ were performed by a level 7 sonographer. The scan consisted of imaging and colour Doppler in the parasternal long-axis and short-axis views, apical four-chamber, two-chamber and three-chamber views and a subcostal view.
Scans were classed as normal if they were completely normal, or if there was no more than: mild aortic or mitral valve thickening; trivial aortic or mitral regurgitation; or mild-to-moderate tricuspid regurgitation. The following were classed as mildly abnormal: mild left ventricular (LV) hypertrophy (septal thickness 12–14 mm); borderline LV systolic dysfunction (LV ejection fraction 50–55%); mild aortic dilatation; mild valve restriction; mild aortic or mitral regurgitation; moderate tricuspid regurgitation. The following were classed as significantly abnormal: LV hypertrophy (septal thickness >14 mm); LV ejection fraction <50%; at least mild valve stenosis; moderate or worse aortic or mitral regurgitation; more than mild aortic dilatation; severe tricuspid regurgitation.
Indications for referral for ‘quick-scans’ were: age ≥75 years, cardiac symptoms (chest pain, palpitations, syncope); murmur; chronic obstructive pulmonary disease (COPD) with disproportionate breathlessness; cardiac history in the GP notes (e.g. myocardial infarction); abnormal electrocardiogram (ECG) including atrial fibrillation; high risk of rheumatic disease. Patients who were under cardiac care or had a recent echocardiogram were not scanned. Standard transthoracic echocardiograms were performed if clinically indicated or if the ‘quick-scan’ detected a significant abnormality.
The analysis was approved as a Trust service evaluation (no. 4,034).
Results
A total of 253 patients underwent a ‘quick-scan’. However, only 163 (64%) fitted the inclusion criteria. These were aged 72 ± 10 years (37% male and 63% female). A further 90 aged 65 ± 8 years (38% male and 62% female) were scanned at the GPs’ request despite not having agreed indications. Of these, 55 (61%) had systemic hypertension. These were analysed separately as a comparator group.
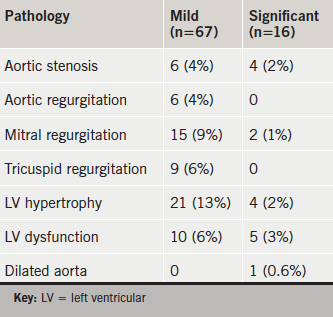
In the 163 patients with the agreed indications, the ‘quick-scan’ was normal in 80 (49%), mildly abnormal in 67 (41%) and significantly abnormal in 16 (10%) (table 1). In those aged ≥75 years, there were 10 (12%) with significant abnormalities. A subsequent standard transthoracic study confirmed moderate aortic stenosis in three, moderate-to-severe aortic stenosis in one, moderate mitral regurgitation in one, moderate-to-severe mitral regurgitation in one, moderate LV systolic dysfunction in four, moderate-to-severe dysfunction in one, LV hypertrophy in four and mild aortic dilatation in one.
Within the 90 patients without agreed indications the ‘quick-scans’ were normal in 64 (71%) and mildly abnormal in 26 (29%), while none were significantly abnormal. The mild abnormalities were: aortic regurgitation in three (3%); mitral regurgitation in 11 (12%); LV hypertrophy in eight (9%); borderline LV dysfunction in two (2%).
Discussion
Summary
This study showed that ‘quick-scans’ detected significant structural disease in 10% of patients with focused indications at a general practice and in 12% of those aged ≥75 years.
Limitations of current screening for heart failure and valve disease
The most common reasons for requesting echocardiography are suspected heart failure or valve disease.3 However, patients with heart failure may be misdiagnosed as having COPD,7 or may have both conditions,8 and may not be screened either with B-type natriuretic peptide levels or echocardiography. We, therefore, included patients with COPD but inappropriate breathlessness as an indication for a ‘quick-scan’. A murmur is an insensitive and nonspecific screen for valve disease and an audit of open-access echocardiography3 suggested that the yield of significant valve disease could be doubled by widening the indications to include an abnormal ECG or cardiac symptoms. In this study, we also included patients aged ≥75 years because the prevalence of valve disease is known to be >10% in this group,2 which we judged was a reasonable sensitivity to justify screening.
Clinical significance of the abnormalities detected
Screening has to be performed judiciously to avoid finding mild abnormalities that do not need treatment and merely raise unnecessary anxiety.9 For this reason, ‘quick-scans’ should not be performed without a reasonable clinical suspicion of structural disease. The ‘quick-scans’ were normal or near-normal in all 90 cases lacking agreed indications in this study. Most of the abnormalities we detected, even with agreed indications, were indeed mild but they were significant in 10%. It is best practice to follow patients with moderate valve disease in a specialist valve clinic,10 to allow correct timing of surgery, so these unsuspected diagnoses were clinically important.
Comparison with existing literature
‘Quick-scans’ have mostly been evaluated in the hospital setting including the intensive care unit or emergency room.5 They are reliable for predicting a normal standard echocardiogram.11,12 As, in our experience, they also detect clinically unexpected abnormalities, 17% in one series13 with no apparent indications for standard echocardiography. We are not aware of any service similar to ours, although portable studies have been shown to be useful at detecting LV hypertrophy in the community,14 and to be cost-effective in triaging the need for standard echocardiography in suspected heart failure.15 Our rate of abnormalities is similar to that in population screening studies,1,2 while there were no abnormalities in the cases without agreed indications suggesting that our method of focusing studies was effective.
Strengths and limitations
The population was necessarily general, and there were few patients fitting the indications for ‘quick-scans’, making it hard to fill a list. In fact a number of patients were referred outside the agreed indications, probably encouraged by the presence of the sonographer. This suggests that a separate list might be a better way to manage the service, although this approach did not work at the second GP practice participating. The logistics of the service, therefore, need to be agreed nationally,4 and are expected to vary according to local resources. It is vital that a service should be provided by suitably qualified and experienced staff, with operating systems including quality control that are as good as those in standard echocardiography services.12,16
Implications for practice
‘Quick-scans’ appear a useful method of screening a relatively high-risk community population, since a clinically useful yield of unexpected significant structural disease is obtained. These results are consistent with a global move to use the hand-held ultrasound machine as an extension of the clinical examination.
Key messages
- A ‘quick-scan’ detects significant abnormalities in 10% of people clinically judged as high risk
- Valve disease and left ventricular dysfunction were the most common abnormalities found
- ‘Quick-scans’ effectively extend the clinical examination and triage the need for standard transthoracic echocardiography.
Conflict of interest
None declared.
Funding
The salary for the level 7 sonographer and the cost of the V scan were supplied by Edwards Life Sciences who played no role in the design, conduct and analysis of the results of the service.
Editors’ note
See also the editorial by Currie and the case report by Workman et al. in this issue.
References
1. McMurray JJV, Adamopoulos S, Anker SD et al. ESC guidelines for the diagnosis and treatment of acute and chronic heart failure 2012. Eur J Heart Fail 2012;14:803–69. http://dx.doi.org/10.1093/eurjhf/hfs105
2. Nkomo VT, Gardin JM, Skelton TN et al. Burden of valvular heart diseases: a population-based study. Lancet 2006;368:1005–11. http://dx.doi.org/10.1016/S0140-6736(06)69208-8
3. Chambers J, Kabir S, Cajeat E. Detection of heart disease by open access echocardiography: a retrospective analysis of general practice referrals. Br J Gen Pract 2014;64:e105–e111. http://dx.doi.org/10.3399/bjgp14X677167
4. Arden C, Chambers J, Ray S et al. Can we improve the detection of heart valve disease? Heart 2014;100:271–3. http://dx.doi.org/10.1136/heartjnl-2013-304223
5. Hothi SS, Sprigings D, Chambers J. Point-of-care cardiac ultrasound in acute medicine – the quick scan. Clinical Med 2014;14:608–11. http://dx.doi.org/10.7861/clinmedicine.14-6-608
6. Panoulas VF, Daigeler AL, Malaweera AS et al. Pocket-size hand-held cardiac ultrasound as an adjunct to clinical examination in the hands of medical students and junior doctors. Eur Heart J Cardiovasc Imaging 2013;14:323–30. http://dx.doi.org/10.1093/ehjci/jes140
7. Miller MR, Levy ML. Chronic obstructive pulmonary disease: missed diagnosis versus misdiagnosis. BMJ 2015;351:h3021. http://dx.doi.org/10.1136/bmj.h3021
8. Rutten FH, Cramer M-JM, Grobbee DE et al. Unrecognized heart failure in elderly patients with stable chronic obstructive pulmonary disease. Eur Heart J 2005;26:1887–94. http://dx.doi.org/10.1093/eurheartj/ehi291
9. McDonald IG, Daly J, Jelinek VM, Panetta F, Gutman JM. Opening Pandora’s box: the unpredictability of reassurance by a normal test result. BMJ 1996;313:329–32. http://dx.doi.org/10.1136/bmj.313.7053.329
10. Lancellotti P, Rosenhek R, Pibarot P et al. Heart valve clinics: organisation, structure and experiences. Eur Heart J 2013;34:1597–606. http://dx.doi.org/10.1093/eurheartj/ehs443
11. Greaves K, Jeetley P, Hickman M et al. The use of hand-carried ultrasound in the hospital setting – a cost-effective analysis. J Am Soc Echocardiogr 2005;18:620–5. http://dx.doi.org/10.1016/j.echo.2004.09.015
12. Spencer K, Kimura B, Korcarz C et al. Focused cardiac ultrasound: recommendations from the American Society of Echocardiography. J Am Soc Echocardiogr 2013;26:567–81. http://dx.doi.org/10.1016/j.echo.2013.04.001
13. Fedson S, Neithardt G, Thomas P et al. Unsuspected clinically important findings detected with a small portable ultrasound device in patients admitted to a general medical service. J Am Soc Echocardiogr 2003;16:901–05. http://dx.doi.org/10.1016/S0894-7317(03)00426-7
14. Senior R, Galasko G, Hickman M, Jeetley P, Lahiri A. Community screening for left ventricular hypertrophy in patients with hypertension using hand-held echocardiography. J Am Soc Echocardiogr 2004;17:56–61. http://dx.doi.org/10.1016/j.echo.2003.09.013
15. Galasko GIW, Barnes SC, Collinson P, Lahiri A, Senior R. What is the most cost-effective strategy to screen for left ventricular systolic dysfunction: natriuretic peptides, the electrocardiogram, hand-held echocardiography, traditional echocardiography, or their combination? Eur Heart J 2006;27:193–200. http://dx.doi.org/10.1093/eurheartj/ehi559
16. Sicari R, Galderisi M, Voigt J-U et al. The use of pocket-size imaging devices: a position statement of the European Association of Echocardiography. Eur J Echocardiogr 2011;12:85–7. http://dx.doi.org/10.1093/ejechocard/jeq184

