Highlights of the recent American College of Cardiology (ACC) meeting, held in New Orleans, USA, on 2nd – 5th April 2011, included the promise of new, less invasive methods that might be an alternative to both aortic and mitral valve surgery. Long-term results from the STICH trial also suggest that CABG may have a role for patients with heart failure and ischaemic heart disease.
PARTNER: transcatheter valves just as good as surgery for high risk aortic stenosis
Transcatheter aortic valve implantation (TAVI) is just as effective at reducing mortality as surgery for severe aortic stenosis in elderly patients whose age and overall health posed high risks for conventional surgery, according to the results of the PARTNER (Placement of AoRTic TraNscathetER Valve trial). However, stroke rates were higher in the trancatheter group.
The transcatheter approach involves delivering a bioprosthetic valve to its target location with a catheter using either transfemoral access or trans-apical access (through the ribs) if peripheral arteries are not large enough.
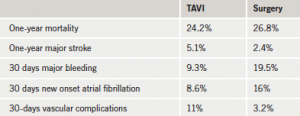
One-year results from the cohort A of the trial (table 1), in which 699 elderly patients with severe aortic stenosis who were randomised to either TAVI or conventional surgery, showed that the transcatheter approach was non-inferior to traditional surgical aortic-valve replacement in terms of all-cause mortality. Major strokes, however, were higher in the catheter-treated group. Major bleeding was far more common in the surgery group, as was new-onset atrial fibrillation whereas vascular complications were higher in the transcatheter group. Symptom improvement was similar in the two groups at one year.
Principal investigator Dr Craig Smith (Columbia University, New York, USA) said: “Surgical aortic valve replacement is one of the most effective operations surgeons offer, and transcatheter aortic valve replacement is the most exciting new treatment for aortic stenosis in the past two to three decades.”
“These results clearly show that the transcatheter approach is an excellent alternative to surgery in high-risk patients,” he continued. “Recommendations to individual patients will need to weigh the appeal of avoiding open-heart surgery, with its known risks, against less invasive transcatheter approach, with different and less well understood risks. Future trials will help delineate the role of transcatheter aortic valve replacement in intermediate risk patients.”
Transcatheter approach also shows cost-effectiveness in surgery-ineligible cohort
In a second part of the PARTNER trial involving patients with severe aortic stenosis who were not candidates for surgery (cohort B), transcatheter aortic valve replacement was linked to a 20% absolute benefit in survival at one year compared to medical therapy. These results were reported at last year’s TCT meeting in the US.
New data from this cohort, presented at the ACC meeting, suggested that the transcatheter valve replacement was cost effective in this group of patients.
Lead investigator, Dr Matthew Reynolds (Harvard Clinical Research Institute, Boston, USA) reported that the initial cost for transcatheter valve replacement, along with care before and after the procedure, was approximately US$78,000, with the commercial cost for the new valve system estimated at about US$30,000.
A higher rate of hospitalisation in the standard care group, however, led to costs during the first 12 months of around US$23,000, which partially offset the initial high cost of the trancatheter procedure.
The investigators projected a gain in life expectancy of approximately 1.9 years for the transcatheter valve replacement group, at an incremental cost-effectiveness ratio of about US$50,200 for each additional year of life gained, or approximately US$62,000 per each quality-adjusted life year gained. Dr Reynolds said this was “within accepted values for commonly used cardiovascular technologies.” He added: “Our perspective is that the costs of this intervention, even in this elderly and extremely high-risk population, are justified by the very significant benefits, and that the ratio of costs to benefits stacks up well compared with other therapies the US health care system pays for today.”
EVEREST II: MitraClip™ an option for mitral regurgitation patients unsuitable for surgery
Percutaneous repair of the mitral valve with the new MitraClip™ device was significantly less effective at reducing mitral regurgitation than surgery, but the percutaneous procedure was safer and resulted in similar improvements in clinical outcomes, latest results from EVEREST II (Endovascular valve edge-to-edge repair study) show.
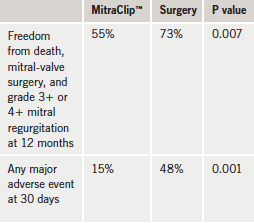
Discussants of the study at an ACC press conference said the clip would be useful for mitral regurgitation patients who were too high risk to undergo surgery. The trial randomised 279 patients with moderately severe or severe (grade 3+ or grade 4+) mitral regurgitation to undergo either percutaneous or surgical repair of the mitral valve. The primary composite end point of freedom from death, from surgery for mitral-valve dysfunction, and from grade 3+ or 4+ mitral regurgitation at 12 months was significantly better in the surgery group
(table 1). The primary safety end point of adverse events at 30 days was significantly reduced in the MitraClip™ group, but this was primarily accounted for by a reduction in blood transfusions.
Commenting on the study, cardiac surgeon Dr Steven Bolling (University of Michigan Cardiovascular Center, Ann Arbor, USA), noted that the clip left many more patients with grade 2+ or more regurgitation. Saying he “wouldn’t be happy to leave the operating room with this situation”, Dr Bolling added that residual mitral regurgitation normally predicts adverse long-term clinical outcomes.
STICH: CABG does appear beneficial in heart failure patients
Long-term results of the STICH (Surgical Treatment for Ischaemic Heart Failure) trial suggest that there may be a benefit after all in performing coronary artery bypass grafting (CABG) in patients with heart failure and ischaemic heart disease.
The trial assigned 1,200 patients with coronary artery disease and heart failure to either CABG plus the best possible medical therapy to aggressive medical therapy alone.
As expected, CABG was associated with an early risk of death as a result of the surgical intervention itself in the study but this disadvantage for surgery disappeared after two years post-procedure.
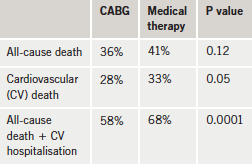
Five-year results (see table 1), presented at the ACC meeting, showed a trend towards a reduced risk of death from any cause in the CABG group. Cardiovascular death and the combined risk of death from any cause plus hospitalisation for heart disease were both significantly lower in the CAGB group.
Rheos® – an implantable device to control blood pressure
A new implantable device helps patients with severe and uncontrolled hypertension achieve and maintain target blood pressure levels, according to a new study.
The Rheos® device is implanted just below the collarbone, like a pacemaker, and delivers four to six volts of electricity to the carotid arteries. The pulses mimic a spike in blood pressure that activates the carotid baroreflex, causing the blood pressure to drop.
In the phase III study, the device was implanted in 265 patients with resistant hypertension of >160/80 mmHg who were taking at least three antihypertensives. The patients were then randomised to two groups. The first group had the device activated for the study’s full 12-month duration, and the second group had the device programmed to start activation at six months. Patients were seen monthly and those who had not reached the target systolic blood pressure of 140 mmHg had their device adjusted on an individual basis to assert more voltage and so further reduce blood pressure.
The trial did not meet all its end points, but blood pressures were generally lower in the active treatment group. Short-term acute response (at least a 10 mmHg reduction in systolic pressure at six months) was achieved in 65% of patients in the active treatment group, but the control group had a higher response than expected, with 45% of patients achieving the goal.
In the first group, systolic pressure decreased to target levels for 41% of patients after six months and 54% after 12 months. In the second group, 21% achieved target pressures during the control phase, and 46% were in the target range after six months of treatment.
Presenting the study, Dr John Bisognano (University of Rochester, USA) commented: “The system is safe, and its effect is as good as two or three drugs for people who are already taking five or six drugs and still can’t control their hypertension. It’s a good additional option for these patients.” He said the data were “encouraging”, but further studies were needed to define which group of patients benefits the most.
EXCELLENT: can clopidogrel be stopped six months after
drug-eluting stent?
Some patients may be able to stop clopidogrel six months following drug-eluting-stent implantation, according to the results of the EXCELLENT study. Although the 1,443-patient study was not powered for hard clinical end points, it did show a similar rate of target vessel failure among patients treated with six- and 12-months of clopidogrel.
Lead investigator Dr Hyeon-Cheol Gwon (Sungkyunkwan University School of Medicine, Seoul, Korea) said: “The take-home message from our study is that, at least in low-risk, nondiabetic patients treated with second-generation drug-eluting stents, we can safely discontinue clopidogrel at about six months, especially if the patient is at high risk for bleeding or is anticipating some surgery”.
But he added that the data should only be considered hypothesis-generating at this point, and larger trials with harder end points were needed to confirm the results.
Commenting on the trial, Dr Sanjay Kaul (Cedars Sinai Medical Center, Los Angeles, USA) noted that there was no significant reduction in the risk of major bleeding, which provided little support for stopping treatment at six months.
Major results of the trial showed the composite end point of cardiac death, myocardial infarction, or target vessel revascularisation at 12 months occurred in 4.7% of those treated with dual antplatelet therapy for six months, and 4.4% of those treated for 12 months, a result that satisfied non-inferiority criteria. However, diabetes patients had more than a three-fold risk of the end point in the six-month treatment group.
OSCAR: which antihypertensive best in elderly?
Treating elderly hypertensive patients with a combination of an angiotensin II receptor blocker (ARB) and a calcium blocker leads to similar rates of cardiovascular events and death compared to therapy with a high-dose ARB alone. This was the main result from the OSCAR study, conducted in a Japanese population.
For the study, 1,164 high-risk elderly hypertension patients unable to manage their high blood pressure through standard-dose monotherapy with the ARB olmesartan, were randomised to receive either high-dose olmesartan (40 mg per day) or a calcium blocker (either amlodipine or azelnidipine) combined with olmesartan 20 mg per day.
At 36 months of follow-up, blood pressure was reduced significantly more in the combination group (by 2.4 mmHg systolic and 1.7 mmHg diastolic). However, no significant difference was seen between the two groups in terms of clinical events – defined as cerebrovascular disease, coronary artery disease, heart failure, other atherosclerotic disease, diabetes complications, the deterioration of renal function, and all-cause death (table 1).
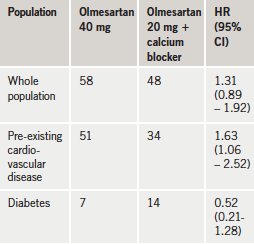
On subgroup analysis, however, patients who had pre-existing cardiovascular disease did have fewer events if on combination treatment. But another subgroup analysis including only patients with diabetes showed a higher incidence of the primary end point in the combination therapy group.
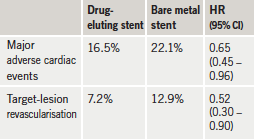
ISAR CABG: drug-eluting stents reduce repeat revascularisation in saphenous vein grafts
Use of drug-eluting stents was associated with fewer clinical events than bare metal stents when treating saphenous vein graft lesions, results of the ISAR CABG (Is Drug-Eluting Stenting Associated with Improved Results in Coronary Artery Bypass Grafts?) trial showed.
Saphenous vein grafts are used extensively in CABG surgery, but these grafts often develop atherosclerotic disease, requiring another revascularisation procedure.
Presenting the ISAR-CABG trial, Dr Julinda Mehilli (German Heart Center, Munich, Germany) explained that drug-eluting stents had proven superior to bare metal stents in all coronary artery lesion subsets examined, but there was a lack of data in saphenous vein graft lesions.
The ISAR-CABG trial enrolled 610 patients who had previously undergone CABG surgery, and had subsequently developed at least one lesion in their saphenous vein graft. They were randomised to receive either a drug-eluting stent or a bare metal stent.
Results (table 1) showed a reduced rate of major adverse cardiac events (death, myocardial infarction, target-lesion revascularisation) at one-year in the drug-eluting stent group. The benefit was primarily due to a significantly lower rate of target-lesion revascularisation. There was no significant difference in the rates of death or myocardial infarction.
RIVAL: radial versus femoral access for stenting in ACS patients
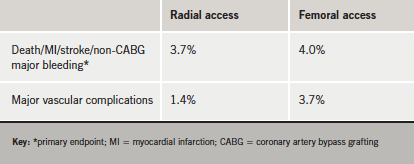
Stenting with radial access was not associated with a reduction in major clinical events compared with femoral access in acute coronary syndrome (ACS) patients, although the newer approach did appear to reduce large vascular-access complications in the 7,000-patient RIVAL (Radial versus Femoral Access for Coronary Intervention) study. 
In addition, subgroup analysis suggested a benefit of the radial approach in patients with ST-elevation myocardial infarction (STEMI), and those treated at centers that perform a high volume of radial access procedures.
Presenting the results (table 1), Dr Sanjit Jolly (McMaster University, Hamilton, Canada) said that the study showed both radial and femoral procedures to be safe and effective, but that radial access might be preferable because of the lower rates of vascular complications.
In an editorial accompanying publication of the study in the April 4th issue of The Lancet (Lancet 2011; DOI:10.1016/S0140-6736(11)60469-8) Drs Carlo Di Mario and Nicola Viceconte (Royal Brompton Hospital, London) suggest that operators with a high workload of acute procedures should consider retraining in transradial PCI, and all new interventionalists should be taught the transradial approach. But they add that the femoral approach may still be more suitable when large guiding catheters are required and prolonged procedural time is expected for complex lesions.
In the RIVAL study, major bleeding was not different in the primarily analysis, but a post hoc exploratory analysis using the ACUITY definition of bleeding did show reduced bleeding with the radial approach. This was said to be because the ACUITY definition includes large access-site haematomas, which is a vascular-site complication.

