The inflammatory component of ischaemic heart disease (IHD) is well recognised. An elderly male, following primary percutaneous coronary intervention (pPCI) for ST-elevation myocardial infarction (STEMI), had, otherwise unexplained, severely elevated C-reactive protein (CRP) prior to sudden cardiac death (SCD). Post-mortem showed only old infarct, no re-stenosis, and no evidence of inflammation elsewhere. The levels of CRP in this case are much higher than those documented previously in IHD. Current guidelines advocate for implantable cardioverter defibrillator (ICD) implantation after acute coronary syndrome (ACS) only in the context of left ventricular ejection fraction <35%, therefore, this patient would not qualify. Multiple risk-stratification tools have been developed to widen ICD prescription after ACS, but have not yet been integrated into the National Institute for Health and Care Excellence (NICE) guidelines. This case is a poignant reminder that we must widen ICD prescription, and CRP should be considered as a likely predictor.
Introduction
Primary percutaneous coronary intervention (pPCI) and stenting are considered first-line management of ST-elevation myocardial infarction (STEMI).1 There is a well-recognised inflammatory component to ischaemic heart disease (IHD), and, thus, C-reactive protein (CRP) has been implicated as a poor prognostic indicator for stent re-stenosis, cardiovascular mortality and all-cause mortality post-myocardial infarction (MI).1,2
Case
An 87-year-old man presented to Accident and Emergency (A&E) for “a one day history of severe neck/parietal headache on background of recent discharge from hospital with an MI”.
Past medical history included gout and essential hypertension, and, six weeks previously, he was managed for STEMI with pPCI and had three coronary stents inserted. He lived alone, independently, was a non-smoker and drank two to three pints of beer three to four times per week. His medications included aspirin, atorvastatin, bisoprolol, clopidogrel, furosemide and pantoprazole.
Thorough examination of neurological, cardiovascular, respiratory and abdominal systems were unremarkable except that the neck was painful on movement.
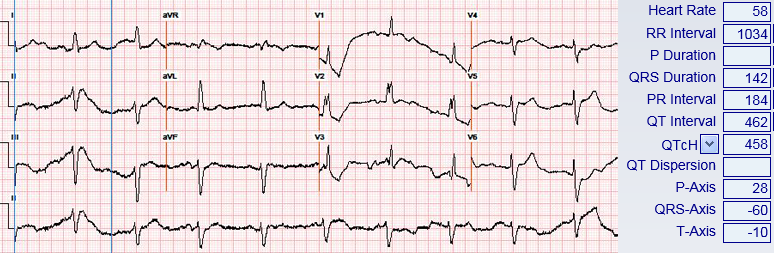
Investigations later attributed the presenting headache to a C6 nerve impingement, as evidenced by magnetic resonance imaging (MRI) of the spine. Incidentally, the admission CRP was 108.4 mg/L. Throughout his admission this fluctuated, reaching as high as 553 mg/L. Electrocardiogram (ECG) showed sinus bradycardia at 58 bpm, left-axis deviation and partial right bundle branch block (RBBB) (figure 1). His echocardiogram showed ‘mild’ left-ventricular systolic dysfunction (LVSD). Despite thorough investigation including infection screen, abdominal ultrasound, computed tomography (CT) of chest/abdomen/pelvis, autoimmune screen, MRI of spine, echocardiography and more, no inflammatory source was found except for an asymptomatic Klebsiella urine infection, which did not respond to antibiotics as per sensitivities. The patient unfortunately died suddenly on day 17, showing asystole throughout unsuccessful cardiopulmonary resuscitation (CPR).
Post-mortem revealed old, full-thickness posterior MI, and no recent changes. No inflammatory pathology was demonstrated elsewhere. Cause of death was documented as IHD.
Discussion
Since the fluctuating CRP showed no real temporal relationship with symptomatology, and did not respond to the management attempts, it is reasonable to suspect that it was raised as part of the chronic inflammatory processes of IHD, and the best efforts to find an alternative source were misguided.
When Wang et al. implicated the prognostic efficacy of high-sensitivity CRP:albumin ratio (CAR) in patients following ACS, they categorised patients in to high CAR (over 0.114) or low CAR (under 0.114) to compare incidence of major cardiac events.3 For comparison, in this case the CAR reached as high as 16.8, over 150 times the cut-off margin for ‘high CAR’.
With this, it is understandable that the cardiology team were not consulted, and multiple other specialties were drawn upon instead, due to better-established differential diagnoses of non-specific inflammation being taken into consideration. Sudden cardiac death (SCD) is responsible for roughly half of post-MI mortality.4 Theories regarding the mechanisms leading to SCD post-MI are varied and poorly understood but include repeated ischaemia or formation of ventricular re-entry circuits during myocardial remodelling. With this in mind, the chance of one test being used to accurately predict SCD is small.4 Multiple risk-stratification tools have been developed to predict SCD post-MI.5 One example of these is the Multicentre UnSustained Tachycardia Trial (MUSTT), which developed a risk-stratification score for arrhythmic death or cardiac arrest based upon the several predictors for SCD. Despite this, current National Institute for Health and Care Excellence (NICE) guidelines only advocate for primary ICD-implantation post-ACS in those with LVEF <35%.5,6
Further work is required to identify the approximately 50% of patients suffering SCD post-MI who do not fall in to the LVEF <35% category, in order to widen ICD-prescription, reducing mortality.6 Based on this case report, as well as previous implicating evidence, it is certainly plausible that elevated CRP may be a predictive factor.
Conclusion
This case is a poignant reminder that current guidelines for ICD prescription post-MI are suboptimal, such that only half of those suffering major cardiac events are identified, based on LVEF <35% being the only predictive factor recognised in the NICE guidelines. Further research to validate more predictive factors is required, and CRP should certainly be considered.
Conflicts of interest
None declared.
Funding
None.
Patient consent
Written consent for publication was obtained from next of kin.
Acknowledgements
Special thanks to Dr Mickey Jachuck, Consultant Cardiologist, and Dr Christopher Benson, Cardiology Registrar, South Tyneside Foundation Trust.
References
1. Rencuzogullari I, Karabağ Y, Çağdaş M et al. Assessment of the relationship between preprocedural C-reactive protein/albumin ratio and stent restenosis in patients with ST-segment elevation myocardial infarction. Rev Port Cardiol 2019;38:269–77. https://doi.org/10.1016/j.repc.2018.08.008
2. Oikawa T, Sakata Y, Nochioka K et al. Association between temporal changes in C-reactive protein levels and prognosis in patients with previous myocardial infarction – a report from the CHART-2 study. Int J Cardiol 2019;293:17–24. https://doi.org/10.1016/j.ijcard.2019.07.022
3. Wang W, Ren D, Wang C, Li T, Yao H, Ma S. Prognostic efficacy of high-sensitivity C-reactive protein to albumin ratio in patients with acute coronary syndrome. Biomark Med 2019;13:811–20. https://doi.org/10.2217/bmm-2018-0346
4. Waks J, Buxton A. Risk stratification for sudden cardiac death after myocardial infarction. Annu Rev Med 2018;69:147–64. https://doi.org/10.1146/annurev-med-041316-090046
5. National Institute for Health and Care Excellence. Implantable cardioverter defibrillators and cardiac resynchronisation therapy for arrhythmias and heart failure. TA314. London: NICE, 2014. Available from: https://www.nice.org.uk/guidance/ta314/
6. Goldenberg I, Vyas A, Hall W et al. Risk stratification for primary implantation of a cardioverter-defibrillator in patients with ischemic left ventricular dysfunction. J Am Coll Cardiol 2008;51:288–96. https://doi.org/10.1016/j.jacc.2007.08.058
