Historically, young people with genetic heart diseases were discouraged from active sport due to concerns about the increased risk of sudden cardiac death during competitive or intensive exercise. The shock resulting from the sudden death of a young athlete, an event often highly publicised, tends to generate concern in the general population, and fear of litigation in a low-evidence area: both influence decision-making by the medical profession when discussing ‘restrictions’, especially in patients with genetic heart diseases, who by definition, are at increased risk of sudden cardiac death. In recent years, however, we have moved to a point where many athletes with certain genetic heart diseases can, with optimal medical therapy, be considered for involvement in various sporting and athletic pursuits. We are cautiously moving away from the assumption that exercise is contraindicated; we are factoring in the wishes of the patient-athlete (shared decision-making), and we are encouraging optimal protection for these athletes during their sporting endeavours (easily available automated external defibrillators [AEDs], and club personnel trained in basic life support [BLS]), while ensuring regular medical assessment to identify alterations in risk status. With dedicated follow-up of all such patient-athletes, we can refine our understanding of how best to advise (and protect) them in terms of exercise for enhanced quality of life.
Introduction
Physical activity and sports play a pivotal role in maintaining overall health and well-being, and as societies become increasingly sedentary, with an epidemic of obesity1,2 and type 2 diabetes3 in the western world, the medical profession seeks to promote meaningful exercise during daily life to maintain physical and mental health. The World Health Organisation (WHO) recommends that all adults engage in a minimum of 150 minutes of moderate intensity exercise, or 75 minutes of vigorous exercise, per week.4 For many years, the causes of sudden death in young people have been under scrutiny, with those events particularly highlighted when occurring during active exercise or competitive sports.
In recent decades, researchers have significantly increased their knowledge of the causes that predominantly contribute to sudden cardiac death (SCD) in young people, and have identified various conditions that are responsible for such outcomes, and which have already been discussed extensively in the first three BJC articles relating to sports cardiology. In the first, Westaby and Sheppard comprehensively detail the epidemiology and aetiology of SCD in athletes.5 In the second, Petrone and co-authors review the differential diagnosis between ‘athletes’ heart’ and cardiomyopathies,6 and, most recently, where Ramesh and Dhutia discuss the benefits and potential challenges of cardiac screening in athletes.7
Physicians have striven to identify those at risk of suffering acute cardiac events during sporting activities, and because of the established risks in subjects with genetic heart disease, and coronary anomalies, the (safe) tendency, historically, has been to advise against competitive sport or significant physical exertion. However, in some cases, this ‘safe’ approach may not be what the patient-athlete wants, and may not be supported by available evidence, and, furthermore, may lead to aggravation of mental health issues, in addition to predisposing to the potential complications of an enforced sedentary lifestyle.
Much has been done to find a balance between the risks and benefits of sporting activities in those with cardiovascular abnormalities. In this review, we summarise currently available recommendations and information to guide specialist cardiologists when advising young people who have been diagnosed with cardiac diseases, so that they may engage in healthy physical and sporting activities, while considering their individual cardiac abnormalities and potential risks.
“When advising such individuals (predominantly asymptomatic young patients with cardiomyopathies who aspire to exercise) it is essential to strike a balance between protecting patients from the potentially adverse effects of exercise, and depriving them of the multiple beneficial effects of exercise”.9
Maron, in assessing eligibility and disqualification recommendations for competitive athletes with cardiovascular abnormalities,8 states: “it is our goal that the recommendations in this document, together with sound clinical judgement, will lead to a healthier, safer, playing field for young competitive athletes.” Ten years on, sound clinical judgement remains key in this difficult area.
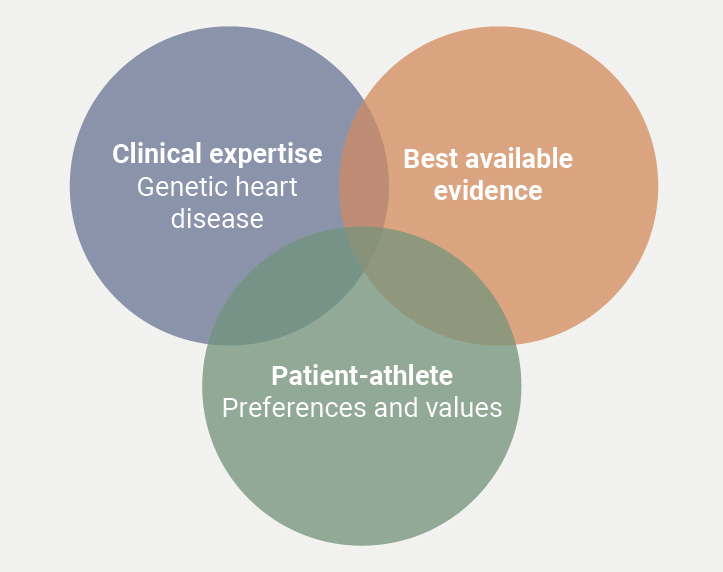
We present a brief two-part overview of some of the more common genetic heart diseases and their implications for exercise and sports participation. Much has been published recently on sport in cardiovascular disease, with significant statements and guidelines published by the European Society of Cardiology (ESC),9 Heart Rhythm Society (HRS),10 and the American Heart Association/American College of Cardiology (AHA/ACC),11 in the past five years, acting as useful reference sources when advising the athlete with cardiovascular disease on the risks and benefits of their chosen sporting activity. We examine the concept of shared decision-making (figure 1), discuss who should be involved, and how best to protect the athlete during their sporting activities.
What do we mean by low-, moderate- and high-intensity exercise?
As one might imagine, there is a considerable degree of subjectivity when discussing different levels of exercise. Table 1 is a reasonable guide to what is understood by the different intensities of exercise. The 2020 ESC guidelines on sports cardiology and exercise in patients with cardiovascular disease provide some useful objective measurements when assessing cardiovascular fitness and performance.9
Table 1. A guide to what is understood by the different intensities of exercise
| Intensity | RPE (1–10) | RPE | %HR max* | Talk test | Perception |
| Low | 0–1 | Resting | <55% | Can talk and sing | No noticeable change in breathing or sweating |
| 2–4 | Very light to light | ||||
| Moderate | 5–6 | Somewhat moderate to moderate | 55–74% | Can talk but not sing | Increased breathing and sweating |
| High | 7–8 | Somewhat hard to very hard | 75–90% | Cannot talk or sing | Feeling out of breath and increased sweating |
| 9–10 | Extremely hard to maximal exertion | ||||
| * %HR max will not be an accurate measure of exercise intensity if your heart rate is influenced by certain medications or conditions. Key: HR max = maximum heart rate; RPE = rating of perceived exertion |
|||||
The changing landscape of advice and management
Hypertrophic cardiomyopathy
Hypertrophic cardiomyopathy (HCM) is the most common genetic cardiomyopathy (prevalence ≥1:500 in the population), and a recognised cause of sudden death in previously undiagnosed young people, including athletes. Inactivity is prevalent among many with HCM.12 Until relatively recently, it was considered that young people with HCM should not participate in vigorous activities, nor should they be allowed to participate in sports. By 2020, the AHA/ACC guidelines recognised the benefits of mild-to-moderate intensity recreational exercise, resulting from the RESET-HCM (Randomized Exploratory Study of Exercise Training in Hypertrophic Cardiomyopathy) trial findings, where those prescribed moderate-to-intense exercise showed significant improvement in capacity and physical functioning, with no major adverse events, however, the study was not powered for safety, and follow-up was 16 weeks long.13 Weisslar-Snir and colleagues14 reported that the incidence of HCM-related SCD in young people in Ontario was substantially less than previously reported, occurring predominantly in previously undiagnosed subjects, and mostly occurred during rest or light activity (83.3%). Lampert et al.,15 in 2023, reported a prospective multi-centre international study looking at the effects of vigorous exercise in patients with HCM. Participants (1,660 subjects followed for three to seven years) were categorised according to self-reported levels of physical activity. They found that, in individuals with clinical HCM or genotype-positive/phenotype-negative HCM who are treated in experienced centres, those exercising vigorously did not experience a higher rate of death or life-threatening arrhythmias than those exercising moderately, or those who were sedentary. The authors observed that one-third of the enrolled subjects had previously been advised to perform no exercise or only light-intensity activities. Of the deaths recorded during follow-up, four occurred during sleep or when home alone: in two the circumstances were unknown, and two deaths were while hunting and driving a car, respectively. Those successfully resuscitated were at school, playing sport (volleyball, karate), running and cycling recreationally, respectively. The overall message appears to indicate that, even with cardiac arrest, the availability of resuscitation personnel and equipment (automated external defibrillator – AED) can markedly alter the outcome, and this should form a very important part of any guidance that specialist cardiologists might give to those who wish to pursue athletic or competitive activities. The subgroup of vigorous competitive athletes were younger, more likely to be genotype-positive/phenotype-negative, more likely to have apical HCM, less likely to have an implanted cardioverter-defibrillator (ICD), and it is likely that they were predominantly asymptomatic, thus, to a certain extent, self-selecting themselves.
Kim et al., in the 2025 scientific statement from the AHA/ACC on clinical considerations for competitive sports participation for athletes with cardiovascular abnormalities,11 refer to the lack of substantial breakthrough cardiac events in athletes with HCM, and retrospective studies demonstrating little difference between athletes with HCM who continued participating in competitive sport, and those who retired for medical reasons. Gray et al. suggest, however, that, while exercise may not significantly increase the risk of sudden cardiac arrest (SCA) for some cardiovascular diseases, such as low-risk HCM, on optimal therapy, these individuals still carry a risk of SCA, which may occur during exercise, and that this should be part of any ‘return to play’ discussion.16 Martinez et al. had previously highlighted that, included in an athletes shared decision-making/return to play counselling session is a transparent discussion that no evaluation process is perfect and that there is no such thing as ‘zero-risk’.17 As a ‘cautionary tale’, Malhotra et al., in their outcomes of cardiac screening in adolescent soccer players, reported that five of the 11,168 elite players screened were diagnosed with HCM, and advised to stop playing. Two continued to compete despite medical advice, and subsequently died during intensive exercise.18
Arrhythmogenic right ventricular cardiomyopathy
Arrhythmogenic right ventricular cardiomyopathy (ARVC) is reported as one of the most common causes of SCD in athletes and young people,19 and has a prevalence of 1:1,000–1:5,000 in the population. Historically, it was believed that it was a right ventricular cardiomyopathy, but we now know it involves both ventricles, and even the left ventricle in isolation in some cases. Diagnosis and treatment have previously been discussed, but ARVC is a particularly complex disease when discussing sporting activity, as there is evidence of increased risk of SCD, and also increased disease acceleration with exercise.20 So while there is a paucity of evidence for increased risk of SCA when competing in sport among genotype-positive/phenotype-negative athletes, phenotypic conversion can be accelerated by that exercise in plakophilin-2 (PKP2)-mediated ARVC20,21 (it is not established whether the same applies for other genotypes or gene-elusive ARVC). After initial studies suggested an association between competitive sport and increased incidence of SCD compared with sedentary individuals, it was reported that athletes were more likely to have impaired biventricular function and earlier onset of ventricular arrhythmias, compared with sedentary patients,22 and ventricular arrhythmias were more common in patients who had previously engaged in high-intensity exercise.23 People with manifestations of ARVC at a young age had frequently participated in endurance sports previously,24 and this is important because paediatric onset is more often associated with SCD or SCA. It has been hypothesised that the ARVC phenotype may be caused by intense exercise in individuals who are gene elusive.25,26 Overall, the message is one of caution with very close surveillance to detect early phenotypic conversion. Dei et al.27 highlight that, as exercise prescription in those with cardiomyopathy is challenging, decision-making for patients with ARVC should be conducted within specialised centres, with a multi-disciplinary approach, including exercise physiologists, and with shared decision-making including patient preferences for type of sport, but considering the many knowledge gaps in the area. Minimal exercise levels appear to be safe for unaffected PKP2 mutation carriers according to the AHA recommendations, with adherence associated with a lower risk of phenotype progression and ventricular arrhythmias.28 Strenuous sports should be avoided in children with ARVC, but participation in non-competitive school activities may be considered. “Although it appears reasonable not to deny patients with ACM the numerous health benefits associated with low-moderate intensity exercise, the evidence on the safety and possible effects on the progression of the disease is lacking”.27 It has been recommended that mild-to-moderate physical activity for up to 150 minutes per week is considered safe, and is recommended in able phenotype-negative individuals.9
Cardiac channelopathies
Unequivocally, everyone with a known cardiac channelopathy should be followed by a specialist with expertise in genetic heart disease. The AHA/ACC scientific statement provides a summary of the general clinical considerations for competitive athletes with cardiac channelopathies.11 They report recent data demonstrating low rates of breakthrough cardiac events in competitive athletes with cardiac channelopathies after initiation of appropriate management plans. Tobert and colleagues,29 in reviewing their 20-year experience of athletes with long QT syndrome (LQTS), many of whom were previously disqualified, and who were subsequently assessed at the Mayo Clinic, and returned to play, reported no LQTS sports-associated mortality (among 494 athletes) over the 20 years. One of the criteria for return-to-play, after informing relevant coaches and administrators, was to obtain a personal AED as part of the ‘sports bag’, a concept that may become more accepted in the future: also sensible advice on ensuring proper hydration, electrolyte replenishment, avoidance of QT-prolonging drugs, and annual follow-up assessment. The authors emphasise that primary-care physicians and general cardiologists should ensure that the patient-athletes are evaluated in a genetic heart disease specialty centre, where risk stratification, tailoring of therapy, and return-to-play approval should be undertaken. Martinez and colleagues17 reported on elite patient-athletes (collegiate, Olympic, professional) who were followed for a mean of seven years after return to play, and where over 70% were initially disqualified from sport after being diagnosed with genetic heart disease (HCM 53%, LQTS 26%), but were subsequently assessed at one of four national specialist units. No fatalities were reported during follow-up. In all cases, it was confirmed that an AED would be readily accessible during all training and competition. In some cases, patients acquired their own AED. The authors highlight: “Although historically, athletes with genetic heart disease predisposing to SCD were restricted from playing competitive athletics, those who return to play after expert risk stratification and tailoring of therapy face a low risk of cardiac events”.
Exercise restrictions have historically been advised for those with catecholaminergic polymorphic ventricular tachycardia (CPVT), due to the adrenergic basis of the ventricular arrhythmias that are the hallmark of the condition.10 Competitive sports participation can be considered in the absence of exercise-induced ventricular ectopy or arrhythmias, or a family history of SCD, but including discussion of the use of prophylactic therapies, and regular surveillance assessment.29,30 If the exercise test is positive, a treatment programme guided by normalisation of the exercise test is recommended before considering return-to-play (RTP).29-31 In those previously symptomatic, where RTP is being considered, combination therapy with beta-blockade and flecainide, and with consideration of left cardiac sympathetic denervation (LCSD) should be introduced, in the hope of normalising the exercise test.31,32
As there are no data showing that exercise increases risk in athletes with Brugada syndrome, there is no evidence to support exercise restrictions for these individuals,10 however, those with Brugada syndrome should avoid the use of certain medications (sodium-channel blocking, a list is provided at www.brugadadrugs.org),33 avoid overheating and exercising during intercurrent illnesses;34 also heavy meals and excessive alcohol have been implicated in increased risk of arrhythmia.35 It would seem especially sensible to avoid prolonged-duration exercise in the heat, to ensure good hydration, and avoid exercising after heavy meals.
As with all patients with genetic heart disease, if subjects require escalation in therapy for whatever clinical reason, there should be clinical stability for at least three months before resumption of competitive sports participation.
Athletes who have any ‘episode’ or ‘event’ should avoid sporting activities until their clinical status is re-assessed and stabilised over a three-month period.
Key messages
- When we advise on exercise for those with genetic heart diseases, we need as much information as possible on the relative risks and benefits
- We do not want to add the long-term complications of a sedentary lifestyle, thus, we need to find the safest ‘happy medium’ in order to ensure that young people with genetic heart diseases live a long and healthy life
- Athletes with diagnosed genetic heart diseases should only be considered for ‘return to play’ after optimal treatment has been introduced, and a plan for regular surveillance is in place at a specialist cardiac unit
Conflicts of interest
None declared.
Funding
None.
Editors’ note
This is the first part of a two-part review, the second part will be published in the next issue (issue 2, 2026). This article is also part of the Sports Cardiology series.
Recommended reading
Hypertrophic cardiomyopathy
Kim JH, Baggish AL, Levine BD et al. Clinical considerations for competitive sports participation for athletes with cardiovascular abnormalities: a scientific statement from the American Heart Association and American College of Cardiology. Circulation 2025;151:e716–e761. https://doi.org/10.1161/CIR.0000000000001297
Lampert R, Ackerman MJ, Marino BS et al. Vigorous exercise in patients with hypertrophic cardiomyopathy. JAMA Cardiol 2023;8:595–605. https://doi.org/10.1001/jamacardio.2023.1042
Ommen SR, Ho CY, Asif IM et al. 2024 AHA/ACC/AMSSM/HRS/PACES/SCMR guideline for the management of hypertrophic cardiomyopathy:a report of the American Heart Association/American College of Cardiology joint committee on clinical practice guidelines. Circulation 2024;149:e1239–e1311. https://doi.org/10.1161/CIR.0000000000001250
Arrhythmogenic right ventricular cardiomyopathy
Kim JH, Baggish AL, Levine BD et al. Clinical considerations for competitive sports participation for athletes with cardiovascular abnormalities: a scientific statement from the American Heart Association and American College of Cardiology. Circulation 2025;151:e716–e761. https://doi.org/10.1161/CIR.0000000000001297
Lupo Dei L, Han J, Romano S et al. Exercise prescription in arrhythmogenic cardiomyopathy: finding the right balance between risks and benefits. J Am Heart Assoc 2025;14:e039125. https://doi.org/10.1161/JAHA.124.039125
References
1. Holmstrom L, Junttila J, Chugh SS. Sudden death in obesity: mechanisms and management. J Am Coll Cardiol 2024;84:2308–24. https://doi.org/10.1016/j.jacc.2024.09.016
2. NCD Risk Factor Collaboration. Worldwide trends in underweight and obesity from 1990 to 2022: a pooled analysis of 3663 population-representative studies with 222 million children, adolescents, and adults. Lancet 2024;403:1027–50. https://doi.org/10.1016/S0140-6736(23)02750-2
3. Khan MAB, Hashim MJ, King JK, Govender RV, Mustafa H, Kaabi JL. Epidemiology of type 2 diabetes – global burden of disease and forecasted trends. J Epidemiol Glob Health 2020;10:107–11. https://doi.org/10.2991/jegh.k.191028.001
4. World Health Organisation. WHO guidelines on physical activity and sedentary behavior. Geneva: WHO, 2020. Available from: https://www.who.int/publications/i/item/9789240015128
5. Westaby J, Shephard MN. Epidemiology and aetiology of sudden cardiac death in athletes. Br J Cardiol 2025;32:49–52. https://doi.org/10.5837/bjc.2025.019
6. Petrone A, Minopoli TC, Papadakis M, Sharma S, Finocchiaro G. Differential diagnosis between ‘athlete’s heart’ and cardiomyopathies. Br J Cardiol 2025;32:91–9. https://doi.org/10.5837/bjc.2025.030
7. Ramesh P, Dhutia H. Cardiac screening in athletes: benefits and potential challenges. Br J Cardiol 2025;32:130–4. https://doi.org/10.5837/bjc.2025.048
8. Maron BJ, Zipes DP, Kovaks RJ. Eligibility and disqualification recommendations for competitive athletes with cardiovascular abnormalities: preamble, principles, and general considerations. A scientific statement from the American Heart Association and American College of Cardiology. Circulation 2015;132:e256–e261. https://doi.org/10.1161/CIR.0000000000000236
9. Pelliccia A, Sharma S, Gahi S et al. 2020 ESC guidelines on sports cardiology and exercise in patients with cardiovascular disease: the task force on sports cardiology and exercise in patients with cardiovascular disease of the European Society of Cardiology (ESC). Eur Heart J 2021;42:17–96. https://doi.org/10.1093/eurheartj/ehaa605
10. Lampert R, Chung EH, Ackerman MJ et al. 2024 HRS expert consensus statement on arrhythmias in the athlete: evaluation, treatment, and return to play. Heart Rhythm 2024;21:e151–e252. https://doi.org/10.1016/j.hrthm.2024.05.018
11. Kim JH, Baggish AL, Levine BD et al. Clinical considerations for competitive sports participation for athletes with cardiovascular abnormalities: a scientific statement from the American Heart Association and American College of Cardiology. Circulation 2025;151:e716–e761. https://doi.org/10.1161/CIR.0000000000001297
12. Sweeting J, Ingles J, Timperio A et al. Physical activity in hypertrophic cardiomyopathy: prevalence of inactivity and perceived barriers. Open Heart 2016;3:e000484. https://doi.org/10.1136/openhrt-2016-000484
13. Saberi S, Wheeler M, Bragg-Gresham J et al. Effect of moderate-intensity exercise training on peak oxygen consumption in patients with hypertrophic cardiomyopathy: a randomized clinical trial. JAMA 2017;317:1349–57. https://doi.org/10.1001/jama.2017.2503
14. Weissler-Snir A, Allen K, Cunningham K et al. Hypertrophic cardiomyopathy-related sudden cardiac death in young people in Ontario. Circulation 2019;140:1706–16. https://doi.org/10.1161/CIRCULATIONAHA.119.040271
15. Lampert R, Ackerman MJ, Marino BS et al. Vigorous exercise in patients with hypertrophic cardiomyopathy. JAMA Cardiol 2023;8:595–605. https://doi.org/10.1001/jamacardio.2023.1042
16. Gray B, Lampert R, Papadakis M. Return to play with genetic heart disease: the importance of developing a personalized emergency action plan. Circulation 2025;151:893–5. https://doi.org/10.1161/CIRCULATIONAHA.124.072830
17. Martinez KA, Bos JM, Baggish AL et al. Return-to-play for elite athletes with genetic heart diseases predisposing to sudden cardiac death. J Am Coll Cardiol 2023;82:661–70. https://doi.org/10.1016/j.jacc.2023.05.059
18. Malhotra A, Dhutia H, Finocchiaro G et al. Outcome of cardiac screening in adolescent soccer players. N Engl J Med 2018;379:524–34. https://doi.org/10.1056/NEJMoa1714719
19. Finocchiaro G, Westaby J, Sheppard MN, Papadakis M, Sharma S. Sudden cardiac death in young athletes: JACC state-of-the-art review. J Am Coll Cardiol 2024;83:350–70. https://doi.org/10.1016/j.jacc.2023.10.032
20. James CA, Bhonsale A, Tichnell C et al. Exercise increases age-related penetrance and arrhythmic risk in arrhythmogenic right ventricular dysplasia/cardiomyopathy-associated desmosomal mutation carriers. J Am Coll Cardiol 2013;62:1290–7. https://doi.org/10.1016/j.jacc.2013.06.033
21. Lie OH, Rootweld-NorbergC, Dejgaard LA et al. Prediction of life-threatening ventricular arrhythmia in patients with arrhythmogenic cardiomyopathy: a primary prevention cohort study. JACC Cardiovasc Imaging 2018;11:1377–86. https://doi.org/10.1016/j.jcmg.2018.05.017
22. Saberniak J, Hasselberg NE, Borgquist R et al. Vigorous physical activity impairs myocardial function in patients with arrhythmogenic right ventricular cardiomyopathy and in mutation positive family members. Eur J Heart Fail 2014;16:1337–44. https://doi.org/10.1002/ejhf.181
23. Lie OH, Dejgaard LA, Saberniak J et al. Harmful effects of exercise intensity and exercise duration in patients with arrhythmogenic cardiomyopathy. JACC Clin Electrophysiol 2018;4:744–53. https://doi.org/10.1016/j.jacep.2018.01.010
24. Te Riele ASJM, James CA, Sawant AC et al. Arrhythmogenic right ventricular dysplasia/cardiomyopathy in the pediatric population: clinical characterization and comparison with adult-onset disease. JACC Clin Electrophysiol 2015;1:551–60. https://doi.org/10.1016/j.jacep.2015.08.004
25. La Gerche A, Robberecht C, Kuiperi C et al. Lower than expected desmosomal gene mutation prevalence in endurance athletes with complex ventricular arrhythmias of right ventricular origin. Heart 2010;96:1268–74. https://doi.org/10.1136/hrt.2009.189621
26. Sawant AC, Bhonsale A, Te Riele ASJM et al. Exercise has a disproportionate role in the pathogenesis of arrhythmogenic right ventricular dysplasia/cardiomyopathy in patients without desmosomal mutations. J Am Heart Assoc 2014;3:e001471. https://doi.org/10.1161/JAHA.114.001471
27. Lupo Dei L, Han J, Romano S et al. Exercise prescription in arrhythmogenic cardiomyopathy: finding the right balance between risks and benefits. J Am Heart Assoc 2025;14:e039125. https://doi.org/10.1161/JAHA.124.039125
28. Wang W, Tichnell C, Murray BA et al. Exercise restriction is protective for genotype positive family members of arrhythmogenic right ventricular cardiomyopathy patients. Europace 2020;22:1270–8. https://doi.org/10.1093/europace/euaa105
29. Tobert KE, Bos M, Garmany R, Ackerman MJ. Return-to-play for athletes with long QT syndrome or genetic heart diseases predisposing to sudden death. J Am Coll Cardiol 2021;78:594–604. https://doi.org/10.1016/j.jacc.2021.04.026
30. Ostby SA, Bos JM, Owen HJ, Wackel PL, Cannon BC, Ackerman MJ. Competitive sports participation in patients with catecholaminergic polymorphic ventricular tachycardia: a single center’s early experience. JACC Clin Electrophysiol 2016;2:253–62. https://doi.org/10.1016/j.jacep.2016.01.020
31. Roston TM, Kallas D, Davies B et al. Burst exercise testing can unmask arrhythmias in patients with incompletely penetrant catecholamine polymorphic ventricular tachycardia. JACC Clin Electrophysiol 2021;7:437–41. https://doi.org/10.1016/j.jacep.2021.02.013
32. Tobert KE, Bos JM, Cannon BC, Ackerman MJ. Outcomes of athletes with genetic heart diseases and implantable cardioverter-defibrillators who choose to return to play. Mayo Clin Proc 2022;97:2028–39. https://doi.org/10.1016/j.mayocp.2022.03.024
33. Postema PG, Wolpert C, Amin AS et al. Drugs and Brugada syndrome patients: review of the literature, recommendations, and an up-to-date website. Heart Rhythm 2009;6:1335–41. https://doi.org/10.1016/j.hrthm.2009.07.002
34. Rattanawong P, Vutthikraivit W, Charoensri A et al. Fever-induced Brugada syndrome is more common than previously suspected: a cross-sectional study from an endemic area. Ann Noninvasive Electrocardiol 2016;21:136–41. https://doi.org/10.1111/anec.12288
35. Nishizaki M, Sakurada H, Mizusawa Y et al. Influence of meals on variations of ST segment elevation in patients with Brugada syndrome. J Cardiovasc Electrophysiol 2008;19:62–8. https://doi.org/10.1111/j.1540-8167.2007.00972.x
