A 32-year-old man presented to the emergency department with shortness of breath and altered mental status. He reported a two-day history of epigastric pain, nausea, and vomiting. His past medical and family history were unremarkable. He was haemodynamically unstable, and his initial electrocardiogram (ECG) revealed a Brugada type 1 ECG pattern. The initial diagnostic assessment revealed significant metabolic derangements consistent with diabetic ketoacidosis, accompanied by hyperkalaemia. Notably, the prompt and effective management of hyperkalaemia resolved the Brugada type 1 ECG pattern, confirming the diagnosis of Brugada phenocopy.
Introduction
Brugada syndrome (BrS) is a rare genetic arrhythmogenic disorder that typically presents with distinctive electrocardiographic (ECG) findings, which can lead to sudden cardiac death in patients without structural cardiac defects.1,2 The Brugada type 1 ECG pattern is a rare finding in severe hyperkalaemia, representing a phenomenon known as Brugada phenocopy (BrP). This phenomenon occurs in the absence of congenital abnormality and typically resolves with the appropriate management of the underlying aetiology.3
Case presentation
A 32-year-old man presented to the emergency department with shortness of breath and altered mental status. He reported a two-day history of epigastric pain, nausea, and vomiting. His past medical history was unremarkable, and he denied any history of palpitation or syncope in the past. His family history was negative for congenital heart disease, coronary artery disease, or sudden cardiac death. In addition, the patient denied any history of alcohol or substance addictions.
On examination the patient’s consciousness level fluctuated, and he demonstrated Kussmaul respiration. He was hypothermic with dry mucous membranes. The patient’s blood pressure was measured at 80/50 mmHg, with a regular pulse rate of 113 beats per minute. Cardiac auscultation revealed normal heart sounds, with no evidence of murmurs being identified. The patient’s chest was clear to auscultation bilaterally with no clinical findings suggestive of volume overload. Moreover, his abdomen was soft and tender with diminished bowel sounds.
An initial blood glucose concentration was 234 mg/dL, along with an arterial blood gas analysis that demonstrated a pH of 6.87, a bicarbonate level of 7.0 mmol/L, and a potassium concentration of 9.0 mmol/L. The subsequent laboratory results were as follows: white blood cell count (WBC) 11,300 per mm³, haemoglobin 17.0 g/dL, blood urea 40 mg/dL, creatinine 2 mg/dL, sodium 135 mEq/L, and chloride 101 mEq/L. In addition, his inflammatory biomarkers, liver function test, coagulation profile, lipid profile, and cardiac enzymes were normal. The 12-lead ECG revealed features consistent with the Brugada type 1 ECG pattern, as demonstrated in figure 1. Furthermore, the patient’s chest X-ray was unremarkable, and his bedside echocardiogram revealed normal cardiac function, with no evidence of structural cardiac defects.
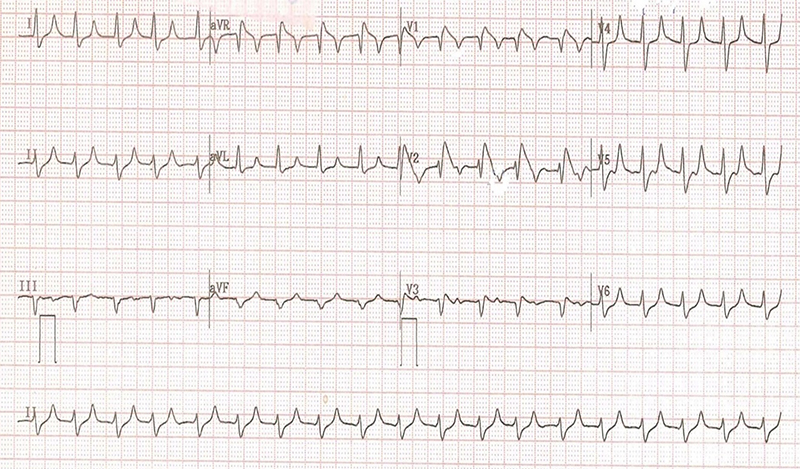
A diagnosis of diabetic ketoacidosis (DKA), hyperkalaemia, and Brugada type 1 ECG pattern was made based on the evidence of clinical findings, laboratory, and ECG results. Therefore, the patient was promptly initiated on the appropriate management protocols for hyperkalaemia and DKA, in accordance with current clinical guidelines. The management commenced with intravenous fluid resuscitation, an initial dose of 30 ml of 10% calcium gluconate, a further dose of calcium gluconate, and the initiation of an insulin infusion at a rate of 0.1 units/kg/hour.
Notably, normalisation of the potassium level to 4.7 mmol/L resulted in the resolution of the Brugada type 1 ECG pattern, as illustrated in figure 2.
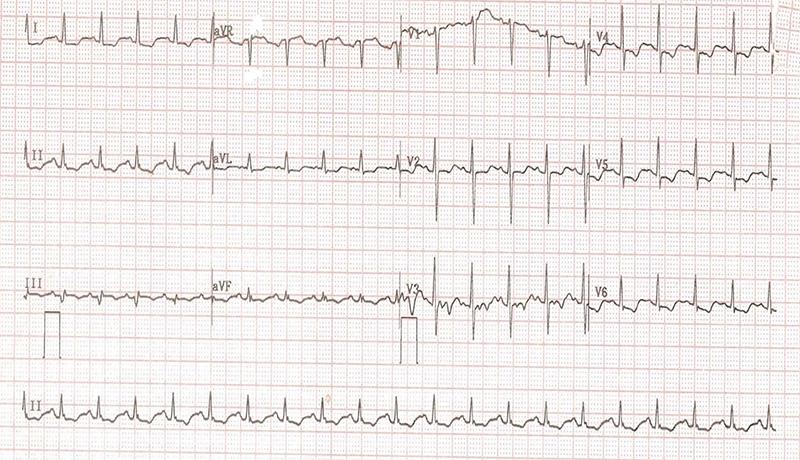
Discussion
BrS is a rare genetic sodium ion channelopathy that typically presents with distinctive ECG findings that lead to malignant ventricular tachycardia (VT) and sudden cardiac death.1 The distinctive ECG findings in BrS are classified into three types, all of which are observed in two or more precordial leads (V1–V3).4,5 Type 1 is characterised by coved-type ST-segment elevation of ≥2 mm, followed by a down-sloping concave ST-segment and symmetrical T-wave inversions. Type 2 is defined by saddle-shaped ST-segment elevation of ≥2 mm, while type 3 features either coved-type or saddle-shaped ST-segment elevation, with an elevation of less than 2 mm.4,5 The diagnosis of BrS requires the presence of distinctive ECG findings in conjunction with at least one clinical criterion.6 The clinical criteria include documented episodes of polymorphic VT or ventricular fibrillation (VF), inducible VT observed during an electrophysiological study, syncope, a family history of sudden cardiac death at <45 years old, or nocturnal agonal respiration.6
Table 1. Dose–response relationship between serum potassium concentration and electrocardiographic (ECG) abnormalities in hyperkalaemia
| Serum potassium level | ECG abnormalities |
| 5.5–6.5 mmol/L | Tall, peaked T-waves with narrow bases, predominantly observed in precordial leads |
| 6.5–8.0 mmol/L | Peaked T-waves Prolongation of the PR interval Decreased amplitude of P-waves Widening of QRS complex |
| >8.0 mmol/L | Absence of P-wave Intraventricular blocks Fascicular blocks Bundle-branch blocks Progressive widening of the QRS with resultant bizarre QRS morphology Ventricular escape rhythm Development of a sine-wave pattern Ventricular fibrillation Asystole |
BrP are clinical entities that mimic the ECG patterns of true BrS but are triggered by various clinical circumstances.7-11 Hyperkalaemia is a prevalent and potentially life-threatening electrolyte disorder, commonly encountered in the hospital setting.12 As demonstrated in table 1, elevated serum potassium levels are associated with progressive ECG abnormalities, ranging from subtle alterations to life-threatening arrhythmias, a sine-wave pattern, and ultimately asystole.13 However, this relationship may not always be observed.14 In this case, the pathophysiology of BrP in the context of hyperkalaemia is hypothesised to involve a reduction in the resting membrane potential, inactivation of sodium channels, and exaggerated potassium efflux within the right ventricular outflow tract.15 These electrophysiological alterations generate a transmural voltage gradient, leading to the development of Brugada-like ECG patterns.15 The definitive treatment for BrS often requires the placement of an implantable cardioverter-defibrillator (ICD) to mitigate the risk of life-threatening arrhythmias.16,17 In contrast, the ECG abnormalities in BrP are transient and resolve entirely with appropriate management of the underlying aetiology, highlighting their reversible and non-genetic nature.18
The diagnosis of BrP in this case is confirmed by the resolution of the Brugada type 1 ECG pattern following the correction of hyperkalaemia, alongside the absence of clinical criteria for BrS.
Conclusion
This case illustrates the reversible and transient nature of the BrP in the context of hyperkalaemia, emphasising the importance of recognising and differentiating it from true BrS. It also underscores the critical need to manage metabolic disturbances in order to reverse potentially life-threatening ECG changes and prevent unwarranted interventions.
Conflicts of interest
None declared.
Funding
None.
Patient consent
Ethical approval and informed consent for the publication of this case report were obtained. All identifying information has been anonymised to ensure patient privacy and confidentiality.
References
1. Brugada P, Brugada J. Right bundle branch block, persistent ST segment elevation and sudden cardiac death: a distinct clinical and electrocardiographic syndrome: a multicenter report. J Am Coll Cardiol 1992;20:1391–6. https://doi.org/10.1016/0735-1097(92)90253-J
2. Antzelevitch C, Brugada P, Borggrefe M et al. Brugada syndrome: report of the second consensus conference: endorsed by the Heart Rhythm Society and the European Heart Rhythm Association. Circulation 2005;111:659–70. https://doi.org/10.1161/01.CIR.0000152479.54298.51
3. Baranchuk A, Nguyen T, Ryu MH et al. Brugada phenocopy: new terminology and proposed classification. Ann Noninvasive Electrocardiol 2012;17:299–314. https://doi.org/10.1111/j.1542-474X.2012.00525.x
4. Wilde AA, Antzelevitch C, Borggrefe M et al. Proposed diagnostic criteria for the Brugada syndrome: consensus report. Circulation 2002;106:2514–19. https://doi.org/10.1161/01.CIR.0000034169.45752.4A
5. de Luna AB, Brugada J, Baranchuk A et al. Current electrocardiographic criteria for diagnosis of Brugada pattern: a consensus report. J Electrocardiol 2012;45:433–42. https://doi.org/10.1016/j.jelectrocard.2012.06.004
6. Priori SG, Wilde AA, Horie M et al. HRS/EHRA/APHRS expert consensus statement on the diagnosis and management of patients with inherited primary arrhythmia syndromes. J Arrhythm 2014;30:1–28. https://doi.org/10.1016/j.joa.2013.07.002
7. Sharon M, Wilson B, End B, Kraft C, Minardi J. Anterior ST-elevation in a patient with chest pain and fever. Ann Emerg Med 2019;74:782–5. https://doi.org/10.1016/j.annemergmed.2019.04.032
8. Yap YG, Behr ER, Camm AJ. Drug-induced Brugada syndrome. Europace 2009;11:989–94. https://doi.org/10.1093/europace/eup114
9. Hunuk A, Hunuk B, Kusken O, Onur OE. Brugada phenocopy induced by electrolyte disorder: a transient electrocardiographic sign. Ann Noninvasive Electrocardiol 2016;21:429–32. https://doi.org/10.1111/anec.12350
10. Anselm DD, Barbosa-Barros R, de Sousa Belém L, Nogueira de Macedo R, Pérez-Riera AR, Baranchuk A. Brugada phenocopy induced by acute inferior ST-segment elevation myocardial infarction with right ventricular involvement. Inn Card Rhythm Manag 2013;4:1092–4. https://doi.org/10.19102/icrm.2013.040108
11. Goldgran-Toledano D, Sideris G, Kevorkian J-P. Overdose of cyclic antidepressants and the Brugada syndrome. N Engl J Med 2002;346:1591–2. https://doi.org/10.1056/NEJM200205163462020
12. Mattu A, Brady WJ, Robinson DA. Electrocardiographic manifestations of hyperkalemia. Am J Emerg Med 2000;18:721–9. https://doi.org/10.1053/ajem.2000.7344
13. Fauci AS, Braunwald E, Kasper DL et al. Harrison’s Principles of Internal Medicine. New York: McGraw-Hill, Health Professions Division, 2008; pp. 2754.
14. Montague BT, Ouellette JR, Buller GK. Retrospective review of the frequency of ECG changes in hyperkalemia. Clin J Am Soc Nephrol 2008;3:324–30. https://doi.org/10.2215/CJN.04611007
15. Rivera‐Juárez A, Hernández‐Romero I, Puertas C et al. Clinical characteristics and electrophysiological mechanisms underlying Brugada ECG in patients with severe hyperkalemia. J Am Heart Assoc 2019;8:e010115. https://doi.org/10.1161/JAHA.118.010115
16. Sacher F, Probst V, Iesaka Y et al. Outcome after implantation of a cardioverter-defibrillator in patients with Brugada syndrome: a multicenter study. Circulation 2006;114:2317–24. https://doi.org/10.1161/CIRCULATIONAHA.106.628537
17. Sarkozy A, Boussy T, Kourgiannides G et al. Long-term follow-up of primary prophylactic implantable cardioverter-defibrillator therapy in Brugada syndrome. Eur Heart J 2007;28:334–44. https://doi.org/10.1093/eurheartj/ehl450
18. Sarquella-Brugada G, Campuzano O, Arbelo E, Brugada J, Brugada R. Brugada syndrome: clinical and genetic findings. Genet Med 2016;18:3–12. https://doi.org/10.1038/gim.2015.35
