A 78-year-old man presented to the cancer assessment bay with a history of progressive fatigue, generalised muscle pain, and bilateral ptosis after completing two cycles of nivolumab/ipilimumab for metastatic renal cancer. Physical examination revealed mild bilateral ptosis (right > left), worsening with upward gaze, binocular diplopia, and fatigable weakness in the right shoulder. Electrocardiogram (ECG) showed a new right-bundle branch block (RBBB), with left-axis deviation, and left ventricular hypertrophy. Blood analyses showed elevated troponin I, brain natriuretic peptide, and creatine kinase with values of 221 ng/L, 114 ng/L, and 1,109 U/L, respectively. He was managed as immune checkpoint inhibitor-related severe myocarditis, myositis, and myasthenia gravis overlap (triple M) syndrome with high-dose steroids and pyridostigmine, resulting in clinical and biochemical improvement. However, immunotherapy was permanently discontinued, and follow-up imaging after three months showed evidence of disease progression. This case explores the importance of a multi-disciplinary approach in the effective management of a rare and severe immune-related toxicity and the impact of toxicities on treatment options and cancer progression.
Introduction
Immune checkpoint inhibitors (ICI) are a class of novel anticancer therapies with significant improvement in outcomes.1 They work by stimulating immune-mediated recognition and clearance of cancer cells. This may lead to immune cell over-activation manifesting as inflammatory toxicities.1,2
While ICI-induced toxicities, like colitis, endocrinopathies and dermatitis, may occur in up to 40% of people, myocarditis is rare, occurring in less than 1% of the treated population. Furthermore, an overlap syndrome of myocarditis, myositis, and myasthenia is extremely uncommon, and only a few cases have been reported so far.3 This overlap syndrome, sometimes called ‘triple M’ syndrome or ‘MMM triad’, poses a significant risk of morbidity and mortality because of its non-specific presentation and risk of rapid deterioration.3
Case
We present a 78-year-old man with a history of metastatic renal cancer, receiving palliative combination immunotherapy (ipilimumab/nivolumab). He attended the cancer assessment bay after the second cycle of immunotherapy, with a 10-day history of progressively worsening fatigue, shortness of breath on exertion, generalised body aches, and progressive drooping of both eyelids for four days, but no history of chest pain. He had a background history of ischaemic heart disease, being treated with bisoprolol, ramipril, and atorvastatin.
Examination revealed bilateral ptosis, binocular diplopia, and fatigable weakness in the right shoulder. There was no dysarthria, nor sensory impairment. He had normal heart sounds with no murmurs or peripheral oedema, and vital signs were normal. Electrocardiogram (ECG) showed a new right-bundle branch block (RBBB), with left-axis deviation, and left ventricular hypertrophy.
Blood analyses revealed raised levels of creatine kinase (CK), troponin I and brain natriuretic peptide (BNP) with values of 1,109 U/L (normal range 40–320 U/L), 221 ng/L (normal range <4 ng/L) and 114 ng/L (normal range <100 ng/L), respectively. On account of these findings, we made a diagnosis of ICI-induced myocarditis, myositis, and myasthenia overlap syndrome (Common Terminology Criteria for Adverse Events/CTCAE grade 4).
He was jointly managed by the oncology, neurology and cardiology teams. He commenced pulsed dosing of methylprednisolone at 1 g daily and pyridostigmine 30 mg three times a day. Further investigations, including autoimmune panel, echocardiogram, nerve conduction study, and electromyography, were normal.
While the generalised muscle pains and fatigue improved within 48 hours of pulsed methylprednisolone therapy, ocular symptoms (ptosis and diploplia) persisted, requiring pyridostigmine dose escalation to 60 mg four times a day on day 3, leading to improvement. Troponin I and CK levels improved steadily with the resulting trend shown in figure 1 and figure 2, respectively.
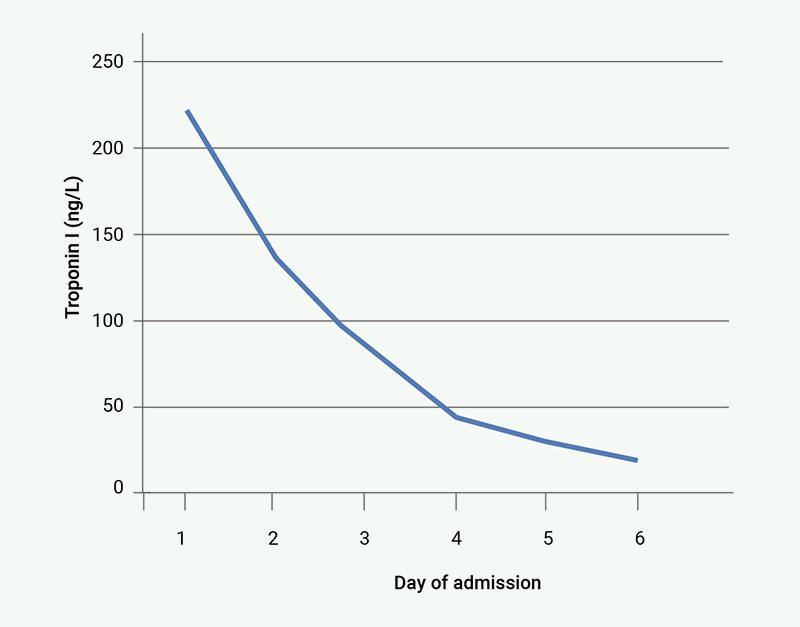
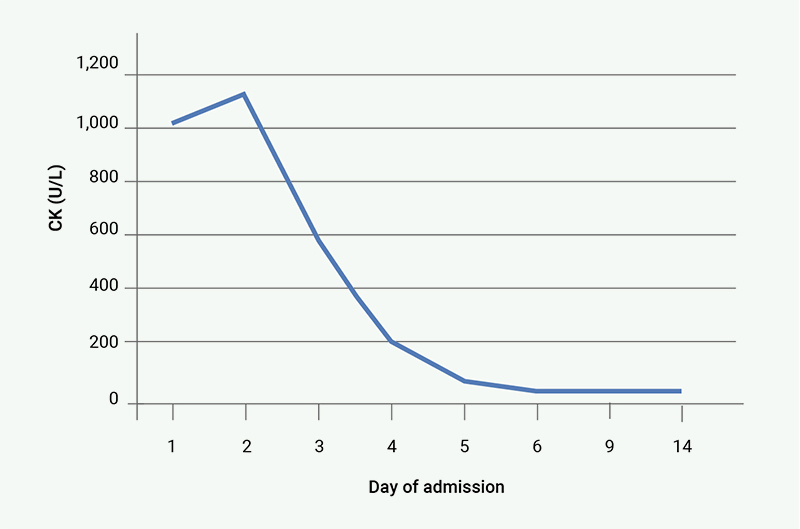
After five days of admission he was discharged on pyridostigmine and a weaning dose of prednisolone. Follow-up ECGs were normal, and biochemical markers normalised on the third week of review after discharge. Immunotherapy was permanently discontinued on account of severe adverse event, and restaging computed tomography (CT) scan unfortunately showed evidence of cancer progression.
Discussion
Current data suggest that ICI-induced myocarditis alone has mortality rates ranging from 25% to 50%. It often occurs as an isolated event, but about 30% of patients develop overlapping myositis and/or myasthenia gravis.3–5
According to a recent review of case reports on triple M syndrome, ocular symptoms of ptosis and diplopia were the most common presenting symptoms, occurring in 58% and 42% of patients, respectively.5 Fatigue was less common, occurring in only 24% of patients.5 Our patient had no chest pain or cardiac-specific symptoms, despite having a raised troponin I level and new ECG changes. Similarly, in cases reported so far, only 8% of patients presented with chest pain, and less than 6% had additional cardiac symptoms, like light-headedness and presyncope.5 This obscure presentation tends to delay diagnosis, which can lead to unfavourable outcomes. Hence, it is important to investigate the other components of the triple M syndrome when one is identified. Bedside ECG may show conduction blocks and arrhythmia, but is normal in many patients. Cardiac magnetic resonance imaging (MRI) is more sensitive in characterising myocardial inflammation. However, this is often not immediately available. Serological markers for myasthenia, such as anti-AChR (acetylcholine receptor) antibodies, are usually negative, hence, clinical examination is important in achieving diagnosis.3,5
The pathophysiology is related to ICI-induced immune dysregulation.1 Ipilimumab and nivolumab, block CTLA-4 (cytotoxic T-lymphocyte associated protein 4) and PD-1 (programmed cell death protein 1), respectively, potentiating T-cell-mediated immune responses against tumour cells. However, these pathways are also important to maintaining immune tolerance and, upon inhibition, may result in autoimmunity. Other mechanisms noted include cross-reactivity between tumour antigens and antigens of normal tissues, and expression of PD-1/PD-L1 (programmed death-ligand 1) by normal tissues.1–5
Management involves the early introduction of high-dose corticosteroids.3 Additional therapies, such as pyridostigmine, intravenous immunoglobulins, and plasmapheresis, are important, especially in steroid-resistant cases.3,5 Given the risk for respiratory compromise from both myasthenia and myocarditis, close monitoring of respiratory and cardiac functions should be done using regular single breath counts, ECGs and troponin trends.
Although this patient had a remarkable recovery from this toxicity, he nonetheless had a side effect related to anticancer treatment, which led to the discontinuation of a potentially effective therapeutic option, a period of inpatient admission and recovery, culminating in a prolonged period without any specific anticancer treatment. This inevitably increases the risk of cancer progression, especially on the background of a metastatic disease.
Key messages
- Ipilimumab/nivolumab can cause severe myocarditis, myositis and myasthaenia overlap syndrome
- Early recognition and multi-disciplinary management can bring about a favourable outcome in a rare and obscure toxicity syndrome with up to 50% mortality risk
- Beyond survivorship, toxicities impact patient’s treatment options and pose a risk for cancer progression
Conflicts of interest
None declared.
Funding
None.
Patient consent
Written informed consent was obtained from the patient for publication of this case report.
References
1. Topalian SL, Drake CG, Pardoll DM. Immune checkpoint blockade: a common denominator approach to cancer therapy. Cancer Cell 2015;27:450–61. https://doi.org/10.1016/j.ccell.2015.03.001
2. Keam S, Turner N, Kugeratski FG et al. Toxicity in the era of immune checkpoint inhibitor therapy. Front Immunol 2024;15:1447021. https://doi.org/10.3389/fimmu.2024.1447021
3. Lipe DN, Qdaisat A, Krishnamani PP et al. Myocarditis, myositis, and myasthenia gravis overlap syndrome associated with immune checkpoint inhibitors: a systematic review. Diagnostics (Basel) 2024;14:1794. https://doi.org/10.3390/diagnostics14161794
4. Jeyakumar N, Etchegaray M, Henry J et al. The terrible triad of checkpoint inhibition: a case report of myasthenia gravis, myocarditis, and myositis induced by cemiplimab in a patient with metastatic cutaneous squamous cell carcinoma. Case Reports Immunol 2020;2020:5126717. https://doi.org/10.1155/2020/5126717
5. Longinow J, Zmaili M, Skoza W et al. Immune checkpoint inhibitor induced myocarditis, myasthenia gravis, and myositis: a single-center case series. Cancer Med 2023;12:2281–9. https://doi.org/10.1002/cam4.5050

