A 27-year-old Arab woman with history of seronegative rheumatoid arthritis for four years presented with a three-day history of multiple joint pains and swelling, along with fever. Clinically, she had bilateral rheumatoid hand deformities with signs of active arthritis of elbows, hands and knee joints. She was in sinus rhythm with normal cardiac examination. She was treated with steroids and was responding well except for mild fever, for which an echocardiogram was requested to rule out infective endocarditis.
Images from her transthoracic and transoesophageal echocardiogram showed two large (2 cm2) left atrial nodular masses; one attached to the atrial surface of the anterior mitral leaflet tip and another free-floating in the left atrium, but not causing any mitral valve obstruction (figure 1). The mitral leaflets appeared normal with no regurgitation and no masses were noted in other chambers or valves including atrial appendages. A differential diagnosis of vegetations, thrombus or tumour mass was considered. Three sets of blood cultures were negative. A thrombophilia screen demonstrated markedly raised IgG anticardiolipin, β2 glycoprotein I antibodies and a positive lupus anticoagulant. Her anti-nuclear antibody and anti-double-stranded DNA were positive, confirming the diagnosis of systemic lupus erythematosus (SLE). Her anti-cyclic citrullinated peptide was positive, suggesting a diagnosis of ‘rhupus’, an overlap between rheumatoid arthritis and SLE.1
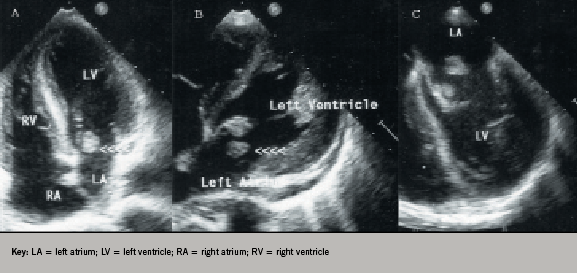
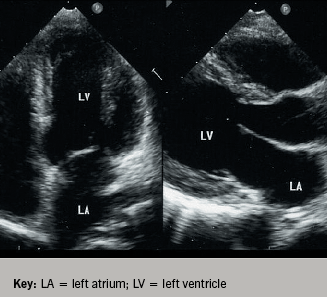
She also had an incidental finding of bilateral external jugular vein thrombosis detected during computed tomography of neck, chest and abdomen. The patient was managed with intravenous heparin followed by high intensity oral warfarin. Her follow-up echocardiograms showed complete resolution of both masses by four weeks without any systemic embolism (figure 2). Her antiphospholipid antibodies along with lupus anticoagulant repeated at three months were positive, confirming secondary antiphospholipid syndrome according to Sapporo criteria.2
Discussion
Antiphospholipid syndrome (APS) is an autoimmune disease characterised by antiphospholipid (aPL) antibodies and at least one clinical manifestation, the most common being venous or arterial thrombosis and recurrent fetal loss. The syndrome occurs in isolation (primary APS) or in association with connective tissue diseases (secondary APS), particularly SLE.2 Heart valve lesions (vegetations, valve thickening and dysfunction) are frequent in APS, independent of SLE.2
Review of literature suggests that there are two distinct types of vegetations in patients with APS associated with or without systemic emboli. Libman-Sacks vegetations3 described specifically in SLE are sterile fibrofibrinous masses that are typically sessile, wart-like (verrucoid) and small, varying from pinhead size to 3–4 mm. In addition, Roldan et al.4 have noted that thrombotic vegetations occur in lupus patients due to a hypercoagulable state and that these vegetations (also called non-bacterial thrombotic endocarditis) cannot be clearly differentiated from Libman–Sacks vegetations on echocardiography. There are many case reports describing the occurrence of thrombotic vegetations in patients with APS either as single or multiple masses, which usually resolve on anticoagulation therapy.5,6
In our patient, the occurrence of free-floating mass in association with an isolated mitral valve leaflet mass, along with evidence of thrombosis at another site and typical laboratory abnormalities, favoured the diagnosis of intracardiac thrombi in a patient with APS. Free-floating ball thrombi are usually seen in patients with rheumatic mitral stenosis and, to our knowledge, this has never been described in a patient with APS and rhupus. Treatment of thrombotic vegetations in patients with APS without significant valve dysfunction is controversial. Individual case reports5,6 have noted favourable response to anticoagulation, as seen in our patient. In contrast, long-term follow-up studies have reported that anticoagulant and/or antiplatelet therapy are ineffective in terms of valvular lesion regression.7
In conclusion, this case illustrates that intracardiac thrombi may precede other manifestations of APS. It also highlights the need to screen young patients with intracardiac masses of unknown aetiology for aPL antibodies.
Conflict of interest
None declared.
References
- Amezcua-Guerra LM, Springall R, Marquez-Velasco R, Gomez-García L, Vargas A, Bojalil R. Presence of antibodies against cyclic citrullinated peptides in patients with ‘rhupus’: a cross-sectional study. Arthritis Res Ther 2006;8:R144. (doi: 10.1186/ar2036)
- Miyakis S, Lockshin MD, Atsumi T et al. International consensus statement on an update of the classification criteria for definite antiphospholipid syndrome (APS). J Thromb Haemost 2006; 4:295–306. (doi: 10.1111/j.1538-7836.2006.01753.x)
- Hojnik M, George J, Ziporen L, Shoenfeld Y. Heart valve involvement (Libman–Sacks endocarditis) in the antiphospholipid syndrome. Circulation 1996;93:1579–87.
- Roldan CA, Shively BK, Crawford MH. An echocardiographic study of valvular heart disease associated with systemic lupus erythematosus. N Engl J Med 1996;335:1424–30. (doi: 10.1056/NEJM199611073351903)
- Agirbasli MA, Hansen DE, Byrd III BF. Resolution of vegetations with anticoagulation after myocardial infarction in primary antiphospholipid syndrome. J Am Soc Echocardiogr 1997;10:877–80. (doi: 10.1016/S0894-7317(97)70050-6)
- Salzberg SP, Nemirovsky D, Goldman ME, Adams DH. Aortic valve vegetation without endocarditis. Ann Thorac Surg 2009;88:267–9. (doi: 10.1016/j.athoracsur.2008.10.006)
- Turiel M, Sarzi-Puttini P, Peretti R et al. Five-year follow-up by transesophageal echocardiographic studies in primary antiphospholipid syndrome. Am J Cardiol 2005;96:574–9. (doi: 10.1016/j.amjcard.2005.04.022)
