Anticoagulation with vitamin K antagonists is recommended for prevention of thromboembolism in patients with mechanical heart valves. However, this treatment carries a notable risk of bleeding. We performed a retrospective analysis of patients with mechanical heart valves who were anticoagulated with warfarin, extracting the number and type of mechanical heart valves and their time in therapeutic range (TTR) for various recommended international normalised ratio (INR) ranges. Following this, we carried out a prospective study, over two separate six-month periods, identifying those with mechanical heart valves who sustained a bleeding or thromboembolic event.
We identified 409 patients with mechanical heart valves with an overall TTR of 68%. Over 12 months, we recorded 32 possible bleeding incidents in 22 patients. There was one thromboembolic event during this period, specifically a cerebrovascular event.
In conclusion, while our data indicate an elevated risk of bleeding, additional larger studies are needed to better understand bleeding risks across different demographic groups.
Introduction
Mechanical valve replacements were first developed in the 1960s with the Starr-Edwards ball-and-cage valve.1 These valves required strict anticoagulation with warfarin, and were subsequently replaced by the less thrombogenic tilting disc valves, and then bileaflet valves. Despite advances in valve technology, even the newer-generation valves require anticoagulation to manage the thromboembolic risk. Currently, vitamin K antagonists (VKAs), such as warfarin, are the only licensed medicines for the reduction of thromboembolic risk, with direct-acting oral anticoagulants (DOACs) currently contraindicated.2,3
What is the current evidence?
The management of anticoagulation for mechanical heart valves (MHVs) is guided by two primary sets of recommendations: the American College of Cardiology/American Heart Association (ACC/AHA) guideline,4 and the European Society of Cardiology/European Association for Cardio-Thoracic Surgery (ESC/EACTS) guideline.5 Both ACC/AHA and ESC/EACTS advocate for the use of VKAs and recommend an international normalised ratio (INR) target tailored to individual risk factors and the type of MHV. According to ESC/EACTS guidelines, specific risk factors for thromboembolic events include having a mechanical mitral or tricuspid valve, a history of thromboembolism, atrial fibrillation, all severities of previous mitral stenosis and left ventricular systolic ejection fraction of under 35%.5 The ACC/AHA guideline encompasses these factors, but also includes a hypercoagulable state and older generation valves. Evidence suggests that there is a greater risk of thromboembolic events associated with mitral rather than aortic MHVs, both in the short6 and long term.7,8
On-X valves are a new generation of valves introduced in the 1990s, which were designed to address some of the limitations associated with earlier mechanical valves, such as the need for high levels of anticoagulation, durability and performance.9 Guidance on the management of On-X valves after three months varies in these guidelines. ACC/AHA states that for On-X valves with no additional risk factors, warfarin could be considered with an INR target of 1.5–2 in combination with aspirin. This recommendation is based on the PROACT (Prospective Randomized On-X Valve Anticoagulation Clinical Trial) study, which found that a lower INR with aspirin was associated with lower bleeding incidents, without affecting the incidence of strokes or thromboembolic events.10 Another study looked at the potential role for dual-antiplatelet therapy, rather than aspirin, with a lower INR, however, this was terminated due to an increased incidence of thromboembolic events in the dual-antiplatelet group.11 Despite the recommendations of the ACC/AHA guideline for consideration of this lower range, ESC/EACTS suggest benefits from the PROACT trial might be partly associated with home monitoring.10 ESC/EACTS, therefore, recommends caution and further studies before revising their recommendations.
Are there alternatives to VKAs?
One of the major disadvantages of VKAs is the potential negative outcomes associated with a labile INR and subsequent low time in the therapeutic window (TTR).12 Studies have sought to compare the non-inferiority of DOACs with warfarin in the context of MHV. Initial studies looking at the non-inferiority of apixaban13 and dabigatran3 in MHVs, were both terminated due to thromboembolic events. Studies looking at rivaroxaban are more promising, but are still very early in their design.14–16 Given this, the use of DOACs in MHVs has been contraindicated and is not recommended in current practice.2,4,5
What was the aim of our study?
The aim of our study was to assess the risk of bleeding and thromboembolic events in our cohort of patients. Furthermore, whether our cohort aligns with current guidelines regarding INR targets and concurrent aspirin use.
Method
We conducted a retrospective analysis of our anticoagulation database for Oxfordshire that contains all patients who are on warfarin. All patients with a MHV were identified and included in our study. Clinical records were reviewed for the type of mechanical valve, the recommended INR range, concomitant aspirin use and the TTR. The TTR was calculated using the Rosendaal method.17 This was approved as a quality improvement project (QIP) at our trust and, therefore, did not require ethical approval.
Data were collected over two separate six-month periods, commencing on 1 April 2019 and 1 January 2021. The data were collected in two different six-month periods to mitigate the risk of bias associated with the impact of Covid-19 on presentations to the emergency department. Patients who presented to an acute hospital setting with a bleed, or suspected bleed, with a MHV were identified. Factors, such as age, type of mechanical valve, bleed site, INR, TTR and the duration of their admission were recorded. This was also registered as a QIP with our trust.
All MHV patients are managed by the local anticoagulation service. The service is run by specialist nurses under the clinical lead of a haemostasis consultant. It manages warfarin dosing using the HiruMed RAID (Rapid Anticoagulant Interpretation & Dosing)18 software, on behalf of all the local GP practices, who remain responsible for the prescribing and supply of warfarin.
The service accepts referrals from primary and secondary care. It provides patient education for patients newly commenced on warfarin, support to the patient and clinicians through peri-operative/procedure management, and supports secondary care in safe and efficient discharges to primary care. It runs a point-of-care (POC) service for INR finger-prick testing, and for training and monitoring of patients who have their own POC device.
These data were analysed in R (version 4.3.2). Plots were produced with the ggplot package and data were analysed via linear regression and t-tests, where appropriate. No post-hoc tests were conducted.
Results
Table 1. Time in therapeutic range (TTR) for different types of valves
| Valve | TTR (n) | Self-test TTR (n) |
| All aortic | 68% (317) | 73% (26) |
| Aortic unknown type | 67% (64) | 76% (6) |
| Aortic On-X | 68% (32) | 73% (6) |
| Aortic bileaflet (excluding On-X) | 71% (181) | 69% (12) |
| Aortic caged ball | 46% (11) | 98% (1) |
| Aortic tilting disc | 67% (29) | 78 (1) |
| All mitral | 62% (66) | 61% (8) |
| Mitral unknown type | 65% (17) | 66% (2) |
| Mitral On-X | 59% (8) | 0 |
| Mitral bileaflet (excluding On-X) | 61% (36) | 60% (6) |
| Mitral tilting disc | 60% (5) | 0 |
| Other | ||
| Mitral and aortic valve | 70% (15) | 70% (1) |
| Tricuspid | 82% (3) | 100% (1) |
| Unknown valve type | 85% (8) | 90% (1) |
In total, 409 patients were identified with mechanical valves, of which 317 (77%) had an isolated aortic MHV, 66 (16%) had an isolated mitral MHV, three (1%) had an isolated tricuspid MHV, 15 (4%) had both an aortic and mitral MHV, and there were eight (2%) whose valves type were not known. The mean age was 66 years with a range of 19 to 94 years. All patients were in the Oxfordshire region.
Of the 317 isolated aortic valves; 213 were bileaflet (32 On-X), 29 were tilting disc, 11 cage and ball, and in 64 the valve type was unknown. Of the 66 mitral valves, 44 were bileaflet (eight On-X), five were tilting disc and 17 were unknown.
The range of INR varied depending on the valve type. There was not a statistical difference between INR ranges between any of the valves.
The overall TTR was 68% for those with a MHV. A subanalysis of those who self-tested their INR showed a TTR of 73% in the aortic MHV group. This demonstrated a significant difference (p=0.003) between the self-testing patients and patients who attended for a blood test, whether at the GP, phlebotomist, or pharmacy, etc. TTR was marginally better in the aortic valve group than the mitral valve group, who had a 62% TTR and a 61% self-TTR (table 1).
Of those with an unknown aortic valve type, 20 of the 64 (31%) patients were also on aspirin. In patients with aortic bileaflet On-X valves, 10 patients were taking aspirin out of a potential 32. For the remainder of valves, the concurrent use of aspirin was aortic bileaflet (excluding On-X) (15/181), aortic tilting disc (2/29), aortic caged ball (2/11) and all mitral valves (3/66). Of those with an aortic bileaflet On-X valve, five out of six of those with a target INR range of 1.5–2.0 were on aspirin.
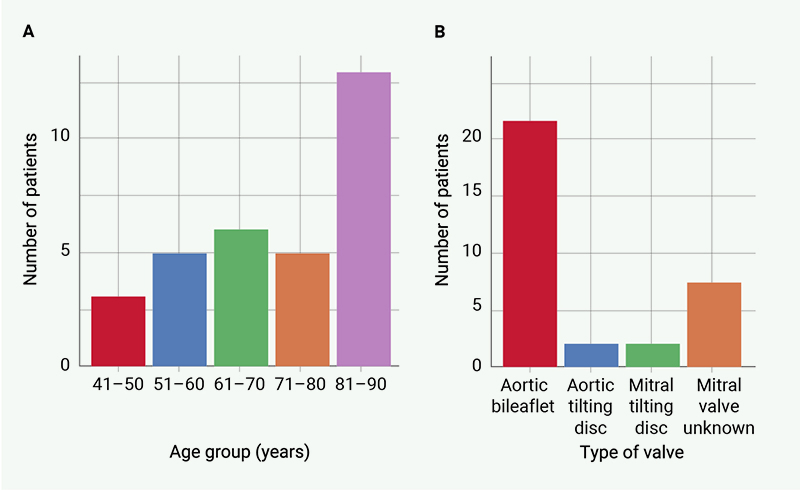
Over the combined 12-month period we observed 32 presentations for possible bleeds with MHVs involving 22 patients. The most common age group presenting to the emergency department was 81–90 years old, accounting for 41% of those attending the emergency department (figure 1A). The most common type of valve associated with potential bleeding was an aortic bileaflet (62%). Other associated valves were the aortic tilting disc, the mitral tilting disc and other unknown mitral valves (figure 1B). Patients presented with epistaxis 34% (11/32), a potential intracranial bleed 22% (7/32), haematuria 16% (5/32), gastrointestinal bleeding 9% (3/32), 13% (4/32) postprocedure or soft tissue, with two unknown sources.
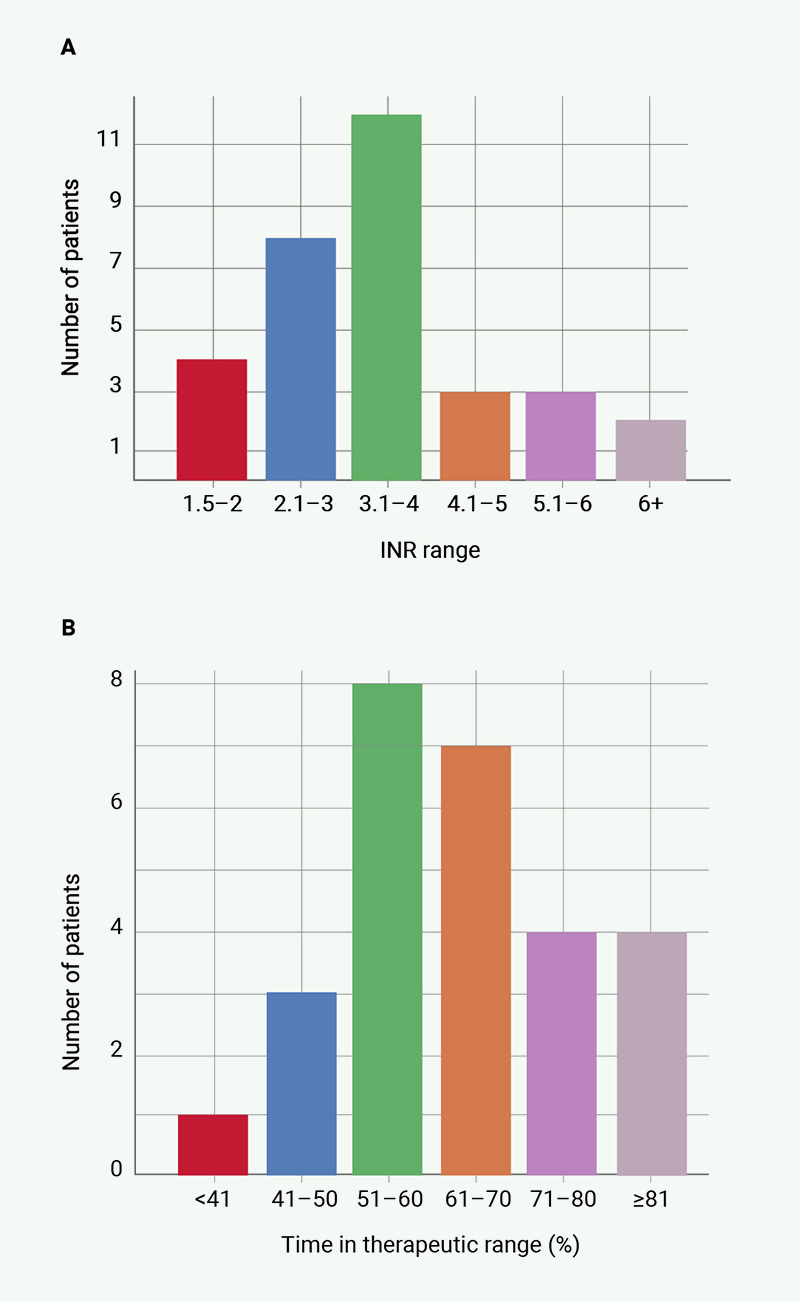
The presenting INRs of those who bled was most frequently in the range of 3–4, however, four patients bled with an INR of 1.5–2 and five patients bled with an INR >5 (figure 2A). The TTR, on average, was lower than our patient population of 68%, with only 25% having a TTR above 70%. The distribution can be seen in figure 2B. Of the 32 presentations to hospital, 23 (72%) of these attended hospital for less than 24 hours, nine stayed longer than 24 hours, and two patients stayed for over 10 days. We did not extract data differences between aortic and mitral valves.
In the same combined 12-month period, there was only one episode of thromboembolism. This was in an 82-year-old who presented with dysphasia and had a confirmed cerebrovascular accident. The TTR was 77%. At presentation, his INR was 2.2 and he had an aortic MHV placed in 1982.
Discussion
Evidence has demonstrated the risk of thromboembolic events with MHVs, however, our data demonstrate that bleeding events are significantly more common. This generated a review of our guidelines on anticoagulation bridging.
Our findings were consistent with current evidence that a lower TTR is associated with negative outcomes.12 In response, patients identified with a lower TTR are now routinely reviewed in pharmacist-led clinics, which has provisionally shown an improvement in TTR. Furthermore, our data confirm that self-testing is associated with a higher time in TTR, however, we note the potential difficulties in acquiring a machine for doing this, and the potential economic implications. As part of service improvement, we have also been exploring better access to POC testing for patients with MHVs in our region. Currently, the ESC recommends bridging therapy with an INR <1.5 with unfractionated or low-molecular-weight heparin cover for major surgery for all MHVs.5 However, guidelines vary, with the ACC not recommending bridging unless the patient has a mechanical aortic valve replacement (AVR) and thromboembolic risk factor, mitral valve or older style AVR.4 This raises the question of whether there should be bridging for AVR. Following our research, we no longer bridge those with bileaflet AVR, and we are monitoring the outcome of this. Our results highlight the critical need to maintain the correct INR range for the type of MHV.
Despite recommendations from both the ESC and ACC for INR targets for patients with MHVs, eight of our patients did not have a correctly documented valve type, which can lead to confusion with anticoagulation dosing. Following the review of our retrospective analysis, we have confirmed site and type of MHV, and have now decreased the number of unknown type MHVs from eight to zero. Our study also identified patients whose INR ranges did not correlate with their valve type and associated lower TTRs. Since completion of the analysis, we have reviewed this and improved the TTRs in this population. Furthermore, the previous type of valve and target INR was not readily visible to clinicians, and this is now available with dosing information on our electronic records.
Our data suggest that those who present with potential bleeds tend to be older, have a low TTR, and a higher INR range. Most frequently, the INR at presentation for those who bled was in the range of 3–4. The TTR, on average, was lower than our overall patient population of 68%, with only 25% having a TTR above 70%. There was a significant difference in the number of bleeding versus thromboembolic events in our population, with bleeding being much more frequent in our study. Considering that age is a significant risk factor for bleeding, we have implemented more frequent reviews in those over the age of 80 years.
At present, the use of DOACs with MHVs is contraindicated.2 A 2013 study looking at the use of dabigatran in MHVs was terminated early due to significantly higher incidence of thromboembolic events. Although a potential limitation of this trial was that dabigatran was started after seven days and within the first three months post-MHV replacement, which is known to be the time of highest risk of thromboembolic complications.19,20 A study analysing the efficacy of apixaban versus warfarin with On-X aortic valves was also terminated due to excessive thromboembolic events in the apixaban group.13 Most promisingly, there have been three small studies looking at rivaroxaban, which have demonstrated the same effectiveness as warfarin in both mitral and aortic valves.14–16 These data reiterate the importance of researching alternatives to VKAs for the management of the risk of thromboembolism, while mitigating the risk of bleeding.
Research has demonstrated the benefit of VKAs on the prevention of thromboembolism, however, there is less evidence regarding the incidence of bleeding in those with MHVs who are established on anticoagulation. One guideline concluded that the benefits in the reduction in thromboembolism outweighed the risk of bleeding, although through indirect evidence.21 A subsequent paper analysed the risk of bleeding and thromboembolic events in those commenced on VKAs with different INR ranges. This demonstrated a small reduction in thromboembolic events, but a larger decrease in bleeding events, 56 to 34, in the lower INR groups.22 Although not the analysis intended, this paper highlights the higher incidence of bleeding than thromboembolism.
One limitation is the size of our data collection. Despite collecting for a year, this produced only 33 episodes of bleeding and thromboembolism. Therefore, although conclusions can be drawn, we need to ensure that further studies are conducted, and the bias associated with a small sample is considered when applying this clinically. Furthermore, the retrospective nature of this data collection could increase the risk of selection bias and, therefore, this needs to be considered when discussing the results of this study.
Finally, we acknowledge a limitation in our data collection regarding potential intracranial bleeds, and that we did not extract data regarding if a significant bleed was present. Given the changes that we have now implemented into our monitoring system and prevention of bleeding, we intend to be more specific in our data collection and extract this information in the future.
Our paper is one of the few studies looking at longer-term anticoagulation therapy with specific analyses regarding the type of bleeding, TTR and INR ranges. It, therefore, raises the question of whether further consideration needs to be given regarding the incidence of bleeding. One could query whether the benefit of prevention of thromboembolism outweighs the risk of bleeding in certain patients with less thrombogenic MHVs. Further research needs to be conducted in order to ascertain whether there might be certain patients where a lower INR with an antiplatelet or alternative anticoagulation strategy, such as rivaroxaban, might be more appropriate.
Conclusion
There was one thromboembolic event in comparison to 32 potential bleeding events over a combined 12-month period for those with MHVs. This raises an important question regarding whether the risk of bleeding is outweighed in all patients with a MHV. Our evidence highlights the importance for larger studies in order to mitigate this risk.
Key messages
- Our study identified 32 potential bleeding incidents compared with one thromboembolic event within our patient cohort. Patients who experienced bleeding were older and had a lower time in therapeutic range (TTR) and a higher international normalised ratio (INR). There was an associated higher TTR in those who used point-of-care testing
- Given the higher risk of bleeding, our research could influence anticoagulation policies and bridging strategies for procedures
- Furthermore, our data have shown the importance of monitoring those on warfarin, and the associated implications of a low TTR, which affects clinical practice
Conflicts of interest
None declared.
Funding
None.
Study approval
The study was a clinical audit, therefore, approval from the local ethics committee was not required, but was registered as a quality improvement project. All patients with mechanical valves have given consent to their anonymised data being collected and audited as part of the NICOR dataset.
Acknowledgements
We would like to thank the haemostasis laboratory and HIRUMED.
References
1. Starr A, Edwards ML. Mitral replacement: clinical experience with a ball-valve prosthesis. Ann Surg 1961;154:726–40. https://doi.org/10.1097/00000658-196110000-00017
2. US Food and Drug Administration. FDA drug safety communication: Pradaxa (dabigatran etexilate mesylate) should not be used in patients with mechanical prosthetic heart valves. FDA, 19 December 2012 [accessed 18 December 2023]. Available at: https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-pradaxa-dabigatran-etexilate-mesylate-should-not-be-used-patients
3. Eikelboom JW, Connolly SJ, Brueckmann M et al. Dabigatran versus warfarin in patients with mechanical heart valves. N Engl J Med 2013;369:1206–14. https://doi.org/10.1056/NEJMoa1300615
4. Otto CM, Nishimura RA, Bonow RO et al. 2020 ACC/AHA guideline for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association joint committee on clinical practice guidelines. Circulation 2021;143:e72–e227. https://doi.org/10.1161/CIR.0000000000000923
5. Vahanian A, Beyersdorf F, Praz F et al. 2021 ESC/EACTS guidelines for the management of valvular heart disease: developed by the task force for the management of valvular heart disease of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J 2022;43:561–632. https://doi.org/10.1093/eurheartj/ehac051
6. Allou N, Piednoir P, Berroëta C et al. Incidence and risk factors of early thromboembolic events after mechanical heart valve replacement in patients treated with intravenous unfractionated heparin. Heart 2009;95:1694–700. https://doi.org/10.1136/hrt.2009.167270
7. Cannegieter SC, Rosendaal FR, Briët E. Thromboembolic and bleeding complications in patients with mechanical heart valve prostheses. Circulation 1994;89:635–41. https://doi.org/10.1161/01.CIR.89.2.635
8. Pruefer D, Dahm M, Dohmen G, Horstkotte D, Bergemann R, Oelert H. Intensity of oral anticoagulation after implantation of St. Jude Medical mitral or multiple valve replacement: lessons learned from GELIA (GELIA 5). Eur Heart J Suppl 2001;3(suppl Q):Q39–Q43. https://doi.org/10.1016/S1520-765X(01)90041-0
9. Gerdisch MW, Hagberg RC, Perchinsky MJ et al. Low-dose warfarin with a novel mechanical aortic valve: interim registry results at 5-year follow-up. J Thorac Cardiovasc Surg 2024;168:1645.e6–1655.e6. https://doi.org/10.1016/j.jtcvs.2024.04.017
10. Puskas J, Gerdisch M, Nichols D et al. Reduced anticoagulation after mechanical aortic valve replacement: interim results from the Prospective Randomized On-X Valve Anticoagulation Clinical Trial randomized Food and Drug Administration investigational device exemption trial. J Thorac Cardiovasc Surg 2014;147:1202.e2–1211.e2. https://doi.org/10.1016/j.jtcvs.2014.01.004
11. Puskas JD, Gerdisch M, Nichols D et al. Anticoagulation and antiplatelet strategies after On-X mechanical aortic valve replacement. J Am Coll Cardiol 2018;71:2717–26. https://doi.org/10.1016/j.jacc.2018.03.535
12. Havers-Borgersen E, Butt JH, Vinding NE et al. Time in therapeutic range and risk of thromboembolism and bleeding in patients with a mechanical heart valve prosthesis. J Thorac Cardiovasc Surg 2020;159:74.e4–83.e4. https://doi.org/10.1016/j.jtcvs.2019.02.061
13. Wang TY, Svensson LG, Wen J et al. Apixaban or warfarin in patients with an On-X mechanical aortic valve. NEJM Evidence 2023;2:EVIDoa2300067. https://doi.org/10.1056/EVIDoa2300067
14. Duraes AR, Bitar Y de SL, Schonhofen IS et al. Rivaroxaban versus warfarin in patients with mechanical heart valves: open-label, proof-of-concept trial – the RIWA study. Am J Cardiovasc Drugs 2021;21:363–71. https://doi.org/10.1007/s40256-020-00449-3
15. Durães AR, Bitar Y de SL, Lima MLG et al. Usefulness and safety of rivaroxaban in patients following isolated mitral valve replacement with a mechanical prosthesis. Am J Cardiol 2018;122:1047–50. https://doi.org/10.1016/j.amjcard.2018.06.015
16. Roost E, Weber A, Alberio L et al. Rivaroxaban in patients with mechanical heart valves: a pilot study. Thromb Res 2020;186:1–6. https://doi.org/10.1016/j.thromres.2019.12.005
17. INRPro.com. Using the Rosendaal method for calculating Therapeutic Time in Range (TTR). Available at: https://www.inrpro.com/rosendaal.asp
18. Hirumed Ltd. Hirumed ‘RAID’. Available at: https://www.hirumed.co.uk/raid.html
19. Sousa-Uva M, Head SJ, Milojevic M et al. 2017 EACTS guidelines on perioperative medication in adult cardiac surgery. Eur J Cardiothorac Surg 2018;53:5–33. https://doi.org/10.1093/ejcts/ezx314
20. Laffort P, Roudaut R, Roques X et al. Early and long-term (one-year) effects of the association of aspirin and oral anticoagulant on thrombi and morbidity after replacement of the mitral valve with the St. Jude medical prosthesis: a clinical and transesophageal echocardiographic study. J Am Coll Cardiol 2000;35:739–46. https://doi.org/10.1016/S0735-1097(99)00598-7
21. Whitlock RP, Sun JC, Fremes SE, Rubens FD, Teoh KH. Antithrombotic and thrombolytic therapy for valvular disease. Chest 2012;141(2 suppl):e576S–e600S. https://doi.org/10.1378/chest.11-2305
22. Acar J, Iung B, Boissel JP et al. AREVA: multicenter randomized comparison of low-dose versus standard-dose anticoagulation in patients with mechanical prosthetic heart valves. Circulation 1996;94:2107–12. https://doi.org/10.1161/01.CIR.94.9.2107

