Held on 2nd March 2017, the 9th British Society for Heart Failure (BSH) Training and Revalidation Day drew heart failure experts to London from around the UK. A superb series of talks addressed fields including the intersection of cardio-oncology and cardio-obstetric care, the role of the primary care heart failure specialist, and the approach to patients with advanced heart failure. Dr Simon Beggs reports on some of the highlights from a day that proved informative and enjoyable in equal measure.
Cardio-oncology and obstetrics
Many cancer therapies are cardiotoxic, and as cancer survival has improved over recent decades so the number of patients living to develop cardiovascular complications of these therapies has risen. A recent position statement by the European Society of Cardiology stresses that “the cured cancer patient of today…[is at risk of becoming]…the heart failure patient of tomorrow”1 and management of these patients increasingly involves a cardiologist.
In a highly educational presentation, Dr Zaheer Yousef (University Hospital of Wales, Cardiff) addressed the management of left ventricular systolic dysfunction (LVSD) in patients who have received, or are receiving, treatments for cancer. It is important for the clinician to recognise that conventional chemotherapy agents have specific cardiotoxic profiles. Anthracyclines, such as doxorubicin, notoriously induce LVSD via mechanisms including free-radical derived myocyte damage; alkylating agents and anti-metabolites (respective examples being cisplatin and 5-FU) traditionally cause myocardial ischaemia, and the anti-microtubule agent paclitaxel classically causes conduction disturbances and arrhythmias, but may induce myocardial dysfunction at higher doses. Cardiovascular risk profiling is thus important in these patients, and collaborative decision making by both an oncologist and cardiologist may be the most appropriate route to selecting a treatment regime.
Anthracyclines are perhaps the prototypical cardiotoxic chemotherapeutic drugs, yet their efficacy as antineoplastic agents means they are used in more than 50% of solid organ tumours and haematological malignancies. Cardiotoxicity is related to total cumulative dose, with over 250 mg/m2 of doxorubicin or equivalent conferring higher risk, and historical data indicate that late-onset effects, when measured over years to decades, include LVSD in nearly 40% of patients.2 Efforts to risk stratify cancer survivors who have received such treatment have led to the definition of cancer therapeutics–related cardiac dysfunction (CTRCD) as a decrease in the left ventricular ejection fraction (LVEF) of >10%, to a value below the lower limit of normal.3 However, even in those patients with preserved resting measures of global systolic cardiac function, significantly blunted markers of cardiac deformation may be unmasked during physiological stress echo. In those patients with LVSD, there is evidence for a benefit of angiotensin converting enzyme (ACE) inhibitors and beta blockers and, specifically, in those patients receiving anthracyclines, co-administration of the iron chelator dexrazone may be considered. The role of cardiac biomarkers such as troponin and natriuretic peptides in the surveillance of patients receiving chemotherapy is an area of ongoing research.
Case study: lessons learned
In a complimentary presentation, Dr Mark Drury-Smith (University Hospital of Wales, Cardiff) described the case of a 27-year old woman admitted with life-threatening acute heart failure on the second post-partum day following delivery of her first child. Her past medical history comprised previous acute lymphoblastic leukaemia when she was aged 4, which had been cured following treatment with high dose anthracyclines and mediastinal radiotherapy. Unfortunately, pre-pregnancy planning and counselling appeared suboptimal despite markers of cardiovascular risk that included left-bundle branch block and an anterior wall motion abnormality, with the significance of these not fully recognised. Collaborative cardio-oncology working is important to identify those cancer survivors at highest risk of cardiovascular complications, with this talk emphasising the potentially disastrous consequences of failing to do so. In this context, appropriate assessment and planning in the pre-pregnancy phase is of particular importance.
Heart failure and pregnancy
Dr Diane Barker (Royal Stoke University Hospital, Stoke-on-Trent) took up the mantle with a talk entitled ‘What to do when a heart failure patient becomes pregnant’. Physiological changes in pregnancy include an increase in plasma volume of up to 40%, physiological anaemia, and transient left ventricular (LV) dilatation. In patients with pre-existing cardiac dysfunction, the danger posed by these changes is potentially substantial. The optimal approach to such patients thus begins in the pre-conception phase, permitting careful assessment of LV function, and ensuring that both the woman and her partner are fully appraised of the risks of pregnancy. Pre-conception risk stratification can be performed using guideline-recommended methods including the modified WHO classification of maternal cardiovascular risk.4 Additional markers of maternal hazard can be sought using objective assessments such as a cardiopulmonary exercise test. In conditions where risk is especially high (WHO class IV), pregnancy is ‘contraindicated’.
In the patient who proceeds to conception and beyond, the cardiologist’s armamentarium of heart failure pharmacotherapies is restricted due to potential teratogenicity. Beta blockers are generally used, although these may result in low birth weight infants. There is no ‘acceptable’ period for an ACE inhibitor or angiotensin receptor blocker (ARB) during pregnancy, and these should be discontinued prior to conception. Hydralazine and diuretics are thought to be safe and are regularly used in pregnant heart failure patients. As labour approaches, expert multidisciplinary involvement is key, and important steps include early epidural analgesia, a facilitated second stage of labour and consideration of corticosteroids for fetal lung maturity where appropriate. Meticulous management must be maintained post-partum, with maternal risk high in the first 48 hours to two weeks after delivery.
In an accompanying case presentation, Dr Anik Appaji (Royal Stoke University Hospital, Stoke-on-Trent) highlighted the important lessons learned from a potentially avoidable death due to worsening heart failure in a pregnant patient. In relation to a tragic and emotive case, the importance of attentive, integrated care in patients with pre-existing heart failure who fall pregnant was made clear, with two points of particular note. First, that women with a known history of cardiac disease, including cardiomyopathy, must always be referred for care in a joint obstetric-cardiology clinic or by a cardiologist with expertise in the care of women with heart disease in pregnancy. Second, that missed diagnoses of pulmonary oedema or acidosis are common mistakes in maternal cardiac deaths.
Primary care
Ms Suzy Hughes (Clinical Nurse Specailist, Gloucestershire) opened this session by describing her role as a community-based heart failure specialist nurse (HFSN) in the care of patients in a rural setting. From the point of diagnosis to the institution of terminal care, the key messages were the importance of working in partnership both with other specialists, and with patients themselves. Patient consultations by community HFSNs are often the trigger for the delivery of appropriate specialist healthcare services to these patients. Specialist involvement might include general practitioners (GPs) with an interest in heart failure, health and social care agencies, community pharmacists, district nurses, or input from secondary care, including cardiologists. In rural Gloucestershire, the community model of working is strengthened by a tele-health service that permits remote assessment of blood pressure, pulse and weight, and resultantly enables more frequent titration of heart failure medications than would otherwise be possible. In parallel, a rapid response nurse team can provide intravenous diuretic therapy for a patient in their own home, with the aim of avoiding hospital admission where feasible and appropriate. The difficulties of independent decision making for the community HFSN were explored, and the utility of expert guidance in hospital clinic and discharge letters was emphasised.
Dr Jim Moore (Stoke Road Surgery, Cheltenham) continued the theme with an exploration of the Gloucestershire model of community heart failure care from a GP’s perspective. The ideal model of care ensures patients receive the right care, at the right time, by the right team and in the right place. A particular challenge to GPs under the current system, however, is that of making heart failure diagnoses in the community, confronted as they are by earlier stages of disease, with limited consultation time, and often hampered by a lack of continuity in decision-making. Where open access echocardiography is available, clinician-led reporting may not be, thus interpretation of echocardiographic data may fall to the GP alone. The benefits of expert support in such situations are clear, and the utility of regular multidisciplinary meetings involving secondary care clinicians – among others – was highlighted.
Advanced heart failure
Cardiogenic shock (CS) is usually fatal if left untreated. Mortality even amongst patients receiving optimal, aggressive, therapeutic interventions approaches 50%.5 Dr Stephen Pettit (Papworth Hospital NHS Foundation Trust, Cambridge) opened the session by discussing the management of this syndrome. Invasive pressure and cardiac output monitoring using a pulmonary artery catheter (PAC) is frequently indicated in patients with CS, and an appropriate understanding of cardiovascular physiology is mandatory (figure 1). PAC-derived haemodynamic data plays an important role in guiding therapy for CS, and may in certain cases reveal an alternative, non-cardiac cause of shock. The severity of CS is graded according to the INTERMACS classification, and this plays a role in selecting a therapeutic strategy. In terms of pharmacotherapy, an array of inotropic and vasoactive drugs may be used to treat CS, although these medications do not improve outcomes, and inotropes worsen them.6
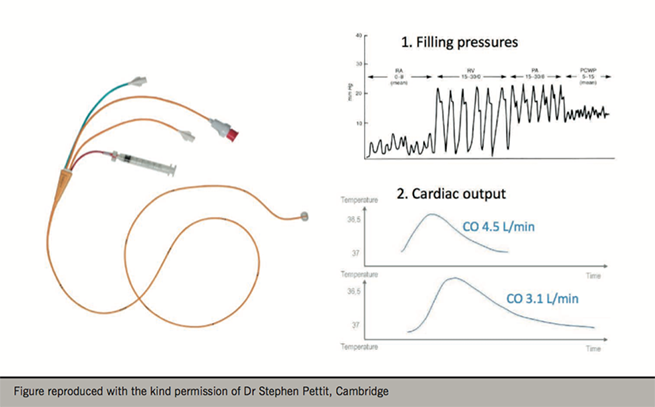
For certain patients, mechanical circulatory support (MCS) may also be an appropriate strategy. The goals of MCS include unloading of the injured ventricle, maintaining end-organ perfusion, and allowing the myocardium to declare its potential for recovery. The most basic form of MCS is the intra-aortic balloon pump, which augments mean arterial pressure but was recently shown not to improve outcomes in shock resulting from acute myocardial infarction.7 More advanced MCS, such as extracorporeal membrane oxygenation and the newer generation of percutaneous left ventricular assist devices (LVADs), such as the TandemHeart™or Impella™, are generally reserved for patients eligible for cardiac transplant. In this respect, the message was to enlist expert help early in CS, in part because appropriate management should ‘start with the end in mind’.
Following on from this with a real world example, Dr Darshan Brahmbhatt (East of England Deanery, Cambridge) presented the case of a patient with presumed CS who had been commenced on intravenous levosimendan and transferred to his tertiary cardiac centre for emergency assessment. Investigation with a pulmonary arterial catheter (PAC) revealed a cardiac index of 3.9 L/min/m2 (surprisingly high) and a systemic vascular resistance of 423 dyn/s/cm5 (markedly low), thus refuting the working diagnosis of CS and pointing instead towards a state of profound vasodilation. Whilst the final underlying diagnosis was thought to be septic shock, ironically the calcium-sensitising inodilator, levosimendan, had potentially exacerbated the vasodilatory effects of systemic infection. The conclusion was that despite a lack of evidence for the benefit of their use,8,9 PACs continue to play a potentially vital role in guiding management of the shocked patient.
Closing the session, Dr Steve Shaw (Wythenshawe Hospital, Manchester) explored the continuing role of cardiac transplantation in the era of LVADs. In 2001, the landmark REMATCH trial heralded the introduction of the HeartMate XVE™ long-term LVAD to bridge patients to cardiac transplant.10 Subsequent studies have suggested incremental benefits from newer LVAD models such as the HeartMate II™, HeartWare™ HVAD and HeartMate III™.11–13 For patients with advanced heart failure, the advantages of LVADs include control over timing of the surgical operation, the lack of requirement for immunosuppression, and improved outcomes with each new generation of device. Conversely, LVADs necessitate substantial lifestyle limitations, carry major cost implications for healthcare providers, and cannot yet compete with the long-term survival rates seen in transplant patients. Importantly, some patients, such as those with biventricular failure, may be eligible for transplantation but not for an LVAD. It was emphasized that the modern advanced heart failure programme will utilize both LVADs and transplantation, and that these should be seen as complimentary, rather than competing treatments.
Acknowledgment
The BSH gratefully acknowledges the support provided by the Friends of BSH: Abbott, Bayer, Biotronik, Boston Scientific, Medtronic, Novartis Pharmaceuticals, Roche Diagnostics, Servier Laboratories and Vifor Pharma.
Conflict of interest
None declared.
Diary dates
Future BSH meetings include:
- 23rd–24th November 2017: The 20th BSH Annual Autumn Meeting, QE II Centre, London
- 1st March 2018: 10th BSH Heart Failure Day for Revalidation and Training, Birmingham
- 2nd March 2018: 9th BSH Heart Failure Nurse and Healthcare Professional Study Day, Birmingham
Further information about the meeting and the Society can be obtained from:
British Society for Heart Failure; email: [email protected]; www.bsh.org.uk; @BSHeartFailure
References
1. Eschenhagen T, Force T, Ewer MS et al. Cardiovascular side effects of cancer therapies: a position statement from the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail 2011;13:1–10. https://doi.org/10.1093/eurjhf/hfq213
2. Shan K, Lincoff AM, Young JB. Anthracycline-induced cardiotoxicity. Ann Intern Med 1996;125(1):47–58. https://doi.org/10.7326/0003-4819-125-1-199607010-00008
3. Zamorano JL, Lancellotti P, Rodriguez Munoz D et al. 2016 ESC position paper on cancer treatments and cardiovascular toxicity developed under the auspices of the ESC Committee for Practice Guidelines. Eur Heart J 2016;37:2768–801. https://doi.org/10.1093/eurheartj/ehw211
4. Regitz-Zagrosek V, Blomstrom Lundqvist C, Borghi C et al. ESC guidelines on the management of cardiovascular diseases during pregnancy. The Task Force on the Management of Cardiovascular Diseases during Pregnancy of the European Society of Cardiology (ESC). Eur Heart J 2011;32:3147–97. https://doi.org/10.1093/eurheartj/ehr218
5. Puymirat E, Fagon JY, Aegerter P et al. Cardiogenic shock in intensive care units: evolution of prevalence, patient profile, management and outcomes, 1997–2012. Eur J Heart Fail 2017;19:192–200. https://doi.org/10.1002/ejhf.646
6. Elkayam U, Tasissa G, Binanay C et al. Use and impact of inotropes and vasodilator therapy in hospitalized patients with severe heart failure. Am Heart J 2007;153(1):98–104. https://doi.org/10.1016/j.ahj.2006.09.005
7. Thiele H, Zeymer U, Neumann F-J et al. Intraaortic balloon support for myocardial infarction with cardiogenic shock. N Engl J Med 2012;367:1287–96. https://doi.org/10.1056/NEJMoa1208410
8. Binanay C, Califf RM, Hasselblad V et al. Evaluation study of congestive heart failure and pulmonary artery catheterization effectiveness: the ESCAPE trial. JAMA 2005;294(13):1625–33. https://doi.org/10.1001/jama.294.13.1625
9. Harvey S, Harrison DA, Singer M et al. Assessment of the clinical effectiveness of pulmonary artery catheters in management of patients in intensive care (PAC-Man): a randomised controlled trial. Lancet 2005;366:472–7. https://doi.org/10.1016/S0140-6736(05)67061-4
10. Rose EA, Gelijns AC, Moskowitz AJ et al. Long-term use of a left ventricular assist device for end-stage heart failure. N Engl J Med 2001;345(20):1435–43. https://doi.org/10.1056/NEJMoa012175
11. Slaughter MS, Rogers JG, Milano CA. Advanced heart failure treated with continuous-flow left ventricular assist device. N Engl J Med 2009;361:2241–51. https://doi.org/10.1056/NEJMoa0909938
12. Aaronson KD, Slaughter MS, Miller LW et al. Use of an intrapericardial, continuous-flow, centrifugal pump in patients awaiting heart transplantation. Circulation 2012;125:3191–200. https://doi.org/10.1161/CIRCULATIONAHA.111.058412
13. Mehra MR, Naka Y, Uriel N. A fully magnetically levitated circulatory pump for advanced heart failure. N Engl J Med 2017;376:440–50. https://doi.org/10.1056/NEJMoa1610426
