Hyperbaric oxygen therapy (HBOT) is successfully implemented for the treatment of several disorders. HBOT is a promising treatment modality for coronary artery disease (CAD), where outcomes are frequently poor despite early revascularisation. The aim of this study is to investigate the effect of HBOT on the left ventricular function of patients with CAD after reperfusion.
Electronic journal searching was performed in PubMed, ScienceDirect, and Cochrane to find studies that investigate the effect of HBOT on the myocardial function of patients with CAD. The primary outcomes were left ventricular end-diastolic volume (LVEDV), left ventricular end-systolic volume (LVESV), and left ventricular ejection fraction (LVEF). Meta-analyses were performed on included studies and mean differences (MD) and 95% confidence intervals (CI) were estimated using Review Manager v5.4.
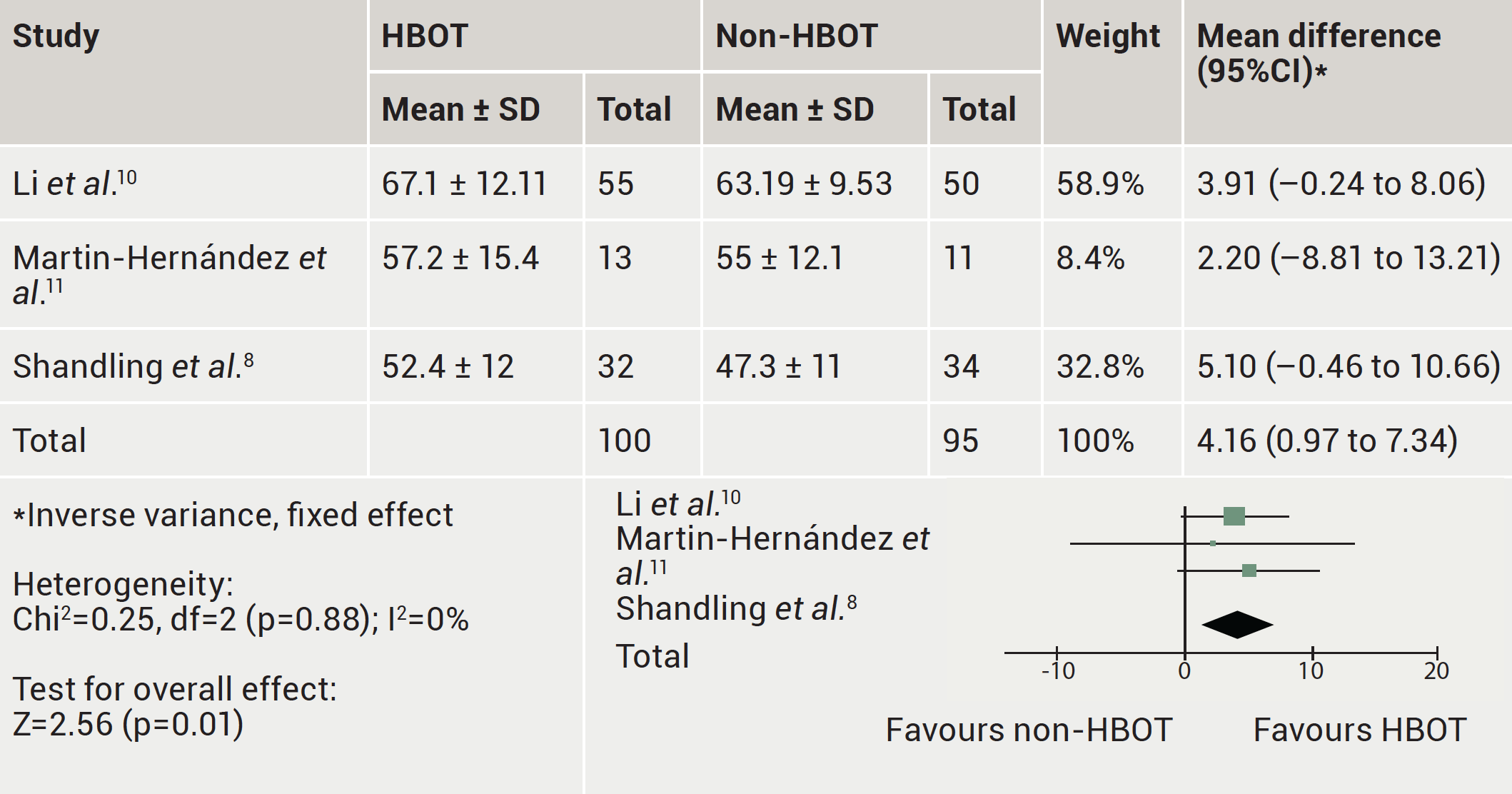
A total of three observational studies enrolling 195 participants were included in our analysis. HBOT significantly increased LVEF by 4.16% in patients with CAD after revascularisation compared with non-HBOT (MD=4.16, 95%CI 0.97 to 7.34, p=0.01). There was no statistical significance observed in the HBOT versus non-HBOT comparison on LVEDV (MD=–1.63, 95%CI –6.52 to 3.26, p=0.51) and LVESV (MD=–1.58, 95%CI –4.06 to 0.90, p=0.21).
In general, this meta-analysis shows HBOT significantly increased LVEF in patients with CAD after revascularisation compared with non-HBOT. There were no significant changes in LVEDV and LVESV in the HBOT group and non-HBOT group.
Introduction
According to projections, coronary artery disease (CAD), which is brought on by the atherosclerotic process in coronary arteries, will continue to be the world’s leading cause of death and morbidity.1,2 The development of coronary lesions may increase the risk of mortality for patients by causing serious adverse cardiovascular events including acute coronary syndrome (ACS). In the treatment of CAD, reperfusion via percutaneous coronary intervention (PCI) combined with the placement of drug-eluting stents (DES) and fibrinolytics has considerably improved patient clinical results.1,2 Even while it significantly lessens post-infarct consequences, there may still be some reperfusion damage.3,4
The use of hyperbaric oxygen treatment (HBOT) has been developed to reduce the after effects of cardiac ischaemia injury. HBOT is a non-invasive adjunctive therapy that can enhance reperfusion after CAD.5–7 In a hyperbaric chamber, patients breathe 100% oxygen to raise their arterial partial pressure of oxygen (PaO2). It has been proposed that this rise in PaO2 has therapeutic value for conditions characterised by systemic or local tissue hypoxia. In the HOT-MI (Hyperbaric Oxygen and Thrombolysis in Myocardial Infarction) study, it was discovered that HBOT improved left ventricular ejection fraction (LVEF) in ST-elevation myocardial infarction (STEMI) patients; there was a 30% decrease in the peak of creatine phosphokinase (CK) at 12 and 24 hours, decreased discomfort, and decreased ST-segment elevations.8 A substantial difference in the left ventricular end-systolic volume (LVESV) and an improvement in the LVEF after HBOT were discovered in thrombolysis patients via another investigation.5 Generally, HBOT has been found to decrease re-infarction and enhance survival after two and five years.9
However, to our knowledge, there has been no meta-analysis report regarding the use of HBOT, specifically after reperfusion in patients with CAD, that assessed left ventricular function. In this study, we aimed to investigate the effect of HBOT on the left ventricular function of patients with CAD after reperfusion.
Method
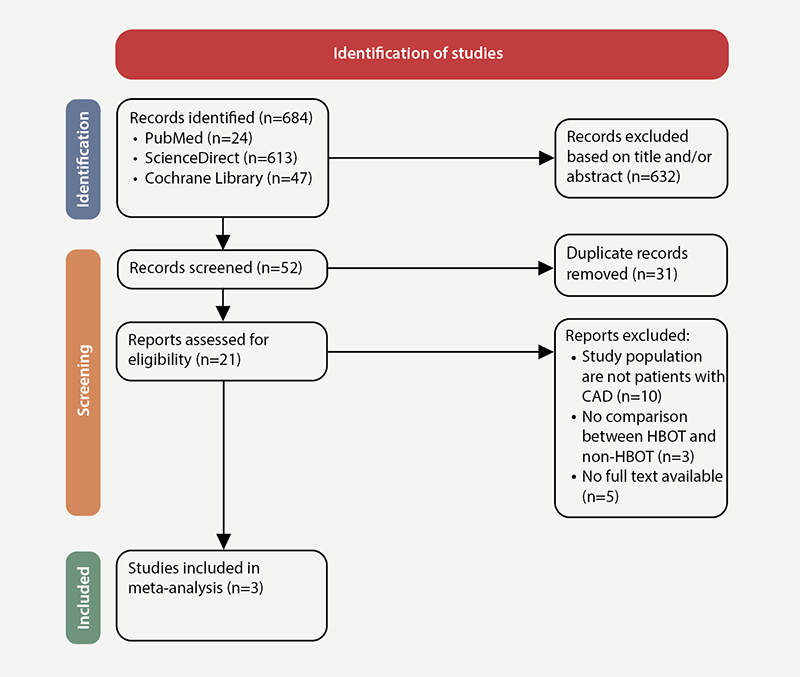
This systematic review and meta-analysis used the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 checklist and the PRISMA extension for searching to conduct the study (figure 1).
Data sources and search strategy
A systematic electronic search of PubMed, ScienceDirect, and Cochrane library was conducted from January 1992 to December 2022, for all randomised-controlled trials (RCTs) and observational studies that assessed the effects of HBOT on the myocardial function of patients with CAD. In addition, bibliographies of pertinent editorials and review articles were manually searched for potentially relevant articles. MeSH terms along with Boolean operators were used to produce a search strategy for each database.
Study selection process
After removing duplicate entries, KAAPP and NGAMSDC independently reviewed first search records based on the title and abstract. After that, a separate complete text examination was carried out. The following criteria for studies were used for inclusion: comparison of the results of HBOT with non-HBOT in patients with CAD; English-language study analyses. Consensus was reached in resolving disagreements, including those of the other author (YP).
Outcome measures
The outcomes of interest for this meta-analysis were left ventricular end-diastolic volume (LVEDV), LVESV and LVEF.
Data extraction and quality assessment
The baseline parameters of included studies were retrieved by KAAPP and NGAMSDC. The chosen Newcastle-Ottawa scale (NOS) for observational studies was then used by KAAPP and NGAMSDC to conduct a comprehensive quality assessment of the included research. The quality of investigations was rated as poor (5 points), moderate (5–7 points), or good (>7 points). Any differences were settled by consensus, incorporating the other author’s viewpoint (YP).
Statistical analysis
The Mantel-Haenszel fixed-effects models using mean difference (MD) as the effect measure and the corresponding 95% confidence interval (CI) were used to pool the data. The Higgins I2 statistic was used to gauge statistical heterogeneity between groups. In particular, an I2=0 meant that there was no heterogeneity, and we defined substantial heterogeneity as I2 values exceeding 50%. Given the study heterogeneity, we utilised the random-effect models to compute the pooled mean difference. We used Egger’s test funnel plot to assess the potential publication bias for this study. For all analyses, Review Manager 5.4.1 was employed. Statistical significance was defined as a two-sided p value of 0.05 or less.
Results
Study search and selection results
The first search method produced 684 entries. Three articles were included in the systematic review and meta-analysis after a complete text analysis of 21 potentially relevant records (figure 1).
Baseline characteristics and quality assessment
The characteristics of the included studies are presented in table 1.8,10,11 All studies were cohort studies and the authors did not find RCT studies suitable for inclusion in this systematic review and meta-analysis. Among the three included studies, the total number of patients was 195; 100 patients received HBOT and 95 patients did not receive HBOT (non-HBOT).
Table 1. Main characteristics of the studies included in the meta-analysis
| Author | Year | Country | Design | Sample size | Follow-up, weeks | Outcome | |
| HBOT | Non-HBOT | ||||||
| Li et al.10 | 2018 | China | Cohort | 55 | 60 | 4 | LVEF, LVEDV, LVESV |
| Martin-Hernández et al.11 | 2020 | Mexico | Cohort | 13 | 11 | 6 | LVEF, LVEDV, LVESV |
| Shandling et al.8 | 1997 | USA | Cohort | 32 | 34 | Not explained | LVEF |
| Key: HBOT = hyperbaric oxygen therapy; LVEDV = left ventricular end-diastolic volume; LVEF = left ventricular ejection fraction; LVESV = left ventricular end-systolic volume | |||||||
In the three studies, quality and bias risk were evaluated using the Newcastle-Ottawa scale (table 2). All observational studies ranked their overall risk of bias as moderate, and some were classified as high-quality research. No observational research has a significant bias risk.
Table 2. Quality assessment of included studies based on Newcastle-Ottawa scale
| Author | Year | Country | Selection | Comparability | Outcome | Total |
| Li et al.10 | 2018 | China | *** | ** | *** | 7 |
| Martin-Hernández et al.11 | 2020 | Mexico | *** | ** | ** | 7 |
| Shandling et al.8 | 1997 | USA | **** | ** | *** | 9 |
Pooled analyses for clinical outcomes
Effect of HBOT on LVEF
The LVEF was reported in three trials. HBOT significantly increased LVEF by 4.16% in patients with CAD after revascularisation compared to non-HBOT (MD=4.16, 95%CI 0.97 to 7.34, p=0.01) (figure 2).
| Key: CI = confidence interval; HBOT = hyperbaric oxygen therapy; SD = standard deviation |
Effect of HBOT on LVEDV
The LVEDV was reported by two trials. There was no statistical significance observed in the HBOT versus non-HBOT comparison on LVEDV (MD=–1.63, 95%CI –6.52 to 3.26, p=0.51) (table 3).
Table 3. The effect of HBOT on left ventricular end-diastolic volume (LVEDV)
| Study | HBOT | Non-HBOT | Weight | Mean difference (95%CI)* | ||
| Mean ± SD | Total | Mean ± SD | Total | |||
| Li et al.10 | 79.33 ± 12.62 | 55 | 81.27 ± 13.19 | 50 | 97.8% | –1.94 (–6.89 to 3.01) |
| Martin-Hernández et al.11 | 111.9 ± 45.3 | 13 | 99.9 ± 36.8 | 11 | 2.2% | 12.00 (–20.85 to 44.85) |
| Total | 68 | 61 | 100% | –1.63 (–6.52 to 3.26) | ||
| Heterogeneity: Chi2=0.68, df=1 (p=0.41); I2=0% | ||||||
| Test for overall effect: Z=0.65 (p=0.51) | ||||||
| *Inverse variance, fixed effect Key: CI = confidence interval; HBOT = hyperbaric oxygen therapy; SD = standard deviation |
||||||
Effect of HBOT on LVESV
The LVESV was reported by two trials. There was no statistical significance observed in the HBOT versus non-HBOT comparison on LVESV (MD=–1.58, 95%CI –4.06 to 0.90, p=0.21) (table 4).
Table 4. The effect of HBOT on left ventricular end-systolic volume (LVESV)
| Study | HBOT | Non-HBOT | Weight | Mean difference (95%CI)* | ||
| Mean ± SD | Total | Mean ± SD | Total | |||
| Li et al.10 | 24.67 ± 5.98 | 55 | 26.3 ± 6.96 | 50 | 99.0% | –1.63 (–4.12 to 0.86) |
| Martin-Hernández et al.11 | 52.5 ± 31.4 | 13 | 47.7 ± 31.4 | 11 | 1.0% | 3.80 (–21.41 to 29.01) |
| Total | 68 | 61 | 100% | –1.58 (–4.06 to 0.90) | ||
| Heterogeneity: Chi2=0.18, df=1 (p=0.67); I2=0% | ||||||
| Test for overall effect: Z=1.25 (p=0.21) | ||||||
| *Inverse variance, fixed effect Key: CI = confidence interval; HBOT = hyperbaric oxygen therapy; SD = standard deviation |
||||||
Discussion
The final meta-analysis included three studies with a combined total of 195 patients. HBOT significantly increased LVEF in patients with CAD after revascularisation compared with non-HBOT. There were no significant changes in LVEDV and LVESV in the HBOT group and non-HBOT group.
According to recent retrospective research, LVEF is the “best indicator of late persistence of severe dysfunction” in STEMI patients.12 In light of this, our study included the change in LVEF caused by the use of HBOT. We found that HBOT significantly increased LVEF by 4.16% in patients with CAD after revascularisation compared with non-HBOT (MD=4.16, 95%CI 0.97 to 7.34, p=0.01). In the HOT-MI research, which employed a single dose of HBOT immediately following thrombolysis, this beneficial impact on LVEF was previously documented in the combination of thrombolysis and HBOT.8
Another study, likewise, discovered a considerable improvement in LVEF, but the only discernible difference was the interval between the onset of thoracic pain relief following thrombolysis and the period immediately following a single exposure to HBOT at 2 ATA (atmospheres absolute) for 30 minutes. Another study used transthoracic echocardiography (TTE) to assess the benefits of HBOT at 2 ATA for 60 minutes in a single exposure, and discovered that, during the third week of the HBOT, the indexed LVESV was lower and the LVEF was considerably higher compared with the control group.13 Reductions in endothelial leukocyte adhesion and an increase in angiogenesis, two of HBOT’s positive side effects, are additional mechanisms that help reduce the size of infarctions and increase the LVEF.14 During the follow-up period, the parameters of LVEDV and LVESV were displayed with no discernible group differences. Because of the brief duration of the disease, the ventricular systolic function may not have been significantly affected.
Conclusion
In conclusion, this systematic review and meta-analysis represents a contemporary evaluation of best available evidence on the effect of HBOT on left ventricular function in patients with CAD after reperfusion. Our analysis demonstrates that HBOT significantly increased LVEF in patients with CAD after revascularisation compared with non-HBOT. There were no significant changes in LVEDV and LVESV in the HBOT group and non-HBOT group. Based on the results of our meta-analysis, further research is needed, especially RCTs or systematic review and meta-analysis that includes RCTs, to determine whether HBOT should be used routinely in patients with CAD.
Key messages
- Hyperbaric oxygen therapy (HBOT) significantly increased left ventricular ejection fraction (LVEF) by 4.16% in patients with coronary artery disease (CAD) after revascularisation compared with non-HBOT
- No statistical significance was observed in the HBOT versus non-HBOT comparison on left ventricular end-diastolic volume (LVEDV) and left ventricular end-systolic volume (LVESV)
- Reductions in endothelial leukocyte adhesion and an increase in angiogenesis, two of HBOT’s positive side effects, are additional mechanisms that may help reduce the size of infarctions and increase the LVEF
Conflicts of interest
None declared.
Funding
None.
References
1. Roth GA, Forouzanfar MH, Moran AE et al. Demographic and epidemiologic drivers of global cardiovascular mortality. N Engl J Med 2015;372:1333–41. https://doi.org/10.1056/NEJMoa1406656
2. GBD 2013 Mortality and Causes of Death Collaborators. Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet 2015;385:117–71. https://doi.org/10.1016/S0140-6736(14)61682-2
3. Crea F, Liuzzo G. Pathogenesis of acute coronary syndromes. J Am Coll Cardiol 2013;61:1–11. https://doi.org/10.1016/j.jacc.2012.07.064
4. Ibanez B, James S, Agewall S et al. 2017 ESC guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: the Task Force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC). Eur Heart J 2018;39:119–77. https://doi.org/10.1093/eurheartj/ehx393
5. Dekleva M, Neskovic A, Vlahovic A, Putnikovic B, Beleslin B, Ostojic M. Adjunctive effect of hyperbaric oxygen treatment after thrombolysis on left ventricular function in patients with acute myocardial infarction. Am Heart J 2004;148:E14. https://doi.org/10.1016/j.ahj.2004.03.031
6. Dotsenko EA, Nikulina NV, Salivonchik DP, Lappo OG, Gritsuk AI, Bastron AS. Low doses of hyperbaric oxygenation effectively decrease the size of necrotic zone in rats with experimental myocardial infarction. Bull Exp Biol Med 2015;158:732–4. https://doi.org/10.1007/s10517-015-2849-1
7. Han C, Lin L, Zhang W et al. Hyperbaric oxygen preconditioning alleviates myocardial ischemic injury in rats. Exp Biol Med (Maywood) 2008;233:1448–53. https://doi.org/10.3181/0801-RM-8
8. Shandling AH, Ellestad MH, Hart GB et al. Hyperbaric oxygen and thrombolysis in myocardial infarction: the ‘HOT MI’ pilot study. Am Heart J 1997;134:544–50. https://doi.org/10.1016/S0002-8703(97)70093-0
9. Dotsenko E, Salivonchyk D, Welcome OM et al. Influence of hyperbaric oxygenation treatment (HBOT) on clinical outcomes (recurrent myocardial infarction and survival rate) during five-year monitoring period after acute myocardial infarction. Health 2014;6:51–6. https://doi.org/10.4236/health.2014.61008
10. Li Y, Hao Y, Wang T et al. The effect of hyperbaric oxygen therapy on myocardial perfusion after the implantation of drug-eluting stents. Ann Clin Lab Sci 2018;48:158–63. Available from: http://www.annclinlabsci.org/content/48/2/158.long
11. Martín-Hernández P, Gutiérrez-Leonard H, Quintana AR et al. Hyperbaric oxygen therapy following percutaneous coronary intervention for ST-segment elevation myocardial infarction. Cardiovasc Revasc Med 2021;27:14–19. https://doi.org/10.1016/j.carrev.2020.04.031
12. Ojha N, Roy S, Radtke J et al. Characterization of the structural and functional changes in the myocardium following focal ischemia-reperfusion injury. Am J Physiol Heart Circ Physiol 2008;294:H2435–H2443. https://doi.org/10.1152/ajpheart.01190.2007
13. Stavitsky Y, Shandling AH, Ellestad MH et al. Hyperbaric oxygen and thrombolysis in myocardial infarction: the ‘HOT MI’ randomized multicenter study. Cardiology 1998;90:131–6. https://doi.org/10.1159/000006832
14. Stolfo D, Cinquetti M, Merlo M et al. ST-elevation myocardial infarction with reduced left ventricular ejection fraction: insights into persisting left ventricular dysfunction. A pPCI-registry analysis. Int J Cardiol 2016;215:340–5. https://doi.org/10.1016/j.ijcard.2016.04.097
