Small-interfering RNA (siRNA)-based therapies, such as inclisiran, offer a novel approach to reducing low-density lipoprotein-cholesterol (LDL-C) and preventing cardiovascular disease (CVD). Inclisiran inhibits proprotein convertase subtilisin/kexin type 9 (PCSK9) synthesis, enhancing LDL-receptor recycling and LDL-particle clearance. Although clinical trials have established its efficacy with LDL-C reductions of 44–52%, real-world studies have reported variable reductions over shorter follow-up periods. This study aimed to assess the long-term efficacy and safety of inclisiran in a diverse real-world cohort.
A total of 238 patients initiating inclisiran between January 2022 and January 2024 at a tertiary lipid service were included. Data on lipid profiles, comorbidities, and medication history were collected from electronic healthcare records. LDL-C reductions were analysed at each dose using a Bayesian hierarchical model, and subgroup analyses explored the influence of familial hypercholesterolaemia (FH) and baseline lipid-lowering therapy.
Inclisiran therapy resulted in a mean LDL-C reduction of 48.4% following the first dose, sustained over 27 months. Patients receiving three or more lipid-lowering therapies at baseline achieved greater LDL-C reductions compared with others (67.4% after first dose vs. 47.6%). No discernible differences in efficacy were observed between patients with and without FH. Inclisiran was well tolerated, with only six patients discontinuing therapy due to adverse events or preference. Approximately 35% of patients met the European Society of Cardiology LDL-C target of <1.4 mmol/L after the first dose, declining to 28% after the fourth dose.
These real-world findings demonstrate that inclisiran is a well-tolerated and effective lipid-lowering therapy, achieving reductions comparable with clinical trial results. Greater reductions in patients on multiple baseline therapies suggest the importance of comprehensive lipid management. While achieving stringent LDL-C targets remains challenging, inclisiran’s practical benefits, including infrequent dosing and good tolerability, underscore its potential to improve CVD outcomes in diverse populations.
Introduction
Small-interfering RNA (siRNA)-based therapies offer a promising approach to cardiovascular disease (CVD) prevention. Inclisiran is a siRNA that reduces the synthesis of proprotein convertase subtilisin/kexin type 9 (PCSK9), thereby increasing low-density lipoprotein (LDL)-receptor recycling and removal of LDL particles from the circulation. In phase 3 clinical trials,1 its effects are akin to anti-PCSK9 monoclonal antibodies, which have documented benefits in the prevention of CVD.2 Indeed, there is a well-established benefit of LDL-cholesterol (LDL-C) reduction regardless of mechanism.3 Inclisiran has some practical benefits compared with other medications, mainly arising from its low frequency of administration (once every 6 months from the second dose) and acceptable side effect profile.
In clinical trials, inclisiran has delivered a LDL-C reduction of between 44%4,5 and 52%,1 with more recent evidence suggesting a reduction of 50% that appears durable up to 6.8 years.6 While these studies have established the efficacy of inclisiran, real-world studies have reported variable degrees of LDL-C reduction, between 26% and 48.6%, with shorter follow-up periods of up to three months.7–10 Previously, this centre has reported a 48.6% LDL-C reduction at two months in 80 patients newly initiated on inclisiran.11 All these real-world studies suffer from a short follow-up time. To address this gap, this study aimed to provide insights into the long-term efficacy and safety of inclisiran in a diverse real-world population.
Method
All 238 patients who initiated inclisiran between January 2022 and January 2024 via the tertiary lipid service at Hammersmith Hospital, London, were included. Electronic healthcare records were interrogated obtaining baseline lipid profiles, glycated haemoglobin (HbA1c) levels, medication history and familial hypercholesterolaemia (FH) diagnosis data. Further data on LDL-C concentrations were obtained for each patient following each dose of inclisiran. ‘Dose of inclisiran’ was utilised as a categorical variable to describe each time point, owing to the asymmetrical dosing strategy (second dose at three months following the first dose, then six-monthly thereafter), with a maximum follow-up time of post the fifth dose (up to 27 months since initial inclisiran dose). Adverse events and decisions to stop inclisiran were recorded.
To mitigate potential confounding and explicitly represent causal assumptions underlying our analysis, we constructed a directed acyclic graph a priori, provided as supplementary material 1. Raw participant LDL-C data at each time point are supplied as supplementary material 2. Missing values for HbA1c (n=8/238) were imputed with the median HbA1c value (40 mmol/mol). HbA1c values were then median-centred for modelling. Bayesian hierarchical linear models were then fitted to the data through the use of the brms R package.12 First, a model was fitted to examine the overall reduction in LDL-C levels following the administration of inclisiran (equations 1–6, available online). Further models were then fitted to explore the interaction between FH and lipid therapy status prior to inclisiran administration (equations 7–10, available online), at each time point, and including the baseline HbA1c as a covariate. Raw data are again supplied as supplementary material 3. Weakly informative priors were specified in order to regularise, but not dominate, our inferences.13 Models were compared by means of efficient approximate leave-one-out cross-validation and these results are provided as supplementary material 4.14 Posterior distributions are summarised as the 2.5th, 50th, and 97.5th percentiles. All Markov Monte-Carlo chains converged for each of the parameters in the model (R̂=1.0). The R code required to reproduce these analyses is provided as supplementary material 5.
Equations
- log (yi ~ T (v, μi , σ )
- μi = α + αpatient[i] + β1timei + β2HbA1ci
- α ~ N (mean [log {y}],2.5⋅sd [log {y}])
- βᵪ ~ N (0,2.5⋅sd [log{y}])
- αpatient ~ N (α,σpatient)
- σpatient , σ ~ Half–T(3,0,2.5)
- v ~ Gamma (2,0.1)
- μi = α + αpatient[i] + β1timei + β2FHi + β3(timei x FHi) + β4HbA1ci
- μi = α + αpatient[i] + β1timei + β2lipidi + β3(timei x lipidi) + β4HbA1ci
- μi = α + αpatient[i] + β1timei + β2FHi + β3lipidi + β4(timei x FHi) + β5(timei x lipidi) + β6HbA1ci
Results
Patient features at baseline
Table 1. Participant characteristics
| Characteristic | Number (%) |
| Patients enrolled at baseline | 238 (100.0) |
| Patient with results following dose 1 | 218 (91.6) |
| Patient with results following dose 2 | 136 (57.1) |
| Patient with results following dose 3 | 85 (35.7) |
| Patient with results following dose 4 | 46 (19.3) |
| Patient with results following dose 5 | 17 (7.1) |
| Assigned male at birth | 150 (63.0) |
| Assigned female at birth | 88 (37.0) |
| Ethnicity: White | 113 (47.5) |
| Ethnicity: Asian | 37 (15.5) |
| Ethnicity: Black | 8 (3.4) |
| Ethnicity: Mixed | 4 (1.7) |
| Ethnicity: Other | 76 (31.9) |
| Patients on a statin at baseline | 142 (59.7) |
| Patients on 3 or more lipid-lowering therapies at baseline | 29 (12.2) |
| Patients on no lipid-lowering therapy at baseline | 42 (17.6) |
| Diagnosis of FH at baseline | 50 (21) |
| Diagnosis of diabetes mellitus at baseline | 42 (17.6) |
| Mean age ± SD, years | 62.2 ± 10.5 |
| Key: FH = familial hypercholesterolaemia; SD = standard deviation | |
All 238 patients who initiated inclisiran were included in the study, with the baseline characteristics and number of patients at each time point described in table 1.
All available patient results were included, even if they ceased inclisiran therapy, and their dose time points were defined by their first dose and dosing schedule rather than when they received the further doses. Some patients continued their inclisiran therapy locally at their GP practice, and we did not have access to further results. Others did not have a blood test within each time point, or had not completed sufficient doses of inclisiran, hence, the reduction in numbers following each time point.
Adverse events and decision to stop inclisiran therapy
Inclisiran was well tolerated in this group, with only six patients ceasing therapy. One patient had a reported severe adverse event (rashes and symptoms of a systemic arthritis) that was felt related to inclisiran therapy and, thus, was no longer offered inclisiran. Five patients made the decision to stop inclisiran therapy, related to the experience of adverse events or other preference.
Table 2. Adverse effects recorded in relation to inclisiran therapy (N=238)
| Adverse event | Number (%) |
| Dizziness | 2 (0.8) |
| Reflux | 1 (0.4) |
| Muscle aches | 5 (2.1) |
| Injection-site reaction | 8 (3.4) |
| Exacerbation of fibromyalgia | 1 (0.4) |
| Hair loss | 2 (0.8) |
| Insomnia | 1 (0.4) |
| Systemic arthritis | 1 (0.4) |
| Skin reaction (non-injection site) | 3 (1.3) |
The recorded adverse events are contained within table 2, with the most common side effect being injection-site reactions (3.4%), followed by muscle aches (2.1%).
Linear modelling
The efficient approximate leave-one-out cross-validation results (supplementary material 4) demonstrated that the model including only the interaction with time since treatment and number of lipid therapies prior to treatment (equation 9), was expected to best predict future data.
Reduction in LDL-C was sustained over time
Figure 1 shows the average reduction in LDL-C over time, suggesting that inclisiran therapy led to a 48.4% reduction in LDL-C after the first dose, that was stable over time, up to 27 months following initiation. The lowest reduction was seen following dose three, with a mean reduction of 40.2% (95% credible interval [CrI] 44.6% to 35.4%). The highest was seen following dose five at 52.1% (95%CrI 58.9% to 44.2%), although this presents with greater uncertainty. Table 3 contains the percentage relative reduction with 95%CrI at each time point.
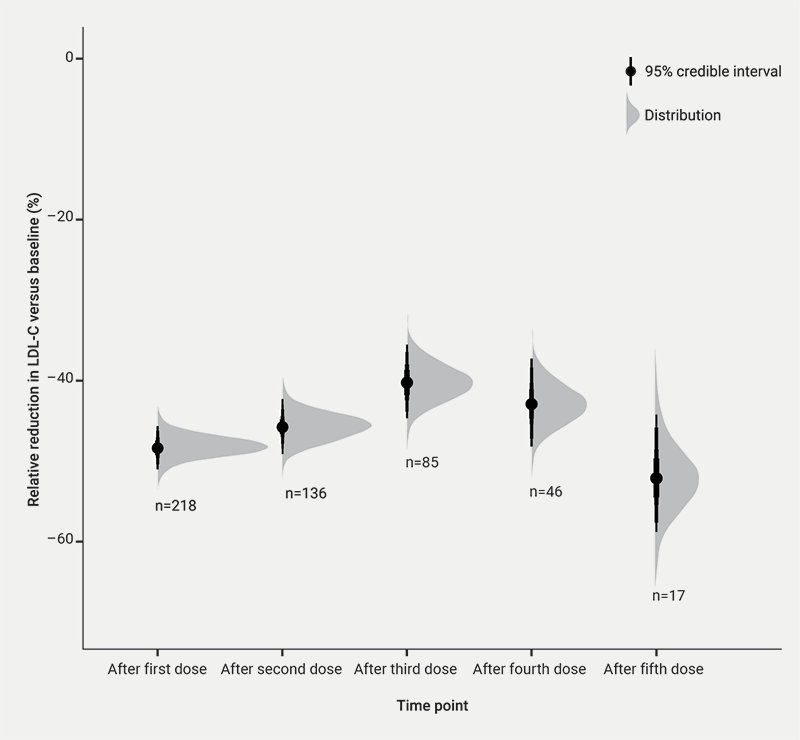
Table 3. Reduction in low-density lipoprotein-cholesterol (LDL-C) over time, compared with baseline LDL-C
| Time point | LDL-C reduction (95% credible interval), % |
| After dose 1 | 48.4 (51.0 to 45.6) |
| After dose 2 | 45.7 (49.1 to 42.1) |
| After dose 3 | 40.2 (44.8 to 35.5) |
| After dose 4 | 42.9 (48.3 to 37.2) |
| After dose 5 | 51.9 (58.7 to 44.1) |
Patients with FH appeared to have a similar benefit from inclisiran to patients without FH
Figure 2 describes the LDL-C reduction across the three groups analysed in this study:
- A ‘reference’ group that includes patients who are FH negative and on less than three other lipid-lowering medications at inclisiran initiation
- Those that were FH positive
- Those who were on at least three other lipid-lowering medications at inclisiran initiation.
There were no discernible differences in inclisiran efficacy in those with or without FH: LDL-C reductions of 41.3% (95%CrI 48.5% to 33.0%) versus 47.6% (95%CrI 50.7% to 44.4%).
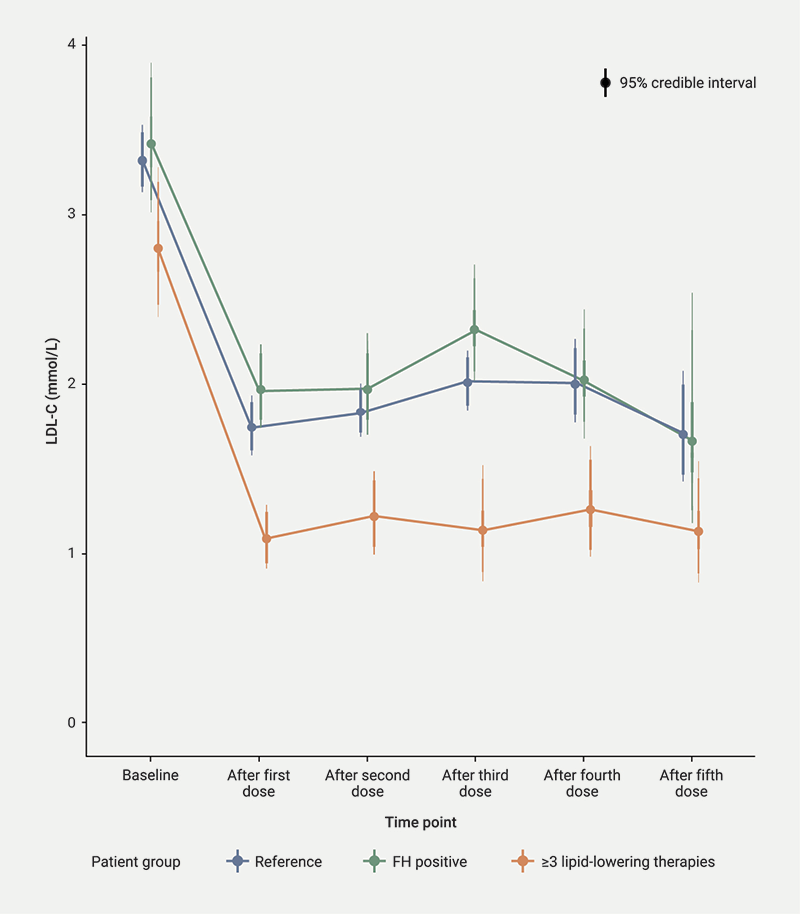
Patients who were established on three other lipid-lowering therapies prior to commencing inclisiran appear to benefit more from the drug
Figure 2 also includes the reduction in LDL-C for patients established on three or more lipid-lowering therapies when starting inclisiran. There is a greater reduction in LDL-C in these patients, at 67.4% (95%CrI 72.6% to 61.1%) following the first dose, which was sustained over the five time points.
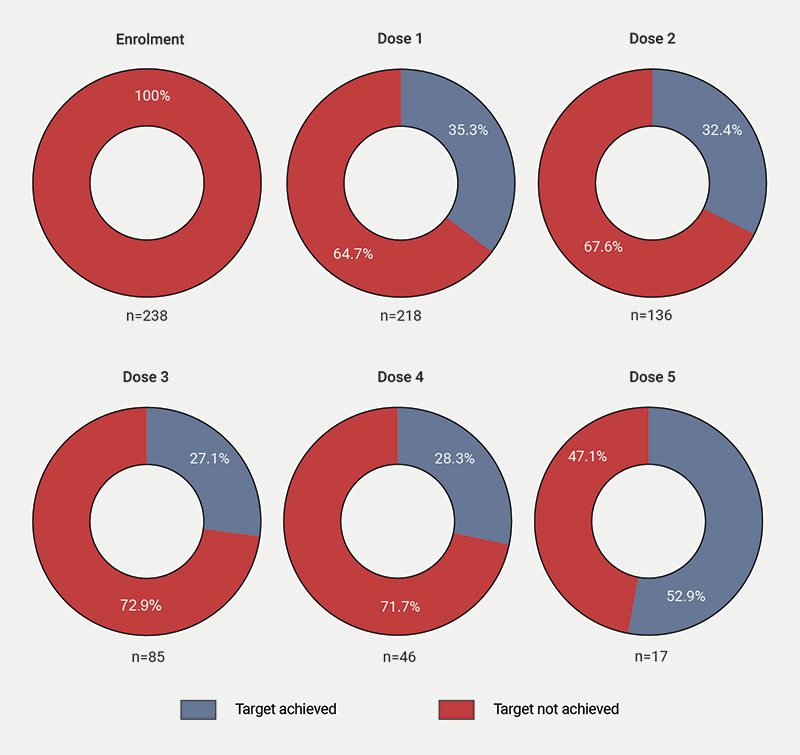
Proportion of patients meeting ESC guidance for LDL-C targets
Figure 3 shows the proportion of patients meeting the European Society of Cardiology (ESC) target for LDL-C (<1.4 mmol/L) in the presence of atherosclerotic CVD. Approximately 35% met target after the first dose, declining to 28% after the fourth dose.
Discussion
The results from this large real-world cohort support the use of inclisiran as a well-tolerated and effective lipid-lowering medication. An observed LDL-C reduction of 48% aligns with findings from the ORION-3 and ORION-8 trials,4,6 but is slightly below the 52% reductions reported in the ORION-10 and ORION-11 trials.1 This discrepancy may be attributed to the real-world nature of the data, including variations in compliance with other lipid-lowering therapies.
This diverse cohort included 52.5% non-white participants, with a notable representation of Asian individuals (n=37), a group typically underrepresented in ORION trials. The recently reported ORION-18 trial,15 conducted across multiple sites in East Asia, demonstrated the efficacy and safety of inclisiran, with 71.7% of participants achieving a ≥50% reduction in LDL-C by day 330. However, further studies are needed to explore its impact on underrepresented groups, such as South Asian populations.
Among the 50 patients with heterozygous FH, LDL-C reduction was consistent with the 39.7% reported in the ORION-9 trial.5 No discernible difference was observed between the FH and non-FH groups. While LDL-C lowering in FH patients was less pronounced than the 59% achieved with monoclonal antibodies targeting PCSK9,2 inclisiran’s infrequent dosing regimen offers a practical advantage.
Patients on three or more lipid-lowering therapies at baseline appeared to derive greater LDL-C reductions from inclisiran. This highlights the importance of continuing oral therapies alongside inclisiran. The aetiology of this observation is unclear, and is unlikely to be due to the pharmacologic or physiologic properties of inclisiran interacting with other lipid-lowering therapies. Instead, the observed benefit may reflect improved adherence fostered by attendance at specialised lipid or cardiovascular risk clinics, underscoring their value in managing high-risk patients.
The ESC recommends a LDL-C target of <1.4 mmol/L for high-risk individuals with atherosclerotic cardiovascular disease.16 In the UK, inclisiran initiation requires baseline LDL-C levels of at least 2.6 mmol/L despite maximum-tolerated therapy. Thus, this cohort is selected by poor response or tolerance to conventional therapy in the face of significant atherosclerotic CVD. Approximately 35% of this cohort achieved the <1.4 mmol/L target after the first dose, declining to 28% by dose four. This decline likely reflects the longitudinal nature of real-world data, rather than a cumulative medication effect, alongside the jump in proportion meeting target at dose five related to the smaller number of patients at this time point. Achieving these stringent targets remains challenging; recent evidence suggests only 18% of very high-risk patients meet them.17
Nevertheless, LDL-C lowering is strongly associated with reduced cardiovascular events and mortality, even at levels below guideline targets.18 Inclisiran, with its significant LDL-C reductions, infrequent dosing, and good tolerability, offers substantial potential for real-world benefits. Early pooled analyses of ORION-9, -10, and -11 indicate a trend towards reduced major adverse cardiovascular events (MACE) at 18 months,19 with forthcoming cardiovascular trials expected to confirm these findings.
Key messages
- Inclisiran, a small-interfering RNA-based therapy targeting proprotein convertase subtilisin/kexin type 9 (PCSK9), demonstrates low-density lipoprotein-cholesterol (LDL-C) reductions of 44–52% in clinical trials, with sustained effects over 6.8 years. However, real-world evidence has been limited to short-term follow-up (≤3 months) and smaller cohorts, with variable efficacy (26–48.6% LDL-C reduction)
- This real-world study confirms inclisiran’s durable efficacy (48.4% LDL-C reduction sustained over 27 months) and safety in a diverse cohort, including underrepresented groups. Patients on ≥3 lipid-lowering therapies achieved greater reductions (67.4%), while no difference was observed between familial hypercholesterolaemia (FH) and non-FH subgroups. Only 28–35% met European Society of Cardiology (ESC) LDL-C targets, highlighting residual risk
- The findings support inclisiran’s role in long-term lipid management, particularly for high-risk patients, and underscore the need for combination therapy adherence. Policymakers may consider broader access given its infrequent dosing and tolerability, while researchers should explore strategies to improve target attainment in real-world settings
Conflicts of interest
JC has received speaker/consultancy fees or research grants from Novartis, Amgen, Sanofi, Amyrt, Pfizer, Akcea, Ultragenyx, Chiesi, Silence Therapeutics, and Verve Therapeutics.
KR has declared research grants to institution from Amgen, Sanofi, Regeneron, MSD, Pfizer, Daiichi Sankyo, and Ultragenyx. Consultancy for Amgen, Sanofi, Regeneron, Pfizer, Viatris, Abbott, AstraZeneca, Lilly, Kowa Pharmaceutics, Novo Nordisk, Boehringer Ingelheim, Esperion, Cargene Therapeutics, Resverlogix, Novartis, Silence Therapeutics, NewAmsterdam Pharma, Scribe Therapeutics, CRISPR Therapeutics, VAXXINITY, Amarin, CSL Behring, Bayer, Cleerly Health, and Emendobio. Stock options in PEMI31, SCRIBE, and New Amsterdam Pharma.
LB has received a consulting fee from Novartis, and conference grants from Linc Medical, Amryt and Ultragenyx.
MMAW, RA, EHW, PP, SW, FdL, AD, BJ: none declared.
Funding
None.
Study approval
This study was not approved by an Ethics Committee as data from routine clinical care were collected respectively, however, it was registered as an audit under the Imperial College Healthcare NHS Trust Audit Committee (CLB_06).
Editors’ note
The equations and supplementary materials referred to in this article are available for download from the online version of this article.
References
1. Ray KK, Wright RS, Kallend D et al.; ORION-10 and ORION-11 Investigators. Two phase 3 trials of inclisiran in patients with elevated LDL cholesterol. N Engl J Med 2020;382:1507–19. https://doi.org/10.1056/NEJMoa1912387
2. Sabatine MS, Giugliano RP, Keech AC et al.; FOURIER Steering Committee and Investigators. Evolocumab and clinical outcomes in patients with cardiovascular disease. N Engl J Med 2017;376:1713–22. https://doi.org/10.1056/NEJMoa1615664
3. Silverman MG, Ference BA, Im K et al. Association between lowering LDL-C and cardiovascular risk reduction among different therapeutic interventions: a systematic review and meta-analysis. JAMA 2016;316:1289–97. https://doi.org/10.1001/jama.2016.13985
4. Ray KK, Troquay RPT, Visseren FLJ et al. Long-term efficacy and safety of inclisiran in patients with high cardiovascular risk and elevated LDL cholesterol (ORION-3): results from the 4-year open-label extension of the ORION-1 trial. Lancet Diabetes Endocrinol 2023;11:109–19. https://doi.org/10.1016/S2213-8587(22)00353-9
5. Raal FJ, Kallend D, Ray KK et al.; ORION-9 Investigators. Inclisiran for the treatment of heterozygous familial hypercholesterolemia. N Engl J Med 2020;382:1520–30. https://doi.org/10.1056/NEJMoa1913805
6. Wright RS, Raal FJ, Koenig W et al. Inclisiran administration potently and durably lowers LDL-C over an extended-term follow-up: the ORION-8 trial. Cardiovasc Res 2024;120:1400–10. https://doi.org/10.1093/cvr/cvae109
7. Makhmudova U, Schatz U, Perakakis N et al. High interindividual variability in LDL-cholesterol reductions after inclisiran administration in a real-world multicenter setting in Germany. Clin Res Cardiol 2023;112:1639–49. https://doi.org/10.1007/s00392-023-02247-8
8. Mulder JWCM, Galema-Boers AMH, Roeters van Lennep JE. First clinical experiences with inclisiran in a real-world setting. J Clin Lipidol 2023;17:818–27. https://doi.org/10.1016/j.jacl.2023.09.005
9. Naoum I, Saliba W, Aker A, Zafrir B. Lipid-lowering therapy with inclisiran in the real-world setting: initial data from a national health care service. J Clin Lipidol 2024;18:e809–e816. https://doi.org/10.1016/j.jacl.2024.05.003
10. Iqbal S, Sabbour HM, Ashraf T, Santos RD, Buckley A. First report of inclisiran utilization for hypercholesterolemia treatment in real-world clinical settings in a Middle East population. Clin Ther 2024;46:186–93. https://doi.org/10.1016/j.clinthera.2023.12.003
11. Padam P, Barton L, Wilson S et al. Lipid lowering with inclisiran: a real-world single-centre experience. Open Heart 2022;9:e002184. https://doi.org/10.1136/openhrt-2022-002184
12. Bürkner P-C. brms: an R package for Bayesian multilevel models using Stan. J Stat Soft 2017;80:1–28. https://doi.org/10.18637/jss.v080.i01
13. Goodrich B, Gabry J, Ali I, Brilleman S. rstanarm: Bayesian applied regression modelling (arm) via Stan. R package version 2.32.1, 2024. Available from: https://mc-stan.org/rstanarm
14. Vehtari A, Gelman A, Gabry J. Practical Bayesian model evaluation using leave-one-out cross-validation and WAIC. Stat Comput 2017;27:1413–32. https://doi.org/10.1007/s11222-016-9696-4
15. Huo Y, Lesogor A, Lee CW et al. Efficacy and safety of inclisiran in Asian patients: results from ORION-18. JACC: Asia 2024;4:123–34. https://doi.org/10.1016/j.jacasi.2023.09.006
16. Mach F, Baigent C, Catapano AL et al.; ESC Scientific Document Group. 2019 ESC/EAS guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J 2020;41:111–88. https://doi.org/10.1093/eurheartj/ehz455
17. Ray KK, Molemans B, Schoonen WM et al. EU-wide cross-sectional observational study of lipid-modifying therapy use in secondary and primary care: the DA VINCI study. Eur J Prev Cardiol 2021;28:1279–89. https://doi.org/10.1093/eurjpc/zwaa047
18. Sabatine MS, Wiviott SD, Im K, Murphy SA, Giugliano RP. Efficacy and safety of further lowering of low-density lipoprotein cholesterol in patients starting with very low levels: a meta-analysis. JAMA Cardiol 2018;3:823–8. https://doi.org/10.1001/jamacardio.2018.2258
19. Ray KK, Raal FJ, Kallend DG et al. Inclisiran and cardiovascular events: a patient-level analysis of phase III trials. Eur Heart J 2022;44:129–38. https://doi.org/10.1093/eurheartj/ehac594

