Ischaemic conditioning is the phenomenon of protection against reperfusion injury via the application of brief, repeated episodes of non-lethal ischaemia. This review has three aims: 1) to briefly explain the various categories of ischaemic conditioning; 2) to explore past clinical trials and their failures; 3) to explore the future of clinical trials in the realm of ischaemic conditioning.
Introduction
Protecting the myocardium against lethal ischaemia necessitates rapid reperfusion. Paradoxically, however, reperfusion injures the myocardium, a phenomenon known as ‘ischaemia-reperfusion injury’ (IRI). There is currently no effective clinical intervention for IRI, in spite of the fact that it may contribute up to 50% of final myocardial infarct (MI) size.1 This review intends to describe past, present, and future clinical trials into ischaemic conditioning, which present one of the few avenues currently being explored clinically to kerb IRI.
The principles of ischaemic conditioning and the supporting proof-of-concept studies
Ischaemic conditioning is a form of protection against IRI produced via brief cycles of non-lethal ischaemia. These cycles may be subclassified as below (figure 1).
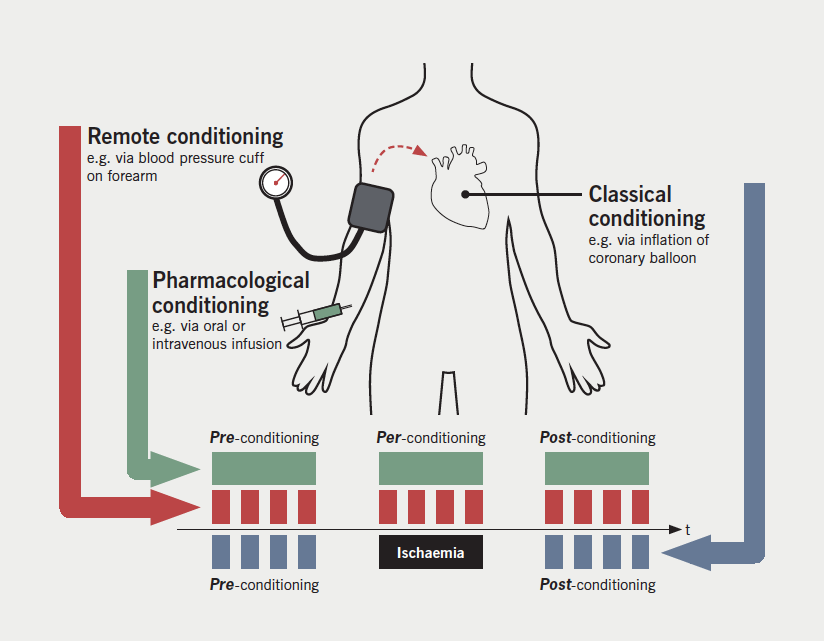
Pre-conditioning
- Classical ischaemic pre-conditioning (IPC): brief cycles of non-lethal ischaemia induced locally and directly to the myocardium prior to an episode of lethal ischaemia, e.g. aortic clamping. IPC has been shown to reduce post-surgical markers of cardiovascular damage (e.g. adenosine triphosphate [ATP],2 troponin T3) when performed during coronary artery bypass surgery (CABG). However, due to the inherent embolic risk of such intervention, it has not been carried over to large-scale clinical trials.
- Remote ischaemic pre-conditioning (RIPC): brief cycles of non-lethal ischaemia induced at a ‘remote’ limb via blood pressure cuff prior to an episode of lethal myocardial ischaemia. The need to apply the conditioning prior to an ischaemic stimulus largely limits its application to the surgical arena, where the time of onset of the injurious ischaemia can be readily predicted. Proof-of-concept studies have shown that RIPC has the capacity to reduce post-CABG troponin release in humans.4,5 Such an intervention is an attractive prospect due to its non-invasive nature. However, the recent outcome trials, RIPHeart (Remote Ischemic Preconditioning for Heart Surgery) and ERICCA (Effect of Remote Ischemic Preconditioning on Clinical Outcomes in Patients Undergoing Coronary Artery Bypass Surgery), have been neutral and will be explored later in this review.
Per-conditioning
- Pharmacological per-conditioning (PPerC): the use of pharmaceutical agents to mimic a conditioning stimulus during an ischaemic insult to protect against subsequent reperfusion injury. Proof-of-concept studies have shown ciclosporin-A (CsA)6 and metoprolol7 to be associated with a significant reduction in infarct size (measured by late-gadolinium enhancement on cardiac magnetic resonance imaging [MRI]). However, the large-scale CsA-based CIRCUS (Cyclosporine to ImpRove Clinical oUtcome in ST-elevation Myocardial Infarction Patients) and CYCLE (CYCLosporinE A in Reperfused Acute Myocardial Infarction) studies have also been neutral and are discussed in the next section (the failure of large-scale ischaemic conditioning trials).
- Remote ischaemic per-conditioning (RIPerC): brief cycles of non-lethal ischaemia induced at a limb via blood pressure cuff during an episode of lethal myocardial ischaemia. The proof-of-concept ERIC-lysis (Effect of Remote Ischemic Conditioning in Heart Attack Patients)8 study randomised ST-elevation myocardial infarction (STEMI) patients to receive thrombolysis with or without RIPerC. The RIPerC cohort had a significant reduction in MI size 24 hours post-intervention. This was shown by a median 32% reduction in troponin T (p=0.020), and 19% reduction in creatinine kinase (p=0.026) levels. The proof-of-concept CONDI (Effect of RIC on Clinical Outcomes in STEMI Patients Undergoing pPCI)9 trial randomised STEMI patients to receive primary percutaneous coronary intervention (PPCI) with or without in-ambulance RIPerC. The RIPerC cohort had a significant reduction in all-cause mortality (p=0.027) on follow-up (median 3.8 years). The large-scale CONDI 2/ERIC-PPCI studies are underway and will be discussed in the final section (the future of clinical trials in ischaemic conditioning).
Post-conditioning
- Pharmacological post-conditioning (PPost): the use of pharmaceutical agents to mimic a conditioning stimulus following onset of reperfusion to reduce subsequent reperfusion injury. The J-WIND (Human atrial natriuretic peptide and nicorandil as adjuncts to reperfusion treatment for acute myocardial infarction) study of 569 MI patients10 showed that those given an atrial natriuretic peptide (ANP) infusion post-reperfusion had a significantly reduced infarct size of 14.7% (p=0.019) and a reduced incidence of readmission for heart failure relative to the control group (median follow-up period 2.7 years) (hazard ratio [HR] 0.267; p=0.011). This same study showed that nicorandil had no effect on infarct size. In a study of 172 STEMI patients,11 exenatide (glucagon-like-peptide-1 analogue) infusion during reperfusion attenuated MI, resulting, on average, in a 15% greater salvage index (i.e. the difference between actual and potential infarct size). In spite of this, however, no significant change in clinical outcome between the two cohorts was found.
- Classical ischaemic post-conditioning (IPost): brief cycles of non-lethal ischaemia applied following angioplasty of the culprit lesion, typically achieved via transient balloon inflation within the culprit vessel. Proof-of-concept studies have produced mixed results12,13 and the large-scale DANAMI 3-iPost (DANish Study of Optimal Acute Treatment of Patients with ST-segment Elevation Myocardial Infarction) (data presented at the American College of Cardiology 2016) clinical trial showed no significant difference in clinical outcomes. This trial will be explored in the next section (the failure of large-scale ischaemic conditioning trials).
- Remote ischaemic post-conditioning (RIPost): brief cycles of non-lethal ischaemia induced at a limb via blood pressure cuff following reperfusion therapy. While proof-of-concept studies have shown some promise,14 no large-scale clinical trials have yet been undertaken to explore RIPost.
The failure of large-scale ischaemic conditioning trials
While proof-of-concept studies have provided a tantalising view of potential clinical efficacy with various ischaemic conditioning interventions, large-scale clinical trials have so far proved fruitless. This section will explore these failed trials and examine why ischaemic conditioning techniques have yet to fulfil their clinical potential.
RIPC
The 2015 RIPHeart study15 by Meybohm et al. was a 1,403 patient, multi-centre, double-blinded, randomised-controlled trial (RCT) exploring RIPC in elective surgical patients requiring cardiac bypass. The primary end point was a composite of acute renal failure, stroke, non-fatal MI or all-cause death up to 14 days after surgery. Four cycles of true or sham RIPC were given post-anaesthetic induction and pre-surgical incision. No significant difference was seen between the primary end points of either cohort. At approximately the same time, the ERRICA study16 by Hausenloy et al. was published. This study was a 1,612-patient, multi-centre, double-blinded RCT of RIPC in patients undergoing CABG surgery. The primary end point was the rate of major adverse cardiac and cerebral events 12 months after the intervention. Four cycles of true or sham RIPC were given post-anaesthetic induction and pre-surgical incision. Once again, no significant difference was seen between the primary end points of either cohort.
Why was it that both of these trials failed to provide positive results? It may be that the injury suffered during CABG surgery is too small for RIPC to provoke a significant protective effect: it is well recognised from the pre-clinical literature that the smaller the primary injury, the smaller the benefit from a conditioning intervention is likely to be. It is also important to consider the other therapies patients undergoing surgical procedures will be exposed to: all patients in RIPHeart and >90% of patients in ERICCA were anaesthetised with propofol, an agent which might abolish RIPC-based cardioprotection, and an important potential confounder.17
PPerC
The 2015 CIRCUS study18 by Cung et al. was a 970 patient, multi-centre, double-blinded RCT exploring CsA as a pre-conditioning agent in patients undergoing PPCI to treat STEMI. The primary end point was a one- and three-year composite of heart failure progression, rehospitalisation for heart failure, adverse left ventricular modelling, and all-cause death. Prior to PPCI, patients were randomised to a CsA or placebo infusion. No significant difference in the primary end point was seen at one year: a neutral outcome. This could be due to the absence of data for left ventricular (LV) end-diastolic volume in 17% of patients. These absent data, combined with the high incidence of adverse modelling in both cohorts, could have made it difficult to detect a significant difference in the other components of the primary end point composite. The 2016 CYCLE study19 by Ottani et al. was a 410 patient, multi-centre, prospective/randomised/open-label/blinded-endpoint (PROBE) trial, which also explored CsA as a per-conditioning agent in patients undergoing PPCI to treat STEMI. The primary end point was the incidence of ≥70% ST-segment resolution 60 minutes after thrombolysis in MI (TIMI) flow grade 3. Prior to PPCI, patients were randomised to a CsA or placebo injection. Once again, no significant difference between the cohorts was detected. As such, the future use of CsA as a per-conditioning agent appears doubtful.
IPost
DANAMI 3-iPost is a treatment subgroup of a 2016 trial programme comparing multiple treatment strategies for STEMI. The iPost arm of the trial was a 1,234 patient, multi-centre, PROBE trial comparing standard PCI to PCI plus IPost. The primary end point was a composite of hospitalisation for heart failure and all-cause death within two years. Patients were randomised to PCI, or PCI plus four 30-second balloon inflations within the reperfused coronary artery following initial reperfusion. No significant difference was detected between the two cohorts (data presented at the American College of Cardiology, 2016). The duration of ischaemia is critical: in the pre-clinical literature, there is a clear ‘therapeutic window’: ischaemic post-conditioning is ineffective where the index ischaemia duration is either very short or very long,20 and it would seem that, in clinical trials, the therapeutic window is typically in the order of one to four hours from symptom onset to revascularisation. Whether or not ischaemic time played a role in the neutral DANAMI 3-iPost study will require further analysis upon official publication.
The future of clinical trials in ischaemic conditioning
A number of large outcome trials exploring ischaemic conditioning are currently underway and may yet still provide evidence for the introduction of conditioning techniques into clinical practice.
RIPerC in PPCI
The CONDI 2 (Effect of RIC on Clinical Outcomes in STEMI Patients Undergoing pPCI) and ERIC-PPCI (Effect of Remote Ischaemic Conditioning on Clinical Outcomes in STEMI Patients Undergoing PPCI) are a pair of collaborative studies investigating the use of RIPerC in STEMI patients.21 The studies are both multi-centre, multi-national, double-blinded RCTs with 2,300 patients and 2,000 patients, respectively. Both trials share identical primary end points of hospitalisation for heart failure or cardiovascular mortality at one year. In these trials, STEMI patients will be randomised to PPCI with or without RIPerC. The RIPerC will be delivered either in-ambulance or on arrival to the PPCI centre depending on average national transit time. The results of these trials are hoped to be released in approximately 18 months.
RIPerC in thrombolysis
Though the ERIC-lysis8 study provided evidence for clinically applied RIPerC, no large-scale trials have explored RIPerC in STEMI patients treated with thrombolysis. While thrombolysis as an intervention has largely been superseded by PPCI in the developed world, in the developing world, thrombolysis remains an important first-line therapy for STEMI. Large-scale human trials are still needed to provide evidence for a cost-neutral, life-saving therapeutic adjunct to thrombolysis.
PPerC in PPCI
Though the aforementioned CIRCUS18 study of PPerC was neutral at one year, the trial has not yet completed follow-up. It may yet show an effect of CsA at the three-year end point and, as such, full conclusions may not currently be drawn. It is also worth noting that CsA is not the sole pharmacological agent: there are multiple potential pharmacological targets within the increasingly well-characterised cell-death pathway that may yet yield an efficacious PPerC option. Indeed, re-purposing existing cardiovascular drugs may yield significant benefit to improve cardiovascular outcomes following an acute MI.
Conclusion
Reperfusion injury plays a significant role in the evolution of the final MI size, yet physicians have no effective tools with which to combat the phenomenon. Recent small-scale human trials exploring the variety of ischaemic-conditioning modalities have shown some promise. If large-scale trials can provide the evidence, reperfusion injury may yet become amenable to treatment. In remote conditioning exists a potentially cheap and non-invasive method of therapy, but as with pharmacological conditioning, the holy grail of an effective clinical intervention against IRI has yet to be realised. However, the search for an effective adjunct to reperfusion, if successful, will be well-rewarded by further reductions in cardiovascular mortality and morbidity and the consequent reduction upon the burgeoning socioeconomic healthcare burden worldwide.
Conflict of interest
None declared.
Key messages
- Rapid reperfusion is essential to reduce myocardial infarction following acute coronary obstruction, but reperfusion brings a cost: reperfusion injury
- Currently, there are no targeted approaches to mitigate against reperfusion injury, but cardioprotective strategies do exist in the form of ‘conditioning’. These strategies may be pharmacological, ischaemic or remote ischaemic that can be applied before (pre-conditioning), during (per-conditioning) or immediately after (post-conditioning) the injurious ischaemic insult
- Conditioning has been demonstrated to work in small, proof-of-concept studies, but have proven far more challenging in respect to larger outcome studies. There are multiple confounders at play, from the size of the initial injury (too small and the efficacy of the intervention is difficult to demonstrate) to concurrent drug interventions (that may either block protective signalling or be protective in their own right).
- Conditioning is an approach that has great potential to further improve patient outcomes beyond that achieved with current interventional strategies: large outcome studies in primary percutaneous coronary intervention (PCI) are currently recruiting and the results are awaited
References
1. Yellon DM, Hausenloy DJ. Myocardial reperfusion injury. N Engl J Med 2007;357:1121–35. https://doi.org/10.1056/NEJMra071667
2. Yellon DM, Alkhulaifi AM, Pugsley WB. Preconditioning the human myocardium. Lancet 1993;342:276–7. https://doi.org/10.1016/0140-6736(93)91819-8
3. Jenkins DP, Pugsley WB, Alkhulaifi AM, Kemp M, Hooper J, Yellon DM. Ischaemic preconditioning reduces troponin T release in patients undergoing coronary artery bypass surgery. Heart 1997;77:314–18. https://doi.org/10.1136/hrt.77.4.314
4. Hausenloy DJ, Mwamure PK, Venugopal V et al. Effect of remote ischaemic preconditioning on myocardial injury in patients undergoing coronary artery bypass graft surgery: a randomised controlled trial. Lancet 2007;370:575–9. https://doi.org/10.1016/S0140-6736(07)61296-3
5. Venugopal V, Hausenloy DJ, Ludman A et al. Remote ischaemic preconditioning reduces myocardial injury in patients undergoing cardiac surgery with cold-blood cardioplegia: a randomised controlled trial. Heart 2009;95:1567–71. https://doi.org/10.1136/hrt.2008.155770
6. Piot C, Croisille P, Staat P et al. Effect of cyclosporine on reperfusion injury in acute myocardial infarction. N Engl J Med 2008;359:473–81. https://doi.org/10.1056/NEJMoa071142
7. Ibanez B, Macaya C, Sánchez-brunete V et al. Effect of early metoprolol on infarct size in ST-segment-elevation myocardial infarction patients undergoing primary percutaneous coronary intervention: the Effect of Metoprolol in Cardioprotection During an Acute Myocardial Infarction (METOCARD-CNIC) trial. Circulation 2013;128:1495–503. https://doi.org/10.1161/CIRCULATIONAHA.113.003653
8. Yellon DM, Ackbarkhan AK, Balgobin V et al. Remote ischemic conditioning reduces myocardial infarct size in STEMI patients treated by thrombolysis. J Am Coll Cardiol 2015;65:2764–5. https://doi.org/10.1016/j.jacc.2015.02.082
9. Sloth AD, Schmidt MR, Munk K et al. Improved long-term clinical outcomes in patients with ST-elevation myocardial infarction undergoing remote ischaemic conditioning as an adjunct to primary percutaneous coronary intervention. Eur Heart J 2014;35:168–75. https://doi.org/10.1093/eurheartj/eht369
10. Kitakaze M, Asakura M, Kim J et al. Human atrial natriuretic peptide and nicorandil as adjuncts to reperfusion treatment for acute myocardial infarction (J-WIND): two randomised trials. Lancet 2007;370:1483–93. https://doi.org/10.1016/S0140-6736(07)61634-1
11. Lønborg J, Vejlstrup N, Kelbæk H et al. Exenatide reduces reperfusion injury in patients with ST-segment elevation myocardial infarction. Eur Heart J 2012;33:1491–9. https://doi.org/10.1093/eurheartj/ehr309
12. Lønborg J, Kelbaek H, Vejlstrup N et al. Cardioprotective effects of ischemic postconditioning in patients treated with primary percutaneous coronary intervention, evaluated by magnetic resonance. Circ Cardiovasc Interv 2010;3:34–41. https://doi.org/10.1161/CIRCINTERVENTIONS.109.905521
13. Freixa X, Bellera N, Ortiz-pérez JT et al. Ischaemic postconditioning revisited: lack of effects on infarct size following primary percutaneous coronary intervention. Eur Heart J 2012;33:103–12. https://doi.org/10.1093/eurheartj/ehr297
14. Crimi G, Pica S, Raineri C et al. Remote ischemic post-conditioning of the lower limb during primary percutaneous coronary intervention safely reduces enzymatic infarct size in anterior myocardial infarction: a randomized controlled trial. JACC Cardiovasc Interv 2013;6:1055–63. https://doi.org/10.1016/j.jcin.2013.05.011
15. Meybohm P, Bein B, Brosteanu O et al. A multicenter trial of remote ischemic preconditioning for heart surgery. N Engl J Med 2015;373:1397–407. https://doi.org/10.1056/NEJMoa1413579
16. Hausenloy DJ, Candilio L, Evans R et al. Remote ischemic preconditioning and outcomes of cardiac surgery. N Engl J Med 2015;373:1408–17. https://doi.org/10.1056/NEJMoa1413534
17. Kottenberg E, Thielmann M, Bergmann L et al. Protection by remote ischemic preconditioning during coronary artery bypass graft surgery with isoflurane but not propofol – a clinical trial. Acta Anaesthesiol Scand 2012;56:30–8. https://doi.org/10.1111/j.1399-6576.2011.02585.x
18. Cung TT, Morel O, Cayla G et al. Cyclosporine before PCI in patients with acute myocardial infarction. N Engl J Med 2015;373:1021–31. https://doi.org/10.1056/NEJMoa1505489
19. Ottani F, Latini R, Staszewsky L et al. Cyclosporine A in reperfused myocardial infarction: the multicenter, controlled, open-label CYCLE trial. J Am Coll Cardiol 2016;67:365–74. https://doi.org/10.1016/j.jacc.2015.10.081
20. Manintveld OC, te Lintel Hekkert M, van den Bos EJ et al. Cardiac effects of postconditioning depend critically on the duration of index ischemia. Am J Physiol Heart Circ Physiol 2007;292:H1551–H1560. https://doi.org/10.1152/ajpheart.00151.2006
21. Hausenloy DJ, Kharbanda R, Rahbek Schmidt M et al. Effect of remote ischaemic conditioning on clinical outcomes in patients presenting with an ST-segment elevation myocardial infarction undergoing primary percutaneous coronary intervention. Eur Heart J 2015;36:1846–8. https://doi.org/10.1093/eurheartj/ehv249

