The changing cardiovascular risk imposed by type 2 diabetes
Once upon a time, type 2 diabetes was considered a coronary heart disease (CHD) risk equivalent, meaning that the risk of a future CHD event was considered equal to patients with existing vascular disease. While this notion has been superseded, the risk for a range of vascular outcomes (myocardial infarction [MI], stroke or deaths due to other vascular diseases) appears approximately double in diabetes compared with the general population (figure 1).1 Less well appreciated is the observation that diabetes increases the risk of fatal events somewhat more than non-fatal, for reasons not well established. Finally, this doubling of cardiovascular disease (CVD) risk is an average and should not be applied equally to all diabetes patients, since CVD risk levels at the point of diabetes diagnosis are far less elevated. By contrast, once a patient has had several years of diabetes or exhibits some microvascular complication, such as albuminuria, low estimated glomerular filtration rate (eGFR) or moderate retinopathy, CVD risk levels can be elevated multi-fold.
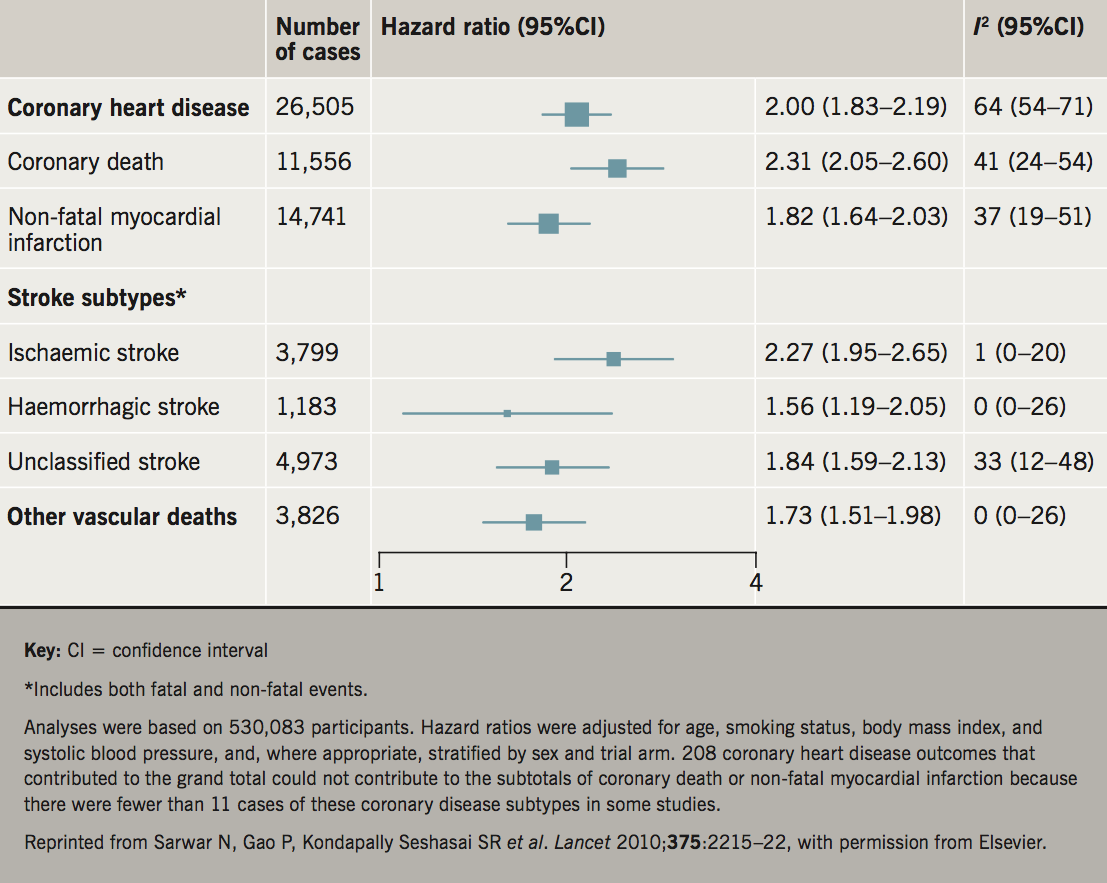
In high-income countries, over the last two or so decades a pattern of substantially reducing risks for MI and stroke in diabetes patients (relative to non-diabetes controls) has been noted, though, even now, such risks still remain far in excess of healthy controls.2 Consequently, recent attention has focused more on the excess risk for other ‘vascular’ outcomes in diabetes, such as heart failure and peripheral arterial disease. In a recent paper, these latter two outcomes were the most common first ‘vascular’ outcomes in diabetes patients in analysis of data from the UK, with MI and stroke3 being less frequent. We appear to have also been more successful at preventing non-fatal CVD events in diabetes patients,4 suggesting we lack full understanding of all the causes for premature CVD mortality in diabetes.
Hence, patterns for vascular risks in high-income countries are changing in diabetes, as earlier diagnosis and better CVD preventative treatment strategies are being employed in the care of diabetes patients, leading to less atherosclerotic events and lower CVD hospitalisations, but there is still much to do to achieve even better reductions in fatal outcomes, and outcomes like heart failure.
Relative risks vary most markedly by age of onset of diabetes
There is a widening appreciation that age of onset of type 2 diabetes matters to magnitude of risks for adverse outcomes and years lost due to diabetes, with younger-onset type 2 diabetes likely more pathogenic. To this end, we recently showed that individuals with younger-onset type 2 diabetes were much more obese, had higher glycaemia levels at diagnosis and worse lipids than those developing type 2 diabetes much later in life.5 Moreover, these differences persisted for several years post-diagnosis and could not be accounted for by differential glycaemia or lipid-lowering treatments, adding support for a more pathogenic phenotype at younger ages. At the other extreme, there is evidence that older patients with diabetes tend to be at much lower excess risk for CVD compared with their non-diabetes counterparts.6 Thus, age at diabetes diagnosis should be an important risk stratifier for diabetes management. There is also some evidence that women’s greater relative risk for CVD risk once they develop diabetes may also be related to greater average body mass index (BMI) levels needed in women to develop diabetes compared with their male counterparts.7
Risk factors driving excess CVD risk in type 2 diabetes
This topic has been well studied by many others, but some things remain far from certain. The following risk pathways are perturbed in diabetes, often well before the diagnosis of diabetes is made:
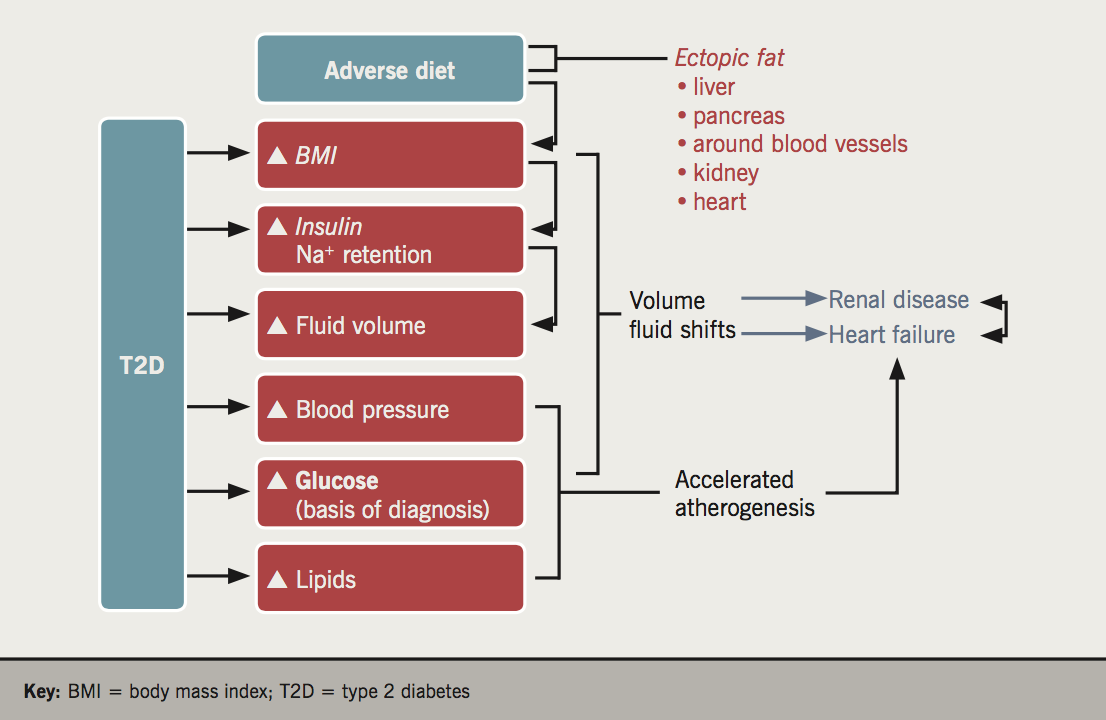
- Obesity – many neglect to consider obesity/excess fat as a risk factor for CVD in diabetes, forgetting that to develop diabetes, particularly at young ages, requires individuals to put on considerable excess fat. There is now ample evidence from higher-quality epidemiological studies linking obesity to CVD risk,8 and particularly striking risks for heart failure,9 with considerable parallel genetic evidence to support these associations being causal.10 There is also ample evidence demonstrating an adiposity pattern linked to diabetes – i.e. excess liver and other ectopic fat – is more pathogenic to CVD risk than excess fat accumulation in subcutaneous tissues.11
- Dyslipidaemia – the dyslipidaemia associated with type 2 diabetes is one of higher triglycerides, and lower high-density lipoprotein cholestrol (HDL-c), a pattern classically linked to insulin resistance. While, for years, researchers focused on increasing HDL-c to try to lessen risk, recent genetic evidence suggests, in fact, higher triglyceride rather than lower HDL-c is likely to be pathogenically linked to vascular risk.12 The higher triglyceride, which could be considered as another example of ectopic fat in diabetes (to add to excess liver, pancreatic and cardiac fat), stems predominantly from overproduction of triglyceride-rich lipoproteins in the liver.
- Hypertension – most patients with type 2 diabetes will develop hypertension at some point, with many having hypertension by the time they are diagnosed. It is also clear that hypertension is a risk factor for future diabetes, independent of BMI and other factors.13 Thus, while higher BMI partially drives greater hypertension in diabetes, other processes linked to insulin resistance/hyperinsulinaemia, or excess sodium and fluid retention, perhaps particularly in the extracellular compartments, must be relevant to hypertension risks in diabetes given rates are higher, even when matched to BMI-similar controls. Excess fat around blood vessels in type 2 diabetes may directly or indirectly (via cytokine release) disturb vascular tone in diabetes and be an additional contributor to hypertension risks in this condition.
- Thrombotic perturbances – linked to insulin resistance and the lipid abnormalities, there is ample evidence of diabetes being associated with a prothrombotic state.14 That noted, current trial evidence goes against a net benefit of aspirin in the primary prevention of CVD in diabetes, a finding that may be related to differences in platelet reactivity in diabetes. This is an area that, despite hundreds of studies, is not at all well understood.
- Qualitative and quantitative differences in body fluid status – as hinted at above, patients with diabetes are often fluid and sodium overloaded, and the processes that lead to this state are inadequately understood. There are many potential contributing pathways, including obesity per se, hyperinsulinaemia-driven sodium retention, plus the more recently proposed upregulation of sodium-glucose co-transporter 2 (SGLT-2) receptors in the kidneys,15 leading to both sodium and glucose re-absorption. Such excess fluid will be relevant to hypertension risks, but whether it also contributes to microvascular damage over and above effects of other risk factors is not well studied. Glomerular hyperfiltration, an early consequence of diabetes, is often linked to excess fluid volume, leading, if not corrected, to nephron death and, over time, a reduction in glomerular filtration rates as more and more nephrons die in this high-pressure state. Sodium and fluid excesses in extracellular and perhaps interstitial compartments may also be relevant to heart failure risks, making diabetes patients more susceptible to heart failure, as is now more widely recognised.
It should be apparent from the aforementioned discussions that some risk factor perturbances in type 2 diabetes drive atherogenesis, whereas others may more strongly promote renal and heart failure risks via fluid disturbances, which are less well studied (figure 2). This new way of thinking needs further development if we are to further reduce CVD risks, especially fatal outcomes, in patients with type 2 diabetes.
Is hyperglycaemia directly relevant to excess vascular risks in diabetes, and if so, to what extent?
One may be surprised to see hyperglycaemia not discussed above as a dominant risk factor for diabetes vascular complications. However, it should be clear that many of the above risk pathways are likely highly perturbed well before frank hyperglycaemia ensues. That noted, hyperglycaemia does add meaningfully to the vascular risk over and above other risk factors, which fits with the finding that frank diabetes but not pre-diabetes states, is predictive of CVD risk independently of other risk factors. In other words, only when glucose levels escalate into the diabetes range is the risk of vascular outcomes enhanced, as confirmed by recent meta-analyses of multiple cohorts,1 as well as a more recent elegant study from Denmark.16 It must also be the case that abnormalities in all the above pathways must be greater in diabetes versus those with pre-diabetes, so, not only are glucose levels higher, other factors (e.g. fluid shifts, lipid abnormalities, thrombotic aspects, etc.) will also be more abnormal in ways not fully documented, so that even in diabetes, one cannot say for certain to what extent glucose, or its co-travellers, influences CVD risk.
There are multiple direct mechanisms by which hyperglycaemia per se might drive blood vessel damage, as frequently reviewed.17 Notably, however, reducing glucose has been shown to have only modest effects in reducing risks of both macrovascular disease (mainly non-fatal)18 and microvascular disease,19 as reported in recent meta-analyses of intensive glucose-lowering trials, albeit conducted using the more established diabetes-lowering drugs. Furthermore, benefits of glucose lowering on macrovascular risks were calculated to be less than with lipid and blood pressure lowering,18 reinforcing the notion that management of other risk factors matters more to MI and stroke risk. Furthermore, more recent trial results with the SGLT-2 inhibiting agents,20 particularly in terms of reducing mortality and heart failure risk, has led researchers to revisit the excess fluid hypothesis in diabetes. These matters are discussed more in subsequent articles but they reinforce the notion that we remain a long way from fully understanding all pathways to CVD or heart failure risks in type 2 diabetes.
Markers of higher CVD risk in diabetes?
Once a diabetes patient has had a vascular event or documented vascular disease, he/she is at markedly elevated vascular risk of future events/premature mortality.21 The presence of microvascular complications, whether retinal, kidney disease or neuropathy, also signals excess vascular risk, and if two or three such complications are present then risks are even higher.22 This latter observation accords with the notion that small-vessel disease begets large-vessel disease, perhaps directly,23 but of course, overlapping risk factor processes also operate on both small and large blood vessels.
Risk scores for CVD risk are also plentiful for use in diabetes patients and some of these (e.g. ADVANCE risk score – http://www.advanceriskengine.com) also capture CVD risk contributions from earlier diabetes development or longer diabetes duration, presence or absence of microvascular complications, in addition to CVD risk from the usual risk factors (blood pressure, lipids), glycaemia control and lifestyle factors (smoking).
Summary
It is clear that diabetes imposes considerable premature vascular risks on individuals. In this short review, the well-established concept of multiple risk factors leading to excess diabetes risk is acknowledged, with confirmation of considerable successes in reducing CVD (non-fatal more than fatal) risks in diabetes. However, the less well appreciated age differentials in risk, whereby younger-onset type 2 diabetes is shown to be much more pathogenic, is better highlighted with a call for more aggressive management in these groups. In addition, a need for a greater focus on abnormal fluid volume shifts in diabetes is made, since such knowledge may further our ability to reduce risk for fatal CVD in diabetes, as well as renal decline and heart failure.
Key messages
- Risk of a range of vascular outcomes, e.g. myocardial infarction, stroke etc., is approximately doubled in patients with diabetes
- Relative risks may vary by age of onset of diabetes
- Aggressive management is warranted in these groups and there is a need for a greater focus on abnormal fluid volume shifts
Conflict of interest
NS has been a speaker and/or consulted for Amgen, AstraZeneca, Boehringer Ingelheim, Eli-Lilly, Janssen, Novo Nordisk, Roche Diagnostics and Sanofi. His unit has received a research grant from Boehringer Ingelheim, with funds administered by the University of Glasgow.
Other articles in this supplement include:
Introduction
2. Diabetes and cardiovascular risk in UK South Asians: an overview
3. Drugs for diabetes: the cardiovascular evidence base
4. Optimising cardiovascular risk reduction in diabetes
References
1. Emerging Risk Factors Collaboration, Sarwar N, Gao P et al. Diabetes mellitus, fasting blood glucose concentration, and risk of vascular disease: a collaborative meta-analysis of 102 prospective studies. Lancet 2010;375:2215–22. https:doi.org/10.1016/S0140-6736(10)60484-9
2. Gregg EW, Sattar N, Ali MK. The changing face of diabetes complications. Lancet Diabetes Endocrinol 2016;4:537–47. https://doi.org/10.1016/S2213-8587(16)30010-9
3. Dinesh Shah A, Langenberg C, Rapsomaniki E et al. Type 2 diabetes and incidence of a wide range of cardiovascular diseases: a cohort study in 1.9 million people. Lancet 2015;385:S86. https://doi.org/10.1016/S0140-6736(15)60401-9
4. Rawshani A, Rawshani A, Franzén S et al. Mortality and cardiovascular disease in type 1 and type 2 diabetes. N Engl J Med 2017;376:1407–18. https://doi.org/10.1056/NEJMoa1608664
5. Steinarsson AO, Rawshani A, Gudbjörnsdottir S, Franzén S, Svensson A-M, Sattar N. Short-term progression of cardiometabolic risk factors in relation to age at type 2 diabetes diagnosis: a longitudinal observational study of 100,606 individuals from the Swedish National Diabetes Register. Diabetologia 2018;61:599–606. https://doi.org/10.1007/s00125-017-4532-8
6. Tancredi M, Rosengren A, Svensson A-M et al. Excess mortality among persons with type 2 diabetes. N Engl J Med 2015;373:1720–32. https://doi.org/10.1056/NEJMoa1504347
7. Wannamethee SG, Papacosta O, Lawlor DA et al. Do women exhibit greater differences in established and novel risk factors between diabetes and non-diabetes than men? The British Regional Heart Study and British Women’s Heart Health Study. Diabetologia 2012;55:80–7. https://doi.org/10.1007/s00125-011-2284-4
8. Emerging Risk Factors Collaboration, Wormser D, Kaptoge S et al. Separate and combined associations of body-mass index and abdominal adiposity with cardiovascular disease: collaborative analysis of 58 prospective studies. Lancet 2011;377:1085–95. https://doi.org/10.1016/S0140-6736(11)60105-0
9. Rosengren A, Åberg M, Robertson J et al. Body weight in adolescence and long-term risk of early heart failure in adulthood among men in Sweden. Eur Heart J 2016;38:ehw221. https://doi.org/10.1093/eurheartj/ehw221
10. Lyall DM, Celis-Morales C, Ward J et al. Association of body mass index with cardiometabolic disease in the UK biobank: a Mendelian randomization study. JAMA Cardiol 2017;2:882–9. https://doi.org/10.1001/jamacardio.2016.5804
11. Yaghootkar H, Lotta LA, Tyrrell J et al. Genetic evidence for a link between favorable adiposity and lower risk of type 2 diabetes, hypertension, and heart disease. Diabetes 2016;65:2448–60. https://doi.org/10.2337/db15-1671
12. Stitziel NO. Human genetic insights into lipoproteins and risk of cardiometabolic disease. Curr Opin Lipidol 2017;28:113–19. https://doi.org/10.1097/MOL.0000000000000389
13. Cho NH, Kim KM, Choi SH et al. High blood pressure and its association with incident diabetes over 10 years in the Korean Genome and Epidemiology Study (KoGES). Diabetes Care 2015;38:1333–8. https://doi.org/10.2337/dc14-1931
14. Morel O, Jesel L, Abbas M, Morel N. Prothrombotic changes in diabetes mellitus. Semin Thromb Hemost 2013;39:477–88. https://doi.org/10.1055/s-0033-1343888
15. Lytvyn Y, Bjornstad P, Udell JA, Lovshin JA, Cherney DZI. Sodium glucose cotransporter-2 inhibition in heart failure: potential mechanisms, clinical applications, and summary of clinical trials. Circulation 2017;136:1643–58. https://doi.org/10.1161/CIRCULATIONAHA.117.030012
16. Vistisen D, Witte DR, Brunner EJ et al. Risk of cardiovascular disease and death in individuals with prediabetes defined by different criteria: the Whitehall II study. Diabetes Care 2018;41:899–906. https://doi.org/10.2337/dc17-2530
17. Shah MS, Brownlee M. Molecular and cellular mechanisms of cardiovascular disorders in diabetes. Circ Res 2016;118:1808–29. https://doi.org/10.1161/CIRCRESAHA.116.306923
18. Ray KK, Seshasai SRK, Wijesuriya S et al. Effect of intensive control of glucose on cardiovascular outcomes and death in patients with diabetes mellitus: a meta-analysis of randomised controlled trials. Lancet 2009;373:1765–72. https://doi.org/10.1016/S0140-6736(09)60697-8
19. Zoungas S, Arima H, Gerstein HC et al. Effects of intensive glucose control on microvascular outcomes in patients with type 2 diabetes: a meta-analysis of individual participant data from randomised controlled trials. Lancet Diabetes Endocrinol 2017;5:431–7. https://doi.org/10.1016/S2213-8587(17)30104-3
20. Zinman B, Wanner C, Lachin JM et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med 2015;373:2117–28. https://doi.org/10.1056/NEJMoa1504720
21. Emerging Risk Factors Collaboration, Di Angelantonio E, Kaptoge S et al. Association of cardiometabolic multimorbidity with mortality. JAMA 2015;314:52–60. https://doi.org/10.1001/jama.2015.7008
22. Brownrigg JRW, Hughes CO, Burleigh D et al. Microvascular disease and risk of cardiovascular events among individuals with type 2 diabetes: a population-level cohort study. Lancet Diabetes Endocrinol 2016;4:588–97. https://doi.org/10.1016/S2213-8587(16)30057-2
23. Gerstein HC, Werstuck GH. Dysglycaemia, vasculopenia, and the chronic consequences of diabetes. Lancet Diabetes Endocrinol 2013;1:71–8. https://doi.org/10.1016/S2213-8587(13)70025-1



