Immune checkpoint inhibitors (ICIs) have revolutionised cancer care, but can cause serious cardiac immune-related adverse events (irAEs), particularly myocarditis, which carries up to 50% mortality. This study explores patient pathways to improve early recognition and management of ICI myocarditis in acute settings.
This was a retrospective, single-centre case series from a UK tertiary hospital with a specialist cardio-oncology multi-disciplinary team (MDT). Ten patients referred to the cardio-oncology MDT between March and August 2024 were included if diagnosed with myocarditis, pericarditis, or arrhythmias linked to ICI treatment. Clinical pathways were described using structured vignettes.
Seven patients had myocarditis, two pericarditis, and one bradyarrhythmia. Fatigue (57%) was the most common presenting symptom in myocarditis. Echocardiography was unremarkable in 86% of cases, while cardiac magnetic resonance imaging (MRI) confirmed myocarditis in 57%. N-terminal pro-B-type natriuretic protein (NT-proBNP) rose 17-fold from baseline compared with a four-fold rise in troponin T. Early MDT referral facilitated prompt diagnosis and treatment, while delayed recognition was associated with worse outcomes.
This case series highlights the importance of early MDT involvement, and the utility of additional cardiac biomarkers, such as NT-proBNP. It provides practical guidance to improve the acute care pathway for patients developing ICI-associated cardiac toxicities.
Introduction
Immune checkpoint inhibitors (ICIs) have transformed cancer treatment, significantly improving survival across many tumour types. For instance, the 10-year results of the CheckMate-067 trial in metastatic melanoma reported a 43% overall survival (OS) rate, compared with under 5% before ICIs.1
ICIs function by inhibiting regulatory proteins expressed by immune cells, reducing self-tolerance and enhancing the immune system’s ability to recognise tumour cells. Most commercially available ICIs target the cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and programmed cell death protein 1 (PD-1)/programmed death-ligand 1 (PD-L1) signalling pathways. Recently, newer ICIs have been developed to target other immune checkpoint proteins, such as lymphocyte-activation gene 3 (LAG-3).1–3
A well-documented side effect of ICIs is autoimmunity, in the form of immune-related adverse events (irAEs). Common irAEs include colitis, thyroiditis, dermatitis, and pneumonitis, with rarer cases affecting neurological, hepatic, renal, and cardiovascular systems.4 As ICIs become increasingly integrated into oncological regimens for early and advanced disease, irAEs are expected to become more prevalent.
IrAEs require structured monitoring and tailored management. Patients should have baseline and periodic assessments, including labs and imaging, as indicated. In the context of cardiac irAEs, baseline assessments should include serum troponin, N-terminal pro-B-type natriuretic peptide (NT-proBNP), creatinine kinase (CK), fasting lipid profile and glycated haemoglobin (HbA1c); 12-lead ECG and transthoracic echocardiogram (TTE) at a minimum. Symptoms and abnormalities must be promptly graded to guide therapy. Management depends on severity, ranging from continuing ICIs in mild cases to suspending or stopping them in severe reactions. Corticosteroids are first line, with escalation to other immunosuppressants if needed. Organ-specific care involves relevant specialists, and ICI rechallenge may be cautiously considered after resolution, particularly for non-severe irAEs.
Cardiac irAEs, once considered rare, are now increasingly recognised. Their under-recognition in earlier studies is partly due to landmark ICI trials not prospectively screening for cardiovascular involvement, such as routine measurement of biomarkers like troponin I (TnI), troponin T (TnT), and NT-proBNP. For example, as of April 2016, safety data from Bristol Myers Squibb – a pioneer in immuno-oncology – reported myocarditis incidences of 0.06% with ICI monotherapy and 0.27% with combination therapy.5 These figures likely underestimate real-world incidence, as more recent studies estimate cardiac irAEs occur in 6–10% of patients.6,7
Myocarditis is the most fatal cardiac irAE, with a mortality rate, most recently, of 38%.8–10 Notably, the mortality risk from ICI myocarditis is nearly double that of an underlying cancer diagnosis.11 Severe sequelae include rapidly progressive heart failure or fulminant myocarditis. Other cardiac irAEs include pericarditis, conduction system defects, and accelerated coronary atherosclerosis (table 1).12,13
Table 1. Overview of immune checkpoint inhibitor cardiac toxicities and associated diagnostic findings
| Conduction | Myocardium | Pericardium | Vascular |
| Atrial or ventricular tachyarrhythmias AV blocks |
Myocarditis Heart failure (HFpEF >HFrEF) Regional myocarditis (Taktsubo-like) |
Pericarditis Pericardial effusion Cardiac tamponade |
Acute coronary syndrome Vasculitis |
| ECG | |||
| Non-specific ST–T changes, PR depression | Non-specific ST–T changes, PR depression | NSR; NS widespread concave STE/PR depression | ST–T changes possibly with reciprocal changes |
| TTE | |||
| Normal | Preserved LV function (MC), reduced LV function | Trace – large pericardial effusion, features of tamponade | Regional wall motion abnormality |
| Markers | |||
| NT-proBNP and TNT mild rise with tachyarrhythmias | NT-proBNP ↑↑ TnT ↑ |
NT-proBNP no rise TNT no rise |
NT-proBNP ↑ TnT ↑ (serial TnT ↑↑) |
| Coronary evaluation | |||
| CMR beneficial | Invasive/CT angiogram | ||
| Key: AV = atrioventricular; CMR = cardiac magnetic resonance; CT = computed tomography; ECG = electrocardiogram; HFpEF = heart failure with preserved ejection fraction; HFrEF = heart failure with reduced ejection fraction; LV = left ventricular; MC = myocardial contractility; NS = non-specific; NSR = normal sinus rhythm; NT-proBNP = N-terminal pro-B-type natriuretic peptide; STE = ST elevation; TnT = troponin T | |||
Diagnosing ICI myocarditis remains challenging due to its non-specific symptoms, such as fatigue and dyspnoea, which mimic chronic heart failure.14 Despite this, echocardiography often reveals normal or hyperdynamic left ventricular function, complicating detection.15 Limited awareness and variable presentations contribute to delayed diagnosis and management, leading to poor outcomes.
Aim
The primary aim of this retrospective, single-centre narrative case series is to evaluate diagnostic and management approaches to cardiac irAEs at our institution. The secondary objective is to provide practical insights to support clinicians in acute care settings.
Materials and method
This retrospective, single-centre study was conducted at a UK tertiary centre with specialist cardiology and oncology services, including a cardio-oncology multi-disciplinary team (MDT). Cases were identified from referrals to the cardio-oncology MDT between March and August 2024. Patients were included if they were considered to have myocarditis, pericarditis, or arrhythmias during MDT discussions.
Data collected included patient demographics, tumour type and stage, ICI regimen, treatment intent, baseline and acute cardiac biomarkers (TnT, NT-proBNP), ECG, TTE and cardiac magnetic resonance (CMR) findings, presenting symptoms, pathway intervals, including between ICI initiation to acute presentation, and between acute presentation to diagnosis, immunosuppressive treatments, and outcomes. CMR images were reviewed twice, initially at our centre’s local cardio-oncology MDT, and subsequently on a regional level. The latter confirmatory data were collected. Survival data were recorded up to November 2024.
Quantitative data were presented as counts, percentages, means, or medians. Clinical pathways for ICI myocarditis cases were qualitatively described using structured vignettes, consisting of background, investigations and clinical course.
Verbal informed consent to publish case-related details was retrospectively obtained from patients or, in the case of deceased individuals, from their next of kin. Voluntary participation and the right to decline without impact on care was emphasised. For cases 2 and 6, posthumous consent could not be obtained due to difficulties in contacting their next of kin. All cases have been maximally anonymised while retaining essential clinical details.
Table 2. Clinicopathological characteristics and relevant investigations for all 10 patients experiencing cardiac immune-related adverse events (irAEs)
| Total N=10 | |
| Male, n (%) | 6 (60) |
| Mean age (range), years | 71 (58–81) |
| Tumour site, n (%) Skin Head and neck Others* |
5 (50) 2 (20) 3 (30) |
| Cancer stage, n (%) Advanced |
10 (100) |
| Treatment intent, n (%) Palliative |
10 (100) |
| Type of ICI(s) received, n (%) Anti-CTLA-4 alone` Anti-PD-1/PD-L1 alone Combination anti-CTLA-4/PD-1 Combination anti-PD-1/LAG-3** |
3 (30) 3 (30) 3 (30) 1 (10) |
| Median number of treatment cycles prior to presentation (range) | 3 (1–7) |
| Mean time from commencing ICI to toxicity (range), days | 95 (21–204) |
| Type of cardiac irAE, n (%) Myocarditis Pericarditis Arrhythmia |
7 (70) 2 (20) 1 (10) |
| Non-cardiac irAEs, n (%) Pneumonitis Others † None |
3 (30) 2 (20) 5 (50) |
| Comorbidities, n (%) Cardiovascular ‡ Autoimmune |
7 (70) 3 (30) |
| ECG findings at presentation, n (%) Sinus rhythm New atrial fibrillation New AV block Ischaemic changes Pericarditis features |
4 (40) 2 (20) 2 (20) 1 (10) 1 (10) |
| Median delay in diagnosis (range), days | 1 (0–95) |
| Immunosuppressive therapy received for cardiac irAE, n (%) Glucocorticoids Immunosuppressants § |
9 (90) 5 (50) |
| Deaths, n (%) | 2 (20) |
| *Synchronous epithelioid mesothelioma and renal cell carcinoma (RCC) (n=1), cholangiocarcinoma (n=1), lung adenocarcinoma (n=1). **Anti-PD-1/LAG-3 combination ICI therapy recently approved within UK. †Colitis (n=1), adrenalitis (n=1), thyroiditis (n=1), myositis and myasthenia gravis (triple M) (n=1). ‡Hypertension (n=4), aortic stenosis (n=2), atrial fibrillation (n=2), type 2 diabetes mellitus (n=2), myocardial infarction (n=1), hyperlipidaemia (n=1), obesity (n=1), pulmonary hypertension (n=1). §Mycophenolate mofetil (n=4), tacrolimus (n=1). Key: AV = atrioventricular; CMR = cardiac magnetic resonance; CTLA-4 = cytotoxic T-lymphocyte-associated protein-4; ECG = electrocardiography; ICI = immune checkpoint inhibitor; irAEs = immune-related adverse events; LAG-3 = lymphocyte-activation gene 3; PD-1 = programmed cell death protein 1; PD-L1 = programmed death-ligand 1; TTE = transthoracic echocardiography |
|
Table 3. Characteristics and relevant investigations for patients identified with immune checkpoint inhibitor (ICI) myocarditis. Data from the included cases are shown as N (%) unless otherwise specified
| Total N=7 | |
| Clinical symptoms at presentation, n (%) | |
| Fatigue | 4 (57.1) |
| Dyspnoea | 2 (28.4) |
| Chest pain | 1 (14.2) |
| Lower limb oedema | 1 (14.2) |
| Fulminant myocarditis | 0 (0) |
| Abnormal TTE at presentation, n (%) | |
| Hyperdynamic LVEF (LVEF >70%) | 0 (0) |
| Preserved LVEF (LVEF 50–69%) | 6 (85.7) |
| New reduction in LVEF | 1 (14.2) |
| Any other relevant TTE findings | 0 (0) |
| CMR suggestive of myocarditis * | 4 (57.1) |
| Mean TnT at presentation (range), ng/L | 184.7 (33–966) |
| Mean ratio of presenting to baseline TnT † | 4 |
| Mean NT-proBNP at presentation (range), pg/ml | 4,664 (571–15,070) |
| Mean ratio of presenting to baseline NT-proBNP † | 17 |
| *CMR was requested for all seven patients. However, two were not performed as these patients were either not able to comply or tolerate the procedure. Four of five CMRs performed were suggestive of myocarditis, with one being non-diagnostic due to movement artefact. †Two out of seven patients did not have baseline readings of TnT and NT-proBNP. Key: CMR = cardiac magnetic resonance; LVEF = left ventricular ejection fraction; NTpro-BNP = N-terminal pro-B-type natriuretic peptide; TnT = troponin T; TTE = transthoracic echocardiography |
|
Results
There were 41 patients discussed at the cardio-oncology MDT between 1 March and 30 August 2024. Eleven patients with cardiac irAEs were identified. One patient with concomitant cardiac sarcoidosis was excluded, due to the potential confounding influence of a pre-existing cardiac autoimmune condition and immunosuppressive therapy on the diagnostic and management pathway.
Ten patients were, therefore, included in this case series (table 2). Myocarditis was the most common irAE, affecting seven patients in this case series (70%) (table 3).
Case 1
Background
A 68-year-old woman with advanced floor-of-mouth squamous cell carcinoma, receiving pembrolizumab (anti-PD-1), presented with shortness of breath and bilateral lower limb oedema 204 days after starting treatment. She had completed six cycles. Medical records revealed a prior presentation to her local hospital three months earlier with mild lower limb oedema. No cardiac biomarkers were tested at that time, and she was discharged with oral diuretics, without consulting her parent oncology team. She subsequently received one further cycle of pembrolizumab.
Investigations
On admission, TnT was 140 ng/L (upper limit of normal [ULN] <14 ng/L, baseline: not performed) and NT-proBNP was 5,745 pg/mL (ULN <400 pg/ml, baseline: not performed). ECG demonstrated new first-degree atrioventricular (AV) block. Echocardiography performed 12 days after admission was unremarkable with preserved left ventricular ejection fraction (LVEF) and normal diastolic function. CMR performed 10 days after admission was non-diagnostic due to movement artefact.
Clinical course
Given the strong clinical suspicion of ICI myocarditis, the patient was treated with intravenous methylprednisolone and oral tacrolimus, resulting in initial improvement. However, her condition deteriorated on discharge, and she passed away from heart failure two months later. It was felt that immunosuppressive therapy had only delayed the progression of chronic myocarditis.
Case 2
Background
An elderly man with advanced cholangiocarcinoma, receiving palliative durvalumab (anti-PD-L1), presented with dyspnoea and bilateral lower limb oedema approximately three months after commencing treatment. He had completed three cycles. His medical history included poorly controlled type 2 diabetes mellitus, hypertension, hypercholesterolaemia, obesity, paroxysmal atrial fibrillation, and a significant smoking history.
Investigations
On admission, TnT was 966 ng/L (ULN <14 ng/L, baseline not performed), and NT-proBNP was 15,070 pg/ml (ULN <400 pg/ml, baseline not performed). An ECG showed ST-segment elevation in leads V2–V6 with atrial fibrillation. Echocardiography revealed a LVEF of 25% (previously 55%), a severely dilated left atrium, raised filling pressures, and regional wall motion abnormalities. CMR five days later reported myocardial ischaemia in the left anterior descending (LAD) territory with underlying inflammatory oedema from myocarditis.
Clinical course
The patient was treated for both ICI myocarditis and acute coronary syndrome with intravenous methylprednisolone, oral mycophenolate mofetil, and dual antiplatelet therapy. In the absence of chest pain, and clinical symptoms compatible with decompensated heart failure, myocarditis was considered as the primary diagnosis. Therefore, invasive angiography was initially deferred. NT-proBNP decreased to 8,743 pg/ml over three days but rose again to 16,331 pg/ml. Angiography nine days after admission revealed 99% LAD occlusion, managed medically with aspirin and apixaban. The patient improved clinically and was discharged two weeks later. National MDT review of CMR images identified subtle oedema, suggestive of concurrent myocarditis. It was hypothesised that myocardial oedema caused by myocarditis had unmasked underlying coronary atherosclerosis, resulting in LAD occlusion.
Case 3
Background
A 71-year-old woman with metastatic melanoma receiving dual ICI treatment with ipilimumab (anti-CTLA-4) and nivolumab (anti-PD-1) presented 71 days after her first treatment with dyspnoea, fatigue and muscular weakness. She had completed three cycles. She had a medical history of type 2 diabetes mellitus with diabetic retinopathy.
Investigations
TnT was 38 ng/L (ULN <14 ng/L, baseline: <3 ng/L) and NT-proBNP was 571 pg/ml (ULN <400 pg/ml, baseline: 82 pg/ml). ECG and echocardiography were unremarkable. CMR seven days after admission showed mild fibrosis in the basal inferolateral wall, suggestive of convalescent myocarditis.
Clinical course
The patient was reviewed on the day of admission by the oncology service and treated for ICI myocarditis. Triple-M syndrome (myocarditis, myasthenia and myositis) was considered due to myalgia, although there was no objective clinical evidence (e.g. CK elevation or easy fatiguability) to confirm myositis or myasthenia. She was treated with intravenous methylprednisolone and oral mycophenolate mofetil. She improved clinically and biochemically and was safely discharged (NT-proBNP 249 pg/ml, TnT 19 pg/L on discharge). ICI treatment was ceased.
Case 4
Background
An 82-year-old man receiving ipilimumab plus nivolumab for synchronous epithelioid mesothelioma and renal cell carcinoma presented 166 days after initiating treatment with fatigue and diarrhoea. He had completed four cycles. His medical history included hypertension, chronic kidney disease and adrenal insufficiency of unknown cause.
Investigations
On admission, TnT was 33 ng/L (ULN <14 ng/L, baseline: 9 ng/L) and NT-proBNP was 3,338 pg/ml (ULN <210 pg/ml, baseline: 57 pg/ml). ECG demonstrated sinus tachycardia with flat T-waves in the precordial leads. Echocardiography was unremarkable.
Clinical course
Cardiology input suggested ICI myocarditis was unlikely, and the patient was not referred to the cardio-oncology MDT. Due to diarrhoea, he was treated for ICI colitis with intravenous methylprednisolone and discharged. Biomarkers improved (TnT 9 ng/L, NT-proBNP 1,804 pg/ml), and he was discharged, receiving another ICI cycle post-discharge.
Four months later, during an oncology outpatient visit, he reported orthopnoea, bilateral pedal oedema, and dyspnoea. ICI was permanently discontinued as myocarditis was suspected, though CMR was not performed at this time.
A year later, he presented in clinical shock and required intensive care unit (ICU) admission with vasopressors and intravenous hydrocortisone for suspected adrenal insufficiency. TnT was 35 ng/L, and NT-proBNP had risen to 16,958 pg/ml. Echocardiography remained unremarkable. CMR, performed 22 days later, confirmed active myocarditis. Treated with intravenous methylprednisolone, he improved and was discharged. Follow-up CMR showed resolving myocarditis.
Despite significant morbidity, the patient had a favourable outcome following appropriate immunosuppression. His initial presentation likely represented a missed diagnosis of ICI myocarditis.
Case 5
Background
A 73-year-old woman receiving cemiplimab (anti-PD-1) for metastatic buccal squamous cell carcinoma complained of dyspnoea during an outpatient review 21 days after her first treatment cycle. Her medical history included end-stage renal failure treated with renal transplantation, hypertension and pulmonary hypertension.
Investigations
Cardiac biomarkers measured in clinic showed TnT 52.5 ng/L (ULN <14 ng/L, baseline: 29 ng/L) and NT-proBNP 1,373 pg/ml (ULN <210 pg/ml, baseline: 796 pg/ml). ECG demonstrated new atrial fibrillation. Echocardiography performed five days later was unremarkable. CMR was not performed as the patient was too unwell to lie flat.
Clinical course
Due to elevated cardiac biomarkers and her symptoms of dyspnoea, the patient was admitted to hospital from the clinic for suspected ICI myocarditis. She was treated with intravenous methylprednisolone. NT-proBNP rose initially, peaking at 3,481 pg/ml, before reducing. The patient improved clinically and was safely discharged. Unfortunately, she later passed away due to progressive cancer.
Case 6
Background
An elderly man with metastatic lung adenocarcinoma receiving pembrolizumab, presented approximately a month after his first treatment cycle with dyspnoea and chest discomfort. His medical history included prior myocardial infarction, aortic stenosis, emphysema, and amiodarone-induced interstitial lung disease.
Investigations
On admission, TnT was 41 ng/L (ULN <14 ng/L, baseline: 22 ng/L) and NT-proBNP was 5,760 pg/ml (ULN <210 pg/ml, baseline: 1,186 pg/ml). ECG demonstrated new atrial fibrillation with a rapid ventricular response and left-bundle branch block morphology. Echocardiography demonstrated a reduced ejection fraction, consistent with previous echocardiograms. CMR was not performed due to severe dyspnoea and inability to comply with breath-holding instructions.
Clinical course
Initially, the elevated NT-proBNP was attributed to atrial fibrillation, but ICI myocarditis was also considered. The cardiology team managed the patient with rate control. After discussion at the cardio-oncology MDT, it was decided that persistent NT-proBNP elevation despite rate control would favour a diagnosis of ICI myocarditis. NT-proBNP levels continued to rise, peaking at 10,147 pg/ml. The patient was treated with intravenous methylprednisolone, leading to clinical and biochemical improvement. He was safely discharged following recovery.
Case 7
Background
An 80-year-old woman with advanced mucosal melanoma receiving dual ICI therapy with nivolumab and relatlimab (anti-LAG-3), developed dyspnoea on exertion and non-specific fatigue after completing two treatment cycles.
Investigations
TNT was 27 ng/L (ULN <14 ng/L, baseline: 12 ng/L) and NT-proBNP was 798 pg/ml (ULN <210 pg/ml, baseline: 322 pg/ml). ECG demonstrated normal sinus rhythm with a previously documented right-bundle branch block. Echocardiography was unremarkable. CMR revealed inflammation consistent with mild or resolving myocarditis.
Clinical course
The sudden onset of symptoms in a previously robust individual raised a strong suspicion of myocarditis. After a prompt discussion at the cardio-oncology MDT, the patient was started on immunosuppression with intravenous methylprednisolone. As the patient remained well clinically, treatment was managed in an ambulatory care setting. She attended regular reviews in the medical ambulatory unit, where she received daily infusions of methylprednisolone. This approach led to complete resolution of her symptoms and normalisation of cardiac biomarkers. ICI treatment was stopped.
Discussion
Cardiac irAEs are increasingly prevalent due to the widespread use of ICIs and improved long-term cancer survivorship. This study examines ten cases of cardiac irAEs and presents a narrative case series of seven patients with ICI myocarditis, the most common and fatal cardiac irAE. We synthesise insights into the clinical presentation, diagnosis, and management of ICI myocarditis, offering practical guidance for frontline clinicians to improve patient outcomes.
Clinical presentation
ICI myocarditis typically presented with fatigue (57%) and symptoms resembling heart failure, including dyspnoea (28%), oedema (14%), and chest discomfort (14%) in the discussed case vignettes. Echocardiograms were unremarkable in six out of seven patients, consistent with heart failure with preserved ejection fraction (HFpEF). One patient had a reduced LVEF due to concurrent ischaemia. None were diagnosed with fulminant myocarditis, a severe form marked by rapid cardiac function decline and haemodynamic instability,16 although one case (case 4) may have been misclassified. No patients presented with regional myocarditis (Takotsubo-like), a variant of ICI myocarditis characterised by ballooning of a regional wall and raised troponin levels.17
The timing of myocarditis onset varied, with most cases occurring within three treatment cycles, though one patient experienced recurrent myocarditis despite discontinuing treatment. Both monotherapy and combination therapies, including anti-CTLA-4, PD-1, PD-L1, and anti-LAG-3 regimens, were implicated.
Concurrent irAEs were observed in three of seven (43%) patients, consistent with literature suggesting an increased risk of multi-system irAEs.18 One patient (case 3) was considered for triple-M syndrome (myocarditis, myasthenia, and myositis), also known as overlap syndrome,19 emphasising the need to consider myocarditis in patients with fatigue or myalgia. Elevated CK may signal concurrent myositis.
Cardiovascular risk factors (CVRFs) were prevalent, with 70% of patients having conditions such as hypertension, diabetes, or chronic kidney disease in the case series. These factors may independently increase the risk of cardiac irAEs. For one patient (case 2), concurrent myocardial ischaemia confounded the diagnosis of myocarditis. It was hypothesised that myocardial oedema from ICI myocarditis can unmask underlying coronary atherosclerosis, due to myocardial oedema causing extrinsic vessel compression. Further, ICI treatment has been demonstrated to accelerate atherosclerosis.12,20 In patients with CVRFs and persistently abnormal cardiac biomarkers despite immunosuppression, clinicians should consider dual pathologies.
Diagnostic findings
Cardiac biomarkers, particularly NT-proBNP, are essential for diagnosing myocarditis and should be measured at baseline and at presentation. NT-proBNP levels were markedly elevated compared with TnT, with a median NT-proBNP of 4,541 pg/ml versus a median TnT of 46.5 ng/L. In five patients with baseline measurements, NT-proBNP showed a 17-fold elevation compared with a four-fold increase in TnT. NT-proBNP levels also responded rapidly to immunosuppression, highlighting its utility in monitoring treatment response. Current evidence suggests that ICI myocarditis causes oedema rather than myocyte necrosis,21 leading to NT-proBNP secretion with modest troponin elevations. This challenges current guidelines that prioritise troponin over NT-proBNP measurements for diagnosing myocarditis.
ECGs are critical for diagnosis. Indeed, a minor diagnostic criterion in the International Cardio-Oncology Society (IC-OS) guidelines is the presence of a new arrhythmia.22 Among the included cases, 29% of myocarditis presented with new atrial fibrillation. A high prevalence of new-onset arrhythmias has been reported in ICI-myocarditis, with bradyarrhythmias in particular associated with increased mortality.23,24 TTEs were often unremarkable (86%), underscoring their limited diagnostic utility. Global longitudinal strain assessments may enhance sensitivity,25 but this requires further study. Point-of-care echocardiography remains valuable for identifying structural abnormalities, such as pericardial effusion or regional wall motion abnormalities, in cases of concurrent ischaemia (case 2).
CMR is the most sensitive imaging modality, and confirmed myocarditis in four out of five cases (80%) where it was performed. CMR is a technically challenging procedure as patients must be rate-controlled and be able to lie recumbent for an extended period, while complying with breath-holding instructions.26 As such, it can be challenging to achieve high-quality images, leaving a degree of diagnostic uncertainty. Indeed, in case 1, motion artefact resulted in CMR being non-diagnostic. Cases 5 and 6 were too unwell to comply with the procedure. Literature supports variability in CMR sensitivity, with detection rates ranging from 27% to 60% across studies.10,27 Reporter expertise also influences accuracy, as two CMRs, initially reported as normal locally, were later identified as abnormal at the national cardio-oncology MDT.
Endomyocardial biopsy (EMB), the diagnostic gold standard,28 was not performed due to limited availability and logistical challenges in the UK. EMB is unlikely to become routine practice in this setting.
The above points highlight the frequent challenges in obtaining gold-standard investigations, such as CMR and EMB, underscoring the need for clinical judgement to guide diagnosis and treatment decisions.
Management
Timely referral to the cardio-oncology MDT was associated with better outcomes. For instance, case 2 was successfully managed despite dual pathologies due to MDT input. Conversely, delays in MDT referral contributed to poorer outcomes in cases 1 and 4. All suspected ICI myocarditis cases should be promptly referred to both local oncology and cardiology services to facilitate rapid review at the regional cardio-oncology MDT.
High-dose intravenous steroids remain the cornerstone of ICI myocarditis management.8 European guidelines recommend initiating a fixed dose of intravenous methylprednisolone 1,000 mg for three to five days, irrespective of body weight, followed by oral prednisolone tapering.22 Ambulatory care may be appropriate for stable patients; however, it is crucial to ensure that arrhythmias are thoroughly ruled out beforehand, as their presence would necessitate closer monitoring in a higher-acuity setting, such as high-dependency unit or cardiac-monitored bed. ICI therapy should be discontinued, with decisions to restart guided by MDT discussions. Clinicians should promptly initiate steroids when myocarditis is suspected, while initiating referral pathways to cardio-oncology services.
Optimising cardiac function with heart failure medications, particularly with sodium-glucose cotransporter type 2 (SGLT-2) inhibitors in those with preserved left ventricular function can also improve symptoms and outcomes.29 Key clinical insights are summarised as takeaways from each case (table 4), five top tips (table 5) and a diagnostic and management flowchart (figure 1).
Table 4. Key takeaways from each case of patients with ICI myocarditis
| Case 1 |
|
| Case 2 |
|
| Case 3 |
|
| Case 4 |
|
| Case 5 |
|
| Case 6 |
|
| Case 7 |
|
| Key: CK = creatine kinase; CMR = cardiac magnetic resonance; ICI = immune checkpoint inhibitor; LV = left ventricular; MDT = multi-disciplinary team; NTpro-BNP = N-terminal pro-B-type natriuretic peptide; TnT = troponin T | |
Table 5. Top tips for frontline clinicians
| Five top tips |
|
| Key: CK = creatine kinase; CMR = cardiac magnetic resonance; ICI = immune checkpoint inhibitor; MDT = multi-disciplinary team; NTpro-BNP = N-terminal pro-B-type natriuretic peptide; TnT = troponin T |
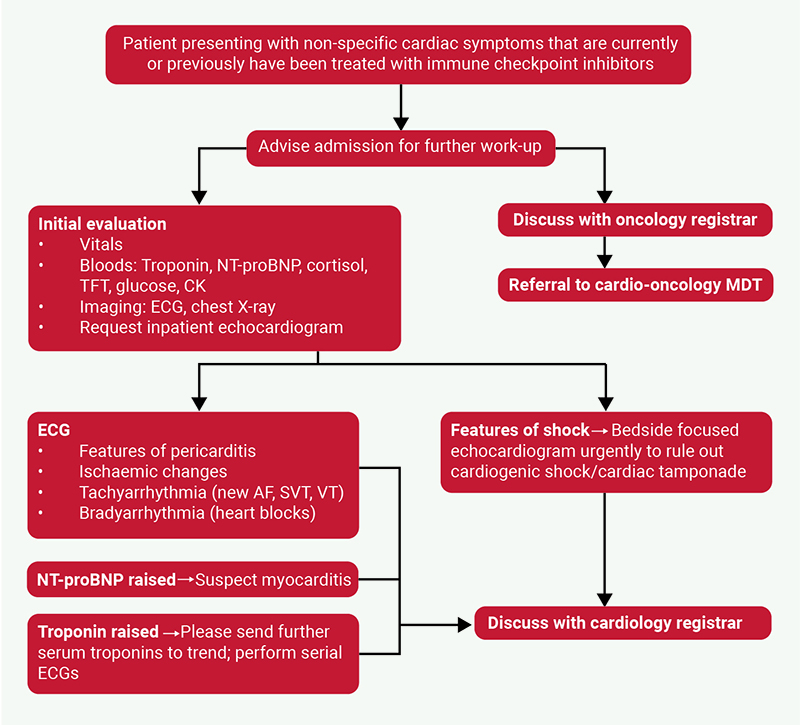
| Key: AF = atrial fibrillation; CK = creatine kinase; ECG = electrocardiogram; MDT = multi-disciplinary team; NTpro-BNP = N-terminal pro-B-type natriuretic peptide; SVT = supraventricular tachycardia; TFT = thyroid function tests; TnT = troponin T; VT = ventricular tachycardia |
Conclusion
This narrative case series highlights the need for heightened clinical vigilance, multi-disciplinary collaboration, and adherence to emerging best practices to optimise outcomes for patients with ICI myocarditis.
A key strength of this qualitative analysis is, unlike prior quantitative studies, it offers detailed insights into the clinical pathways of affected patients. Limitations include the small sample size, retrospective verbal consent process, absence of histological or definitive imaging confirmation in all patients, and lack of long-term outcome data.
This article underscores the need to validate current diagnostic guidelines. For instance, NT-proBNP appears to be a more sensitive biomarker than TnT in ICI myocarditis. Most patients had preserved systolic function, calling into question the utility of the IC-OS guidelines’ minor criteria.29 Current guidance should also emphasise the importance of clinical judgement in the diagnosis and management of ICI myocarditis, given the specialist nature of gold-standard investigations, such as CMR.
Overall, this study emphasises early diagnosis, multi-disciplinary input, and evidence-based management to improve outcomes for patients with ICI myocarditis.
Key messages
- Immune checkpoint inhibitor (ICI) myocarditis should be considered in patients with current or prior receipt of ICIs who present with chronic heart failure symptoms. Failure to suspect ICI myocarditis can lead to delayed diagnoses and significant morbidity and mortality. Clinical judgement is central to its diagnosis
- Comparing cardiac biomarker (troponin T [TnT], N-terminal pro-B-type natriuretic protein [NT-proBNP] and creatine kinase [CK]) measurements to baseline values is essential in the work-up of patients with suspected ICI myocarditis
- Cardiac magnetic resonance (CMR) is the gold-standard imaging modality for ICI myocarditis. However, it is not often routinely available, and its sensitivity can be limited by patient non-compliance (e.g. with breath-holding instructions) and lack of radiologist expertise. Transthoracic echocardiography is often normal
- All patients with suspected ICI myocarditis should be promptly referred to the regional cardio-oncology multi-disciplinary team (MDT) for discussion via local pathways, as multi-disciplinary input can resolve diagnostic uncertainty and improve outcomes
- ICI myocarditis, when clinically suspected, should be promptly treated with intravenous steroids, even if imaging or biomarkers are inconclusive. Monitor cardiac biomarkers and clinical status closely over 48–72 hours to assess treatment response before making changes to therapy
Conflicts of interest
None declared.
Funding
None.
Patient consent
Verbal informed consent to publish case-related details was retrospectively obtained from patients or, in the case of deceased individuals, from their next of kin. Voluntary participation and the right to decline without impact on care was emphasised. For cases 2 and 6, posthumous consent could not be obtained due to difficulties in contacting their next of kin. All cases have been maximally anonymised while retaining essential clinical details.
References
1. Wolchok JD, Chiarion-Sileni V, Rutkowski P et al. Final, 10-year outcomes with nivolumab plus ipilimumab in advanced melanoma. N Engl J Med 2025;392:11–22. https://doi.org/10.1056/NEJMoa2407417
2. Waldman AD, Fritz JM, Lenardo MJ. A guide to cancer immunotherapy: from T cell basic science to clinical practice. Nat Rev Immunol 2020;20:651–68. https://doi.org/10.1038/s41577-020-0306-5
3. Tawbi HA, Schadendorf D, Lipson EJ et al. Relatlimab and nivolumab versus nivolumab in untreated advanced melanoma. N Engl J Med 2022;386:24–34. https://doi.org/10.1056/NEJMoa2109970
4. Mallio CA, Bernetti C, Cea L et al. Adverse effects of immune-checkpoint inhibitors: a comprehensive imaging-oriented review. Curr Oncol 2023;30:4700–23. https://doi.org/10.3390/curroncol30050355
5. Johnson DB, Balko JM, Compton ML et al. Fulminant myocarditis with combination immune checkpoint blockade. N Engl J Med 2016;375:1749–55. https://doi.org/10.1056/NEJMoa1609214
6. D’Souza M, Nielsen D, Svane IM et al. The risk of cardiac events in patients receiving immune checkpoint inhibitors: a nationwide Danish study. Eur Heart J 2021;42:1621–31. https://doi.org/10.1093/eurheartj/ehaa884
7. Laenens D, Yu Y, Santens B et al. Incidence of cardiovascular events in patients treated with immune checkpoint inhibitors. J Clin Oncol 2022;40:3430–8. https://doi.org/10.1200/JCO.21.01808
8. Palaskas N, Lopez‐Mattei J, Durand JB, Iliescu C, Deswal A. Immune checkpoint inhibitor myocarditis: pathophysiological characteristics, diagnosis, and treatment. J Am Heart Assoc 2020;9:e013757. https://doi.org/10.1161/JAHA.119.013757
9. Mahmood SS, Fradley MG, Cohen JV et al. Myocarditis in patients treated with immune checkpoint inhibitors. J Am Coll Cardiol 2018;71:1755–64. https://doi.org/10.1016/j.jacc.2018.02.037
10. Nielsen DL, Juhl CB, Nielsen OH, Chen IM, Herrmann J. Immune checkpoint inhibitor-induced cardiotoxicity: a systematic review and meta-analysis. JAMA Oncol 2024;10:1390–9. https://doi.org/10.1001/jamaoncol.2024.3065
11. Li C, Bhatti SA, Ying J. Immune checkpoint inhibitors-associated cardiotoxicity. Cancers (Basel) 2022;14:1145. https://doi.org/10.3390/cancers14051145
12. Barcia Durán JG, Das D, Gildea M et al. Immune checkpoint landscape of human atherosclerosis and influence of cardiometabolic factors. Nat Cardiovasc Res 2024;3:1482–502. https://doi.org/10.1038/s44161-024-00563-4
13. Ball S, Ghosh RK, Wongsaengsak S et al. Cardiovascular toxicities of immune checkpoint inhibitors: JACC review topic of the week. J Am Coll Cardiol 2019;74:1714–27. https://doi.org/10.1016/j.jacc.2019.07.079
14. Norwood TG, Westbrook BC, Johnson DB et al. Smoldering myocarditis following immune checkpoint blockade. J Immunother Cancer 2017;5:91. https://doi.org/10.1186/s40425-017-0296-4
15. Mahmood SS, Fradley MG, Cohen JV et al. Myocarditis in patients treated with immune checkpoint inhibitors. J Am Coll Cardiol 2018;71:1755–64. https://doi.org/10.1016/j.jacc.2018.02.037
16. Hang W, Chen C, Seubert JM, Wang DW. Fulminant myocarditis: a comprehensive review from etiology to treatments and outcomes. Signal Transduct Target Ther 2020;5:287. https://doi.org/10.1038/s41392-020-00360-y
17. Serzan M, Rapisuwon S, Krishnan J, Chang IC, Barac A. Takotsubo cardiomyopathy associated with checkpoint inhibitor therapy: endomyocardial biopsy provides pathological insights to dual diseases. JACC CardioOncol 2021;3:330–4. https://doi.org/10.1016/j.jaccao.2021.02.005
18. Wan G, Chen W, Khattab S et al. Multi-organ immune-related adverse events from immune checkpoint inhibitors and their downstream implications: a retrospective multicohort study. Lancet Oncol 2024;25:1053–69. https://doi.org/10.1016/S1470-2045(24)00278-X
19. Adeoye FW, Jaffar N, Surandran S, Begum G, Islam MR. Durvalumab-induced triple-M syndrome. Eur J Case Rep Intern Med 2024;11:004729. https://doi.org/10.12890/2024_4729
20. Drobni ZD, Alvi RM, Taron J et al. Association between immune checkpoint inhibitors with cardiovascular events and atherosclerotic plaque. Circulation 2020;142:2299–311. https://doi.org/10.1161/CIRCULATIONAHA.120.049981
21. Palaskas NL, Segura A, Lelenwa L et al. Immune checkpoint inhibitor myocarditis: elucidating the spectrum of disease through endomyocardial biopsy. Eur J Heart Fail 2021;23:1725–35. https://doi.org/10.1002/ejhf.2265
22. Lyon AR, López-Fernández T, Couch LS et al. 2022 ESC guidelines on cardio-oncology developed in collaboration with the European Hematology Association (EHA), the European Society for Therapeutic Radiology and Oncology (ESTRO) and the International Cardio-Oncology Society (IC-OS): developed by the Task Force on Cardio-Oncology of the European Society of Cardiology (ESC). Eur Heart J 2022;43:4229–361. https://doi.org/10.1093/eurheartj/ehac244
23. Song W, Zheng Y, Dong M et al. Electrocardiographic features of immune checkpoint inhibitor-associated myocarditis. Curr Probl Cardiol 2023;48:101478. https://doi.org/10.1016/j.cpcardiol.2022.101478
24. Atallah-Yunes SA, Kadado AJ, Kaufman GP, Hernandez-Montfort J. Immune checkpoint inhibitor therapy and myocarditis: a systematic review of reported cases. J Cancer Res Clin Oncol 2019;145:1527–57. https://doi.org/10.1007/s00432-019-02927-x
25. Coustal C, Vanoverschelde J, Quantin X et al. Prognosis of immune checkpoint inhibitors-induced myocarditis: a case series. J Immunother Cancer 2023;11:e004792. https://doi.org/10.1136/jitc-2022-004792
26. Kramer CM, Barkhausen J, Bucciarelli-Ducci C, Flamm SD, Kim RJ, Nagel E. Standardized cardiovascular magnetic resonance imaging (CMR) protocols: 2020 update. J Cardiovasc Magn Reson 2020;22:17. https://doi.org/10.1186/s12968-020-00607-1
27. Arcari L, Tini G, Camastra G et al. Cardiac magnetic resonance imaging in immune check-point inhibitor myocarditis: a systematic review. J Imaging 2022;8:99. https://doi.org/10.3390/jimaging8040099
28. Asher A. A review of endomyocardial biopsy and current practice in England: out of date or underutilised? Br J Cardiol 2017;24:108–12. http://doi.org/10.5837/bjc.2017.019
29. Liu Z, Fan Y, Guo J, Bian N, Chen D. Fulminant myocarditis caused by immune checkpoint inhibitor: a case report and possible treatment inspiration. ESC Heart Fail 2022;9:2020–6. https://doi.org/10.1002/ehf2.13912
