Risk stratification for sudden cardiac death (SCD) and the selection of patients for prophylactic implantable cardiac defibrillator (ICD) in hypertrophic cardiomyopathy (HCM) are still evolving and far from ideal. I present a historical case of HCM that did not have recognised SCD risk factors. This case highlights the deficiency of the present risk-stratification strategy for HCM and European Society of Cardiology (ESC) risk-scoring system.
Introduction
The ways of risk stratification for sudden cardiac death (SCD) and the selection of patients for prophylactic implantable cardiac defibrillator (ICD) in hypertrophic cardiomyopathy (HCM) are still evolving and far from ideal. The following is a historical case (initial presentation was in 2019) of HCM that did not have recognised SCD risk factors. The calculated European Society of Cardiology (ESC) RISK-SCD score1 was a 1.88% chance of SCD in five years and, hence, an ICD was not recommended. The patient died suddenly, 18 months after the diagnosis was made. The ESC had modified guidelines for prophylactic ICD implantation in HCM in 2023. However, even by present guidelines, this patient may not have satisfied the criteria for a prophylactic ICD. This case highlights the deficiency of the present risk-stratification strategy for HCM and ESC risk-scoring system.
Case presentation
A 50-year-old man presented to the emergency department with chest pain and dizziness. This happened after giving blood samples for tests requested by his general practitioner. The pain was across the chest and lasted for 20 minutes. There was no radiation to the left arm or jaws. He did not have any nausea or sweating. There was no shortness of breath, palpitation or syncope.
He had a history of high blood pressure, gout and dyslipidaemia. He was on amlodipine and ramipril for blood pressure, and colchicine for gout. He was not diabetic, a nonsmoker, and he drank alcohol occasionally. He did not have any cardiac history before. There was no history of coronary artery disease, sudden death or cardiomyopathy in the family.
Clinical examination revealed a blood pressure of 150/90 mmHg, pulse 80 bpm, jugular venous pressure (JVP) was not elevated, no pedal oedema, and apex was in the left fifth space on the midclavicular line and of normal character. There was a grade 2/6 systolic murmur in the aortic area.
Serum high-sensitivity troponin was 41 ng/L on admission and 50 ng/L after six hours (normal 0–17.5 ng/L). Routine bloods, including renal and liver function, were within normal limits. Electrocardiogram (ECG) showed sinus rhythm, normal QRS axis, and deep T-wave inversion in I, aVL, II and V2–V6.
Echocardiogram revealed left ventricular (LV) hypertrophy with a thickness of interventricular septum of 16 mm and posterior wall 14 mm at the end of diastole, preserved LV systolic function, no valvular abnormality and mild left atrial enlargement. Coronary angiogram showed normal unobstructed coronaries.
Cardiac magnetic resonance imaging (MRI) showed asymmetric septal and apical LV hypertrophy (maximum 22 mm in apical inferior wall), patchy mid-wall fibrosis of basal to mid-cavity of left ventricle with near confluent and circumferential apical subendocardial and mid-wall fibrosis (figures 1–3). Apical cavity obliteration with flow acceleration in mid and distal left ventricular outflow tract (LVOT) was noted associated with mild systolic anterior motion of the mitral valve and mitral regurgitation at rest into a moderately dilated left atrium (LA).
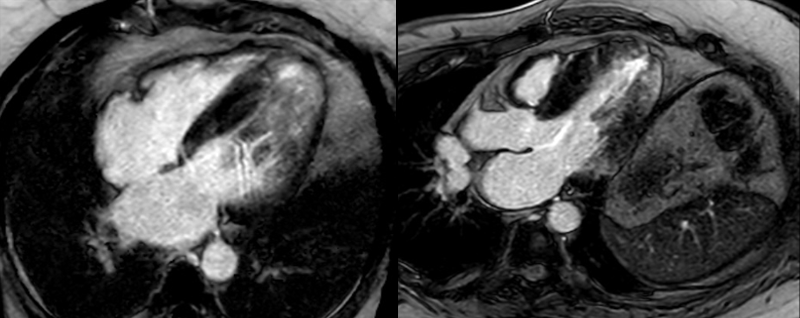
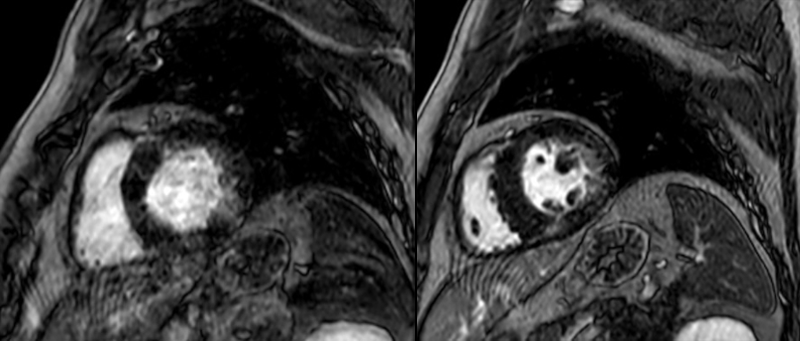
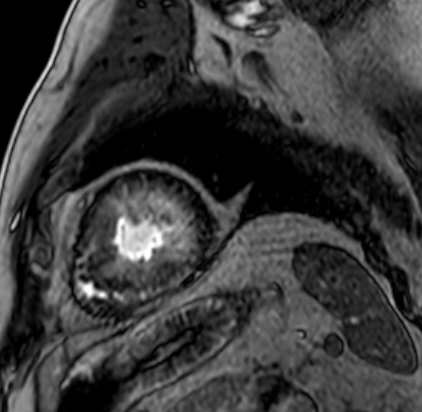
Initial differential diagnoses before cardiac MRI were hypertensive heart disease and HCM. Cardiac MRI was consistent with HCM, so a diagnosis of HCM was made.
A 48-hour ECG showed no evidence of non-sustained ventricular tachycardia (NSVT). ESC RISK-SCD score was calculated to be a 1.88% chance of sudden death in five years (age 51 years, maximum LV wall thickness 16 mm, LA size 52 mm, LVOT gradient 5 mmHg, no family history of SCD, no NSVT, no unexplained syncope). The diagnosis and management plan were explained to the patient. An ICD was not recommended.
Referral to the inherited cardiac disease clinic in a tertiary centre for further evaluation and genetic studies was offered, but the patient was not keen, as he needed to travel some distance to attend this clinic, and this was in the middle of the Covid-19 pandemic.
He was advised to take bisoprolol 2.5 mg once a day and ramipril 10 mg once a day. First follow-up was done at six months after the diagnosis, second follow-up at 12 months after the diagnosis. His blood pressure was well controlled at follow-up visits and there was no history of syncope. Third follow-up was arranged in another 12 months, with a plan to reassess SCD risk with history, 48-hour ECG and echocardiogram. The patient died suddenly before the third follow-up and in the post-mortem examination the cause of death was mentioned as hypertrophic heart disease.
Discussion
HCM is one of the common causes of SCD in the young population, and effective prevention of SCD with ICD has reduced more than 10-fold overall HCM-related mortality compared with the pre-ICD era. This patient, who had a diagnosis of HCM, had an untoward outcome. This was devastating for the patient’s family and the physicians involved in his care. Risk-stratification strategy failed on this occasion.
The ESC risk model has high specificity, but it is criticised for its poor sensitivity. The sensitivity for predicting SCD events averages 33% in the RISK-SCD model compared with the 95% sensitivity of the major risk factor-based strategy recommended by the American College of Cardiology/American Heart Association (ACC/AHA) 2020 guidelines.2 The ESC risk score system in the 2015 guidelines did not include novel risk factors like LV ejection fraction (LVEF) <50%, apical aneurysm and late gadolinium enhancement (LGE) on cardiac MRI. However, in the more recent guidelines in 2023, presence of LV dysfunction and LGE more than 15% have been mentioned as class 2b indications for ICD, even in the presence of a low RISK-SCD risk score.3 The ESC risk score is based on an echocardiogram as imaging modality, though MRI may be more accurate in estimating maximum LV wall thickness in unusual areas, and also in detecting apical aneurysm.
Extensive LGE on cardiac MRI has been identified as a risk factor for SCD in HCM. ACC/AHA guidelines (2020) recommend that the presence of extensive LGE can be considered as a 2b indication for ICD implantation in the absence of any other major risk factors. This patient had significant LGE. The definition of extensive LGE has been suggested by some authors to be comprising more than 15% of LV mass, though the methods used to measure varied.4 Some studies suggest that the location of LGE may be important, non-interventricular septal location was associated with higher risk of sudden death.5 There is a strong correlation between extensive LGE and NSVT on ECG monitoring, suggesting scarring can be a source of malignant ventricular arrhythmia in HCM.4 The extent of LGE, particularly the cut-off value of 15%, has been questioned by recent studies, as thresholds of 5% and 10% have also been shown to associate with higher SCD events, as compared with LGE below 5% after multi-variable adjustment.6 When LGE information was combined with T1 mapping, to represent both focal and diffuse fibrosis in personalised in silico modelling, the functional consequences of such structural changes were better predicted, and virtual induction stimulation protocol improved sensitivity, specificity and accuracy, outperforming current clinical risk predictors.7 Further research is needed in this area.
Key messages
- The ways of risk stratification for sudden cardiac death in hypertrophic cardiomyopathy are not ideal and foolproof
- The sensitivity of the European Society of Cardiology (ESC) RISK-SCD for identifying high-risk cases for sudden cardiac death is low
- Extensive late gadolinium enhancement (LGE) on cardiac magnetic resonance imaging (MRI) has been mentioned as a risk factor in the American College of Cardiology/American Heart Association (ACC/AHA) 2020 guidelines, and presence of extensive LGE can be considered as a 2b indication for defibrillator implantation in the absence of any other major risk factors. Recent ESC guidelines on cardiomyopathy also mention considering ICD as a 2b indication in the presence of >15% LGE, even in the presence of a low RISK-SCD score
Conflicts of interest
None declared.
Funding
None.
Patient consent
Consent to publish was obtained from the next of kin.
References
1. Elliott PM, Anastasakis A, Borger MA et al. 2014 ESC guidelines on diagnosis and management of hypertrophic cardiomyopathy: the task force for the diagnosis and management of hypertrophic cardiomyopathy of the European Society of Cardiology (ESC). Eur Heart J 2014;35:2733–79. https://doi.org/10.1093/eurheartj/ehu284
2. Maron BJ, Desai MY, Nishimura RA et al. Management of hypertrophic cardiomyopathy: JACC state-of-the-art review. J Am Coll Cardiol 2022;79:390–414. https://doi.org/10.1016/j.jacc.2021.11.021
3. Priori SG, Blomström-Lundqvist C, Mazzanti A et al.; ESC Scientific Document Group. 2015 ESC guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: the task force for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death of the European Society of Cardiology (ESC). Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC). Eur Heart J 2015;36:2793–867. https://doi.org/10.1093/eurheartj/ehv316
4. Ommen SR, Mital S, Burke MA et al. 2020 AHA/ACC guideline for the diagnosis and treatment of patients with hypertrophic cardiomyopathy: a report of the American College of Cardiology/American Heart Association joint committee on clinical practice guidelines. J Am Coll Cardiol 2020;76:e159–e240. https://doi.org/10.1016/j.jacc.2020.08.045
5. Li X, Lai L, Luo R et al. The clinical prognosis of presence and location of late gadolinium enhancement by cardiac magnetic resonance imaging in patients with hypertrophic cardiomyopathy: a single-centre cohort study. J Cardiovasc Transl Res 2021;14:1001–16. https://doi.org/10.1007/s12265-021-10107-x
6. Prasad SK, Akbari T, Bishop MJ, Halliday BP, Leyva-Leon F, Marchlinski F. Late gadolinium enhancement imaging and sudden cardiac death. Eur Heart J 2025;46:3555–68. https://doi.org/10.1093/eurheartj/ehaf464
7. O’Hara RP, Binka E, Prakosa A et al. Personalized computational heart models with T1-mapped fibrotic remodeling predict sudden death risk in patients with hypertrophic cardiomyopathy. Elife 2022;11:e73325. https://doi.org/10.7554/eLife.73325

