Introduction
In this module, we will cover medical therapy for the treatment of valvular heart disease (VHD). Pharmacotherapy of VHD can be divided into two broad areas – supportive treatment and disease-modifying treatment. With the exception of a number of large-scale clinical trials in patients with aortic stenosis, we must rely on small studies and expert opinion. We will discuss medical therapy for the major valvular lesions, indications for anticoagulation in native valve disease, and finally examine recommendations for endocarditis prophylaxis – an area that has seen major changes in guidelines over the past decade.
Aortic stenosis
With the growing evidence that calcific aortic stenosis (AS) is not a “degenerative” process, but rather a process heavily influenced by inflammation, fibrosis, and subsequent calcification came the search for potential medical therapies to halt (or at least slow) progression of the disease. Similarities between the lesions of aortic stenosis and atherosclerosis, high crossover of risk factors amongst those developing atherosclerosis and AS, and results from observational studies and experimental models led to a number of large scale trials examining the role of statin therapy in AS.1–3
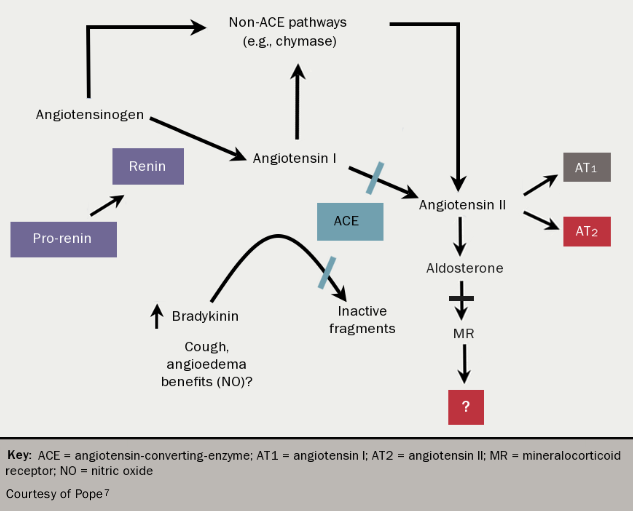
Unfortunately, these trials uniformly showed that statins (see figure 1) do not affect the progression of AS, although a reduction in ischaemic cardiovascular endpoints was seen in the largest study.2 This is likely to reflect the fact that whilst similar, the two atherosclerotic disease processes have important differences, perhaps most notably the advanced calcific response observed in the aortic valve. Strategies targeting calcification directly may be most likely to prove successful. For example, bisphosphonates reduce vascular calcification in experimental settings and these agents may therefore have beneficial effects within the aortic valve. Other potential disease modifying therapies are angiotensin converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs), as the renin-angiotensin-aldosterone system (RAAS) is upregulated at valve level in AS4 and beneficial effects have been seen with these agents in animal studies.5,6 (see figure 27). Population-based studies have shown improved outcomes in patients with all degrees of severity of AS using ACEIs and ARBs.8 However, in the Losartan Intervention for End Point Reduction in Hypertension (LIFE) study, neither the ARB losartan nor the beta blocker, atenolol, prevented the progression of aortic valve sclerosis.9
An alternative therapeutic approach may be to protect the myocardium from the effects of pressure overload in AS. For example, RAAS modulators and beta-blockers may exert beneficial effects by slowing the development of myocardial interstitial fibrosis, a marker of worse prognosis after aortic valve replacement (AVR),9 through down-regulation of transforming growth factor beta (TGF-β).10 Disappointingly, a trial of the aldosterone antagonist eplerenone in 65 patients did not show any benefit with regards to either disease progression or change in left ventricular function.11 However, the recent Ramipril in Aortic Stenosis (RIAS) trial randomised 100 patients with moderate or severe AS to ramipril or placebo, with a primary (non-clinical) endpoint of altered left ventricular mass over 12 months.12 There was a small but statistically significant difference in left ventricular mass in those allocated ramipril treatment, and a further trial examining the effect of ramipril on reduction of clinical endpoints is now planned.