A non-fatal myocardial infarction (MI) is an inconvenience; a non-fatal stroke is a catastrophe. While this is a simplification, it draws attention to the fact that most patients with a non-ST-segment elevation MI (NSTEMI) do quite well. Conversely, most patients who suffer a non-fatal stroke suffer a massive change to their lives and their families’ lives, and experience a devastating change in their quality of life.
 As cardiologists we spend a great deal of our time, and a considerable amount of National Health Service (NHS) money, treating MI, and the improvements in care have been terrific over the last quarter century. Yet our understanding of stroke prevention has not changed very much. Hypertension, dyslipidaemia, atrial fibrillation (AF) and diabetes are among the major risk factors and, while treatment of many of these has improved dramatically, AF stands out as the one treatable area that is yet really to be developed.
As cardiologists we spend a great deal of our time, and a considerable amount of National Health Service (NHS) money, treating MI, and the improvements in care have been terrific over the last quarter century. Yet our understanding of stroke prevention has not changed very much. Hypertension, dyslipidaemia, atrial fibrillation (AF) and diabetes are among the major risk factors and, while treatment of many of these has improved dramatically, AF stands out as the one treatable area that is yet really to be developed.
The new National Institute for Health and Care Excellence (NICE) AF guideline1 goes a long way to addressing this. Paramount is the identification of individuals with AF and, when individuals are identified, the two cornerstones of AF treatment can be employed.
Cornerstones of AF treatment
Management of AF has two goals:
- Alleviation of symptoms.
- Prevention of stroke.
Professor Cowan and colleagues provide a beautifully written guidance on how to address symptoms, with rate control, rhythm control and sometimes with intervention.
But prevention of stroke is to most of us the most important reason for treating AF. Most patients can be rate controlled; some will be much better with rhythm control; and younger patients may have spectacular results with intervention. But the huge burden of AF, and its cost to individuals and to the nation, is its relationship with stroke.
Stroke burden and AF burden in the UK
About a million people live with the effects of stroke, with half of these dependent on other people for everyday activities.2 Around another million people in the UK have AF,3 and this incidence will likely double over the next 30 years.4 It is estimated that around 12,500 strokes are caused directly by AF in the UK each year,5 and the majority of these are preventable with appropriate anticoagulation.
Anticoagulation and stroke prevention in non-valvular AF
We have had the vitamin K antagonist warfarin for 50 years, as anticoagulation to prevent stroke in non-valvular AF. Aspirin offers little protection. Clopidogrel plus aspirin perhaps a little more, but until recently, warfarin has been the gold standard for anticoagulation. Things changed in 2009 with the publication of the RE-LY (Randomised Evaluation of Long-term Anticoagulation Therapy) trial, and there are now four major outcome studies using the new anticoagulants: dabigatran, rivaroxaban, apixaban and edoxaban.6-9 These major outcome trials compare well-managed warfarin with one of the non-vitamin K oral anticoagulant drugs (NOACs), for prevention of stroke and embolic events in patients with non-valvular AF.
Most of us were cautiously testing the water with these new drugs until the publication of the Lancet meta-analysis of these four major trials, online in December 2013, and in print in March 2014.10
Meta-analysis of the NOAC trials
Ruff and colleagues released their data in December 2013 with a meta-analysis of 71,683 patients from the four major trials of the NOACs.10 The meta-analysis showed a 19% relative risk reduction in stroke or systemic embolic event; a 10% reduction in all-cause mortality; a 50% reduction in haemorrhagic stroke and 14% reduction in major bleeds. Particularly noted was a 52% reduction in intracerebral haemorrhage. Warfarin was slightly safer in regard to incident gastrointestinal (GI) bleeding, although, in this regard, there appear to be differences between the individual NOACs. Thus, the meta-analysis would seem absolutely clear, that the new anticoagulants were much better than warfarin. Concern has been expressed about the risk of bleeding in patients taking NOACs, and the lack of an antidote for the currently available drugs. However, recent data from the major trials of each of the NOACs versus warfarin has shown that the risk of fatality associated with major bleeding is 30–50% lower with the NOACs than with warfarin.11-13
NICE exists, fundamentally, to balance available evidence to guide therapy for the population. Yet, the authors of the new NICE AF guideline were prevented from including the crucial meta-analysis data, which demonstrated significant benefits of the NOACs over warfarin. The NICE AF guideline was published in June 2014. The data from the meta-analysis was available in December 2013 and published three months before the NICE guideline. In March 2014, various stakeholders identified to the NICE AF writing team that the NOAC meta-analysis should be considered and included, but this was not permitted by NICE.
Why was the NOAC meta-analysis not included in the NICE guidance?
This seems to be a lost opportunity to address a real need for high-quality anticoagulation. It is likely that the authors of the new NICE AF guidance would have wished to use the NOAC meta-analysis data, but were prevented from doing so.
By implication, it seems then that warfarin control is satisfactory if a patient is in therapeutic range for more than 65% of the time. Yet, for the management of hypertension or lipids, blood pressure or cholesterol, being uncontrolled for a third of the time would be considered poor control. The NOACs offer better, safer anticoagulation at lower overall risk, proven in the meta-analysis of over 70,000 patients. So where do we stand in relation to other countries?
UK position in NOAC prescribing
Data recently available show that our use of NOACs is modest compared with most other European countries (figure 1).14 Germany is way out in front, with Belgium not far behind. The UK is using NOACs at about 10% of the level of the Germans. Implementation of the evidence from the Ruff meta-analysis could have redressed this, with likely clear benefits for the nation and for individuals with AF.
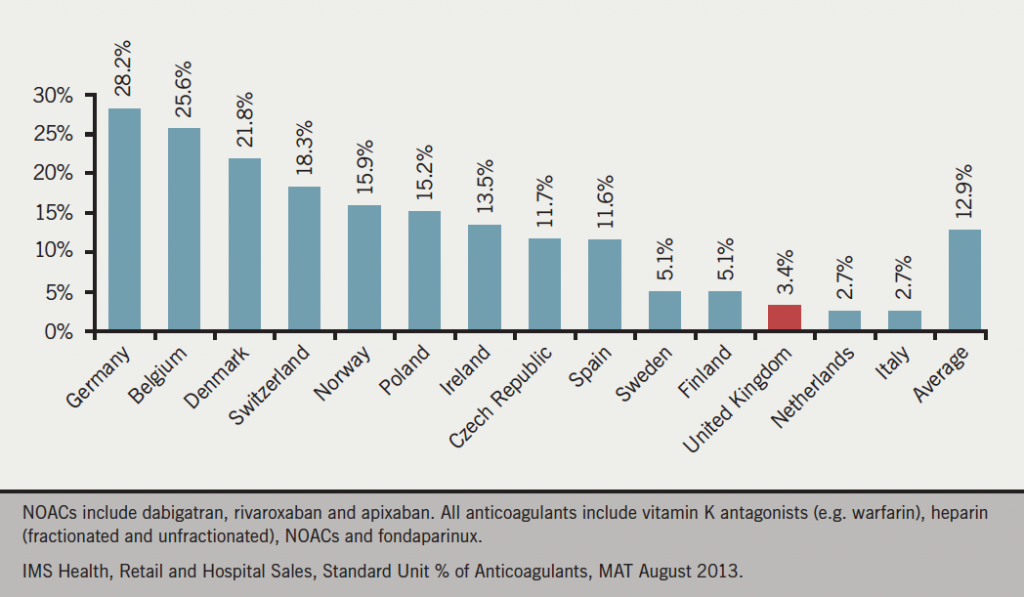
Interestingly, this is similar to the position of the UK with statins in 2002, where statin usage in the UK was modest compared with the coronary heart disease (CHD) burden as shown in figure 2.15
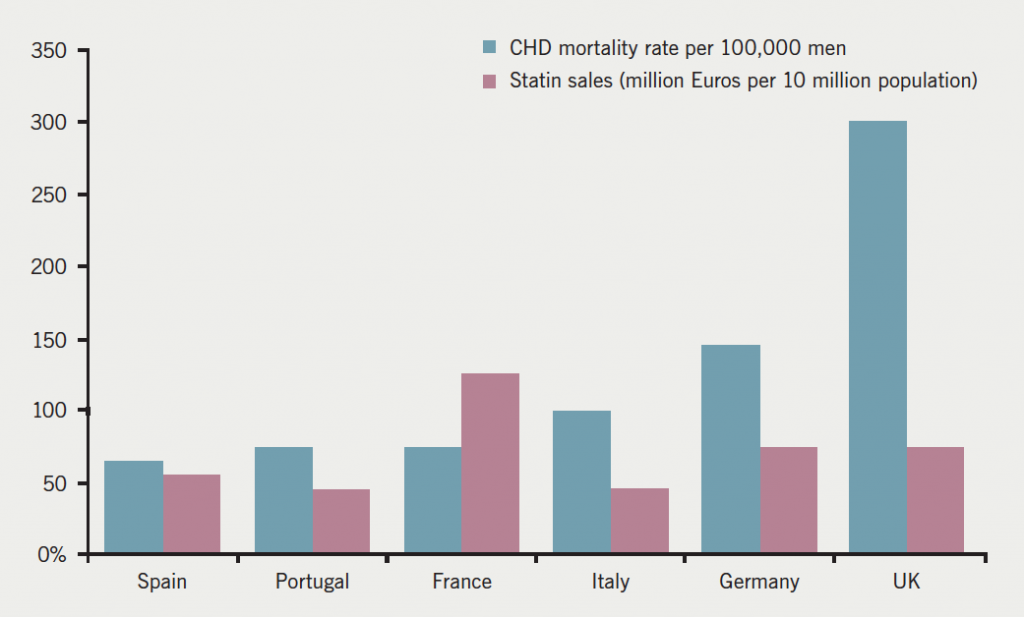
So what should we do?
It seems, to ourselves at least, that for patients with non-valvular AF, NOACs offer considerable superiority for stroke prevention compared with well-managed warfarin. In our own practices, the vast majority of patients prefer NOACs over warfarin. Indeed, some patients beg not to be commenced on warfarin because of its attendant risks, considerable inconvenience, discomfort, and lifestyle change. It seems, to us, to be an opportunity missed, to deny a population with substantial risk and substantial potential benefit, recommended access to these new drugs.
Conclusion
Like the Prussians at Waterloo, we in the UK may come along last but in great numbers. This is what happened with statins, where the terrific work of Sir Roger Boyle and others took Britain to the world-leading position in lipid-lowering therapy. NICE should address new separate guidance on the role of NOACs for stroke prevention in AF, and it is hoped that NICE will engage with this quickly, so that the million UK individuals with AF can be appropriately protected.
Conflict of interest
AB and DC have received honoraria for lecture fees from pharmaceutical companies whose products include the non-vitamin K oral anticoagulants. AD: none declared.
Editors’ note
A response from Chairman of the Guideline Development Group, Campbell Cowan et al. follows in this issue.
References
1. National Institute for Health and Care Excellence. Atrial fibrillation: the management of atrial fibrillation. CG180. London: NICE, June 2014. Available from: http://www.nice.org.uk/guidance/cg180
2. National Audit Office. Reducing brain damage: faster access to better stroke care. Report by the Comptroller and Auditor General. HC 452 Session 2005–2006. London: The Stationery Office, 2005. Available from: http://www.nao.org.uk/wp-content/uploads/2005/11/0506452.pdf
3. Brown V. Using the GRASP-AF audit tool to help prevent strokes in patients with AF. Presentation to NHS Alliance, 2012.
4. Go AS, Hylek EM, Phillips KA et al. Prevalence of diagnosed atrial fibrillation in adults: national implications for rhythm management and stroke prevention; the AnTicoagulation and Risk Factors in Atrial Fibrillation (ATRIA) study. JAMA 2001;285:2370–5. http://dx.doi.org/10.1001/jama.285.18.2370
5. NHS Improvement. Commissioning for stroke prevention in primary care – the role of atrial fibrillation. Leicester: NHS Improvement, 2009. Available from: http://www.atrialfibrillation.org.uk/files/file/AF_Commissioning_Guide.pdf
6. Connolly SJ, Ezekowitz MD, Yusuf S et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med 2009;361:1139–51. http://dx.doi.org/10.1056/NEJMoa0905561
7. Patel MR, Mahaffey KW, Garg J et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med 2011;365:883–91. http://dx.doi.org/10.1056/NEJMoa1009638
8. Granger CB, Alexander JH, McMurray JJ et al. Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med 2011;365:981–92. http://dx.doi.org/10.1056/NEJMoa1107039
9. Giugliano RP, Ruff CT, Braunwald E et al. Edoxaban versus warfarin in patients with atrial fibrillation. N Engl J Med 2013;369:2093–104. http://dx.doi.org/10.1056/NEJMoa1310907
10. Ruff CT, Giugliano RP, Braunwald E et al. Comparison of the efficacy and safety of new oral anticoagulants with warfarin in patients with atrial fibrillation: a meta-analysis of randomised trials. Lancet 2014;383:955–62. http://dx.doi.org/10.1016/S0140-6736(13)62343-0
11. Majeed A, Hwang HG, Connolly SJ et al. Management and outcomes of major bleeding during treatment with dabigatran or warfarin. Circulation 2013;128:2325–32. http://dx.doi.org/10.1161/CIRCULATIONAHA.113.002332
12. Hylek EM, Held C, Alexander JH et al. Major bleeding in patients with atrial fibrillation receiving apixaban or warfarin. The ARISTOTLE trial (Apixaban for Reduction in Stroke and Other Thromboembolic Events in Atrial Fibrillation): predictors, characteristics, and clinical outcomes. J Am Coll Cardiol 2014;63:2141–7. http://dx.doi.org/10.1016/j.jacc.2014.02.549
13. Piccini JP, Garg J, Patel MR et al. Management of major bleeding events in patients treated with rivaroxaban vs. warfarin: results from the ROCKET AF trial. Eur Heart J 2014;35:1873–80. http://dx.doi.org/10.1093/eurheartj/ehu083
14. IMS Health, MIDAS, August 2013. NOACs include dabigatran, rivaroxaban and apixaban. All anticoagulants include vitamin K antagonists (e.g. warfarin), heparin (fractionated and unfractionated), NOACs and fondaparinux.
15. Brady AJB. Britain: still the sick man of Europe? Br J Cardiol 2002;9:1–3. Available from: https://bjcardio.co.uk/2002/01/britain-still-the-sick-man-of-europe/

