Platelet activation is an important part of the pathophysiology of acute coronary syndromes. Inhibition of this by antiplatelet agents forms an important part of the management of this condition. Recently, there has been considerable interest in the variability of platelet response to these drugs and the need to tailor the dose of antiplatelet agents according to the response.
Here, we present a patient who had repeated episodes of stent thrombosis and was found to have decreased response to clopidogrel and aspirin (clopidogrel and aspirin resistance) with the use of the Accumetrics VerifyNowTM analyser. We have also reviewed the literature on this subject.
Our recommendation is that patients who have recurrent stent thrombosis or thrombotic events on dual antiplatelet agents should have their antiplatelet therapy response checked and have their antiplatelet agents tailored accordingly.
Introduction
Activated platelets play a pivotal role in the pathophysiology of acute coronary syndromes, and dual antiplatelet therapy with both aspirin and clopidogrel has become one of the cornerstones of their treatment.1 Similarly, dual antiplatelet therapy is mandated following percutaneous coronary intervention (PCI) with stent insertion to prevent stent thrombosis.2
Recently, there has been considerable interest in the phenomenon of inter-patient variability of clopidogrel response (sometimes termed ‘clopidogrel resistance’)3 and, to a lesser degree, the variability of aspirin response. This has led to interest in tailoring the dose of clopidogrel per patient according to the responsiveness of the platelets to the drug.4 In this report, we describe a patient who suffered recurrent ischaemic coronary events despite dual antiplatelet therapy, and we discuss the various implications of poor antiplatelet response and the current treatment options for similar patients.
Case report
A 41-year-old woman first presented to the rapid access chest pain clinic in May 2007 with a history of exertional angina. Her risk factors were hypertension, hypercholesterolaemia, and a positive family history of ischaemic heart disease (IHD). She had given up smoking three months earlier after a 21 pack-year history. She had a high body mass index of 31 kg/m2 and suffered from hypothyroidism. Exercise electrocardiography (ECG) was positive for symptoms and ECG changes in stage 2 of the Bruce protocol. Subsequent elective coronary angiography demonstrated minor diffuse disease in the left coronary system with a severe ostial right coronary artery (RCA) lesion (figure 1).
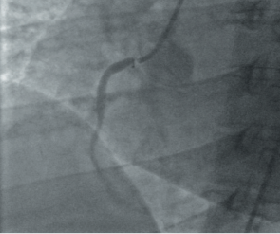
While waiting to undergo elective PCI to her ostial RCA lesion, she was admitted urgently with troponin-positive chest pain and infero-lateral ST-segment shift on an ECG two weeks after her initial presentation. Urgent PCI was performed and a 3 x 16 mm paclitaxel eluting (TaxusTM) stent was placed in the ostium of the RCA. She was discharged on dual antiplatelet therapy.
She presented again a month later, to a local district general hospital with an acute infero-lateral ST-elevation myocardial infarction (STEMI) for which she was treated with thrombolysis. There was no resolution of her ST segments or symptoms and she was transferred to our institution for rescue angioplasty. Angiography at this time revealed a patent RCA stent, but with a thrombotic occlusion of a small posterior left ventricular (PLV) branch. It was likely that she had subacute stent thrombosis, and the thrombolysis had largely dissolved the thrombus, but with a small remnant in the distal vessel. The vessel was wired and a balloon used to disrupt the thrombus. A stent was not deployed as the vessel was of small calibre. She was treated for 24 hours with the glycoprotein IIb/IIIa antagonist, tirofiban, and low molecular weight heparin, and her condition stabilised such that she was subsequently discharged on dual antiplatelet therapy.
She remained fairly well, until 18 months later, when she presented once again to her local hospital with chest pains. ECG on this occasion again showed an inferior STEMI. She was successfully treated with thrombolysis and was transferred for further assessment.
As part of a separate research protocol, platelet function studies were performed using the Accumetrics VerifyNowTM rapid platelet function analyser (RPFA). This showed a raised level of P2Y12 resistance units (PRU) of 363 after 30 days of clopidogrel 75 mg once daily, indicating a poor platelet response to clopidogrel. The same device also demonstrated significant aspirin resistance with aspirin resistance units (ARU) of 630.
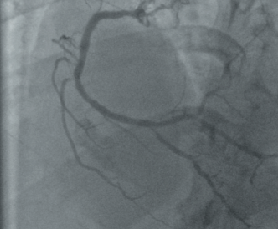
Repeat angiography now showed new pressure damping in left main stem (LMS) with mild disease only in the left anterior descending (LAD) and circumflex coronary vessels. There was new late ostial in-stent restenosis (amounting to 50% of the luminal caliber), and a tight ostial PLV lesion (figure 2). A pressure wire study to the LMS was performed and this was positive with a fractional flow reserve (FFRmyo) of 0.77 using adenosine via a central venous catheter. Intravascular ultrasound of the RCA stent revealed that it was well sized and well deployed and the degree of in-stent restenosis confirmed.
In view of the left main stem lesion and recurrent thrombotic episodes, she was referred for coronary artery bypass graft surgery, and following this is taking aspirin alone. She continues to do well following this intervention.
Discussion
There are a number of variables associated with stent thrombosis following PCI, including interventional factors (for example the complexity of the lesion, bifurcating lesions and poor stent expansion and apposition to the vessel wall) and patient variables (such as diabetes mellitus and renal failure).5 Increasingly, it has become clear that poor response to the antiplatelet agents aspirin and/or clopidogrel is associated with ischaemic events post PCI.6,7 A recent study has indicated that the incidence of definite/probable stent thrombosis at six months in patients undergoing PCI with drug-eluting stents and dual antiplatelet therapy resistance (as in our case above) is over 13%.6 Similarly, reduced clopidogrel activity, as measured using Accumetrics VerifyNow RPFA, has recently been shown to be independently predictive of cardiovascular death and nonfatal myocardial infarction 12 months following stent insertion.8
While the gold-standard test of platelet activity, optical platelet aggregometry, has been extensively used to assess the response to clopidogrel and aspirin in research protocols, it is a complex and time-consuming technique, requiring a high degree of expertise. It is unlikely, therefore, to find widespread acceptance as a routine clinical tool. The recent development of simple near-patient platelet function analysers, such as the VerifyNow RPFA, allows rapid and reproducible assessment of platelet response to aspirin and clopidogrel.9 There is also an increasing body of evidence indicating that poor RPFA results might predict clinical outcome, however, as yet, there are no definite guidelines providing a clinical definition of clopidogrel or aspirin resistance. It has been suggested that an aspirin resistance unit (ARU) level >550 indicates clinically important hyporesponsiveness, and this level has been shown to predict post-PCI myonecrosis.10 Two recent papers have identified the clinically relevant cut-off point of clopidogrel resistance to predict subsequent negative clinical events for RPFA PRU as 23511 and 240.12 As the weight of evidence supporting the use of near-patient tests for this purpose increases, interventional cardiologists need to decide whether to apply this technology in their own departments.
An ongoing issue is that if patients are identified as non-responders to clopidogrel, there is no current consensus on how best to manage their risk of recurrent ischaemic events. However, there is increasing evidence that in poor responders, increasing the clopidogrel dose from 75 mg to 150 mg daily improves antiplatelet activity,13,14 and that the increased doses may reduce clinical adverse events.15
This case illustrates the importance of antiplatelet resistance and we suggest that all patients with acute or subacute stent thrombosis should have their stent investigated using intravascular ultrasound to exclude a mechanical cause for the thrombosis. If none is found, platelet function testing using a validated method should be undertaken to exclude significant antiplatelet hyporesponsiveness.
Conflict of interest
None declared.
References
- Anderson JL, Adams CD, Antman EM et al. ACC/AHA 2007 guidelines for the management of patients with unstable angina/non-ST-elevation myocardial infarction: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines for the Management of Patients With Unstable Angina/Non-ST-Elevation Myocardial Infarction) developed in collaboration with the American College of Emergency Physicians, the Society for Cardiovascular Angiography and Interventions, and the Society of Thoracic Surgeons endorsed by the American Association of Cardiovascular and Pulmonary Rehabilitation and the Society for Academic Emergency Medicine. J Am Coll Cardiol 2007;50:e1–e157. (doi: 10.1016/j.jacc.2007.02.013)
- King SB, Smith SC, Hirshfeld JW et al. 2007 focused update of the ACC/AHA/SCAI 2005 Guideline Update for Percutaneous Coronary Intervention: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines: (2007 Writing Group to Review New Evidence and Update the 2005 ACC/AHA/SCAI Guideline Update for Percutaneous Coronary Intervention). Circulation 2008;117:261–95. (doi: 10.1161/CIRCULATIONAHA.107.188208)
- Serebruany VL, Steinhubl SR, Berger PB et al. Variability in platelet responsiveness to clopidogrel among 544 individuals. J Am Coll Cardiol 2005;45:246–51. (doi: 10.1016/j.jacc.2004.09.067)
- Bonello L, Camoin-Jau L, Arques S et al. Adjusted clopidogrel loading doses according to vasodilator-stimulated phosphoprotein phosphorylation index decrease rate of major adverse cardiovascular events in patients with clopidogrel resistance: a multicenter randomized prospective study. J Am Coll Cardiol 2008;51:1404–11. (doi: 10.1016/j.jacc.2007.12.044)
- Kuchulakanti PK, Chu WW, Torguson R et al. Correlates and long-term outcomes of angiographically proven stent thrombosis with sirolimus- and paclitaxel-eluting stents. Circulation 2006;113:1108–13. (doi: 10.1161/CIRCULATIONAHA.105.600155)
- Gori AM, Marcucci R, Migliorini A et al. Incidence and clinical impact of dual nonresponsiveness to aspirin and clopidogrel in patients with drug-eluting stents. J Am Coll Cardiol 2008;52:734–9. (doi: 10.1016/j.jacc.2008.05.032)
- Buonamici P, Marcucci R, Migliorini A et al. Impact of platelet reactivity after clopidogrel administration on drug-eluting stent thrombosis. J Am Coll Cardiol 2007;49:2312–17. (doi: 10.1016/j.jacc.2007.01.094)
- Marcucci R, Gori AM, Paniccia R et al. Cardiovascular death and nonfatal myocardial infarction in acute coronary syndrome patients receiving coronary stenting are predicted by residual platelet reactivity to ADP detected by a point-of-care assay: a 12-month follow-up. Circulation 2009;119:237–42. (doi: 10.1161/CIRCULATIONAHA.108.812636)
- Godino C, Mendolicchio L, Figini F et al. Comparison of VerifyNow-P2Y12 test and Flow Cytometry for monitoring individual platelet response to clopidogrel. What is the cut-off value for identifying patients who are low responders to clopidogrel therapy? Thromb J 2009;7:4. (doi: 10.1186/1477-9560-7-4)
- Chen WH, Lee PY, Ng W, Tse HF, Lau CP. Aspirin resistance is associated with a high incidence of myonecrosis after non-urgent percutaneous coronary intervention despite clopidogrel pretreatment. J Am Coll Cardiol 2004;43:1122–6. (doi: 10.1016/j.jacc.2003.12.034)
- Price MJ, Endemann S. Prognostic significance of post-clopidogrel platelet reactivity assessed by a point-of-care assay on thrombotic events after drug-eluting stent implantation. Eur Heart J 2008;29:992–1000. (doi: 10.1093/eurheartj/ehn046)
- Marcucci R, Gori AM, Paniccia R et al. Cardiovascular death and nonfatal myocardial infarction in acute coronary syndrome patients receiving coronary stenting are predicted by residual platelet reactivity to ADP detected by a point-of-care assay: a 12-month follow-up. Circulation 2009;119:237–42. (doi: 10.1161/CIRCULATIONAHA.108.812636)
- Angiolillo DJ, Shoemaker SB, Desai B et al. Randomized comparison of a high clopidogrel maintenance dose in patients with diabetes mellitus and coronary artery disease: results of the Optimizing Antiplatelet Therapy in Diabetes Mellitus (OPTIMUS) study. Circulation 2007;115:708–16. (doi: 10.1161/CIRCULATIONAHA.106.667741)
- Aleil B, Jacquemin L, De Poli F et al. Clopidogrel 150 mg/day to overcome low responsiveness in patients undergoing elective percutaneous coronary intervention: results from the VASP-02 (Vasodilator-Stimulated Phosphoprotein-02) randomized study. J Am Coll Cardiol Cardiovasc Interv 2008;1:631–8. (doi: 10.1016/j.jcin.2008.09.004)
- Bonello L, Camoin-Jau L, Arques S et al. Adjusted clopidogrel loading doses according to vasodilator-stimulated phosphoprotein phosphorylation index decrease rate of major adverse cardiovascular events in patients with clopidogrel resistance: a multicenter randomized prospective study. J Am Coll Cardiol 2008;51:1404–11. (doi: 10.1016/j.jacc.2007.12.044)

