We conducted a pilot study to evaluate the safety of the shuttle walking test (SWT) and exercise training for cardiac patients in community-based cardiac rehabilitation settings. Overall, 33 cardiac patients were tested (19 males and 14 females, 67 ± 8 years). Eleven cardiac patients (testing group) and 22 cardiac patients (training group) underwent ambulatory electrocardiogram (ECG) monitoring during the SWT and exercise training during a long-term cardiac rehabilitation programme. Frequency of ECG events was reported for the two groups. Chi-square test was performed to determine associations between the incidence of cardiovascular events and poor functional capacity (SWT <450 m).
The findings showed only minor events provoked during the SWT or exercise training, and no event-related hospitalisation, syncope episodes or fatality. The most important cardiac event was silent myocardial ischaemia (testing group: 27.3%; training group: 18%). Poor functional capacity was not associated with the risk of a cardiac event during exercise (testing group: χ2=0, p=0.99, phi=0.24; training group: χ2=2.1, p=0.15, phi=–0.42).
In conclusion, supervised exercise testing and training are accompanied only by minor cardiovascular events and they can be carried out safely in community-based cardiac rehabilitation settings.
Introduction
 Exercise is well recognised as a tool for assessment, prevention and management of cardiovascular disease.1 Cardiac patients are encouraged to attend cardiac rehabilitation programmes including elements of supervised exercise. Such programmes can reduce mortality and morbidity rates by up to 27%.2,3
Exercise is well recognised as a tool for assessment, prevention and management of cardiovascular disease.1 Cardiac patients are encouraged to attend cardiac rehabilitation programmes including elements of supervised exercise. Such programmes can reduce mortality and morbidity rates by up to 27%.2,3
Despite the benefits derived from participation in exercise-based cardiac rehabilitation, exercise itself may act as a trigger for myocardial ischaemia or cardiac arrest in patients with established coronary heart disease.4 During rehabilitation, cardiovascular event rates range from 12.3 to 37.4 per million patient hours of exercise.4-8 Fatal cardiac events are more rarely reported, but range from 8.6 per million patient hours of exercise in 19784 to zero in the present day.5-8 Event rates during exercise are also lower in more recent studies of supervised cardiac rehabilitation exercise programmes,5-8 events during exercise sessions do still occur, however.
The American Heart Association9,10 emphasise the importance of pre-training cardiovascular risk assessment, including medical history, physical examination and ambulatory electrocardiogram (ECG) monitoring during exercise testing. These actions are recommended to minimise exercise-induced cardiovascular events by identifying patients at greatest risk.
Cardiovascular events during exercise testing using traditional laboratory exercise protocols (treadmill, cycle ergometer) are also rare,4,8,11 and appear difficult to predict.5,8 Laboratory-based exercise protocols are often replaced in daily clinical practice by functional capacity tests, such as the incremental shuttle walking test (SWT). This test is reliable,12 relatively simple to perform and inexpensive.13 Only one study has evaluated the safety of this test in a cardiac population.14 The authors found a very low number of cardiovascular events during exertion, but questioned whether this was the result of low individual effort during exertion or successful treatment of coronary arteries during surgical intervention. The small and selective subsample (19 post coronary artery bypass graft surgery patients) studied, increases the necessity for new studies in this field.
The Scottish Intercollegiate Guidelines Network (SIGN) recommends the use of exercise testing15 and the British Association for Cardiac Rehabilitation (BACR) recommends the use of exercise training,16 to detect exercise-induced cardiovascular events. Thus, the aim of this study was to verify to what extent a recommended exercise testing protocol (SWT) and an exercise training session are safe for a mixed cohort of cardiac rehabilitation patients.
Materials and methods
Study participants
Overall, 33 cardiac patients were tested (19 males and 14 females, 67 ± 8 years). Eleven patients were assessed while undertaking a sub-maximal SWT (testing group) and 22 patients were assessed during cardiac rehabilitation exercise training (training group).
All patients were clinically stable and had been enrolled in a community-based, phase IV cardiac rehabilitation programme for at least 10 weeks. Patients with severe locomotor limitations were excluded from this study.
All procedures were approved by the University ethical committee and conformed to the declaration of Helsinki guidelines for research with human subjects.17 Patients were verbally recruited by instructors and informed written consent was obtained from all patients before enrolment.
Protocol and measurements
Anthropometric and cardiovascular assessment
Patients were assessed once, each received a primary health assessment (pre-exercise health questionnaire, medical, pharmacological history, resting heart rate, resting blood pressure) and anthropometric assessment (stature, body mass, waist circumference) prior to testing. Body mass index (BMI) was calculated (kg/m2). Table 1 summarises the participants’ clinical characteristics and baseline measurements.

All patients were monitored with an ambulatory ECG event monitor (C.NET5000, version 1.2, Cardionetics Ltd., United Kingdom), either during the SWT (testing group) or during the cardiac rehabilitation class (training group). A single-lead ECG was recorded using an electrode configuration analogous to lead standard V5 in a traditional 12-lead clinical ECG monitoring. The recording device was placed into a pouch fitted to the patient’s waist using a particular type of belt.14
Major cardiovascular events were defined as one of the following: myocardial infarction requiring hospitalisation, ventricular fibrillation, ventricular tachycardia requiring treatment, atrial arrhythmias requiring treatment, asystole, stroke and death. Minor cardiovascular events were defined as: isolated ventricular arrhythmias or atrial arrhythmias not requiring intervention, chest pain and bradycardia not requiring intervention.11
Exercise testing
Patients performed a SWT13 by walking back and forth between two cones set 0.5 m from either end of a 10 m course. Initial walking speed, indicated by an audible signal, was 0.5 m/s and increased by 0.13 m/s each minute. During the test, heart rate was also recorded with a heart rate monitor (Polar Electro Sports Tester S810I, Heart Rate Monitor, Kempele, Finland) and ratings of perceived exertion (RPE) were measured using the Borg (6–20) scale.18
The SWT was terminated when the patient (a) felt too breathless or fatigued to continue at the required speed, (b) failed to complete the shuttle within the allowed time, (c) reached 85% of the predicted maximal heart rate: 210 – (0.65×age), (d) reached RPE ≥15 (Borg 1998), or (e) completed all the levels.
Exercise training programme
The programme offers the opportunity of life-long supervised exercise for cardiac patients. The programme consisted of two supervised sessions per week, made up of 60 minutes of circuit-based exercise classes. Supervisory staff included a physiotherapist specialised in cardiac rehabilitation, also trained in immediate life support methods. Exercise sessions comprised: a 15-minute warm-up, a 35-minute main conditioning component and a 10-minute cool-down period. The conditioning component of the session included cardiovascular and strength exercises at an intensity of 60–80% of the age-predicted maximal heart rate, or at 12 to 15 on the RPE scale. During this component patients had access to a variety of exercise equipment, such as arm ergometers, rowing ergometers, progressive resistance equipment, balance equipment, steppers and free weights.
Data analysis
A cardiologist verified the presence or absence of each cardiovascular event. Frequency of the cardiovascular events was reported for both groups. Patients were divided into event-free versus cardiac-event subgroups. Differences in functional capacity between the two subgroups were examined using a two-sample t-test. A chi-square test was performed to determine associations between incidences of cardiovascular events with poor functional capacity (SWT <450 m).19
All statistical analyses were performed using SPSS version 16.0 (SPSS nc., Chicago, IL, USA). The statistical significance was set at p<0.05.
Results
Event rates
No major event related to hospitalisation was detected in both testing and training groups. The most clinically important event was silent myocardial ischaemia, which occurred in 27.3% of the testing group patients, and in 18% of the training group patients. There was no significant difference in functional capacity between the event-free subgroup and the subgroup with cardiovascular events in both testing and training group (p>0.05).
Frequency of cardiovascular events during exercise testing
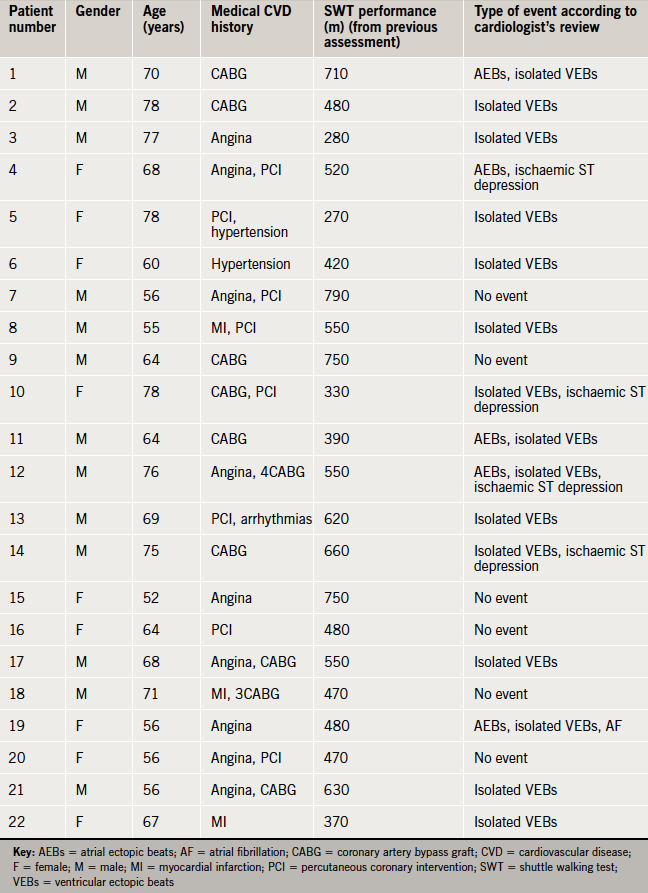
Eight cardiovascular events were detected automatically by the ECG monitoring system during exercise testing. Seven (82%) were verified by the cardiologist. According to the cardiologist’s review of the ECG recordings, of the eleven patients, 5 (45.5%) had atrial ectopic beats, four had isolated ventricular ectopic beats (36.4%), three (27.3%) had ischaemic ST segment depression, two (18.2%) had an atrial fibrillation event, one (9.1%) had a bradycardia event (under 50 beats per minute), while four (36.4%) of the patients were free from cardiovascular events (table 2).

Frequency of cardiovascular events during exercise training
Among 18 cardiovascular events automatically detected during exercise training, 16 (73%) were verified by the cardiologist. According to the cardiologist’s review of the ECG recordings; 15 patients (68%) had isolated ventricular ectopic beats, four (18%) had atrial ectopic beats, four (18%) had ischaemic ST segment depression, one (4.6%) had atrial fibrillation. Six (27.3%) of the patients were free from cardiovascular events (table 3).
In all, 76.5% of thecardiovascular eventswere detected around the time of the main conditioning component of the exercise training, 11.8% during the warm up and 23.5% during the cool down.
Association between frequency of cardiovascular events and patient functional capacity
A chi-square test showed no significant association between poor functional capacity (SWT <450 m) and risk for cardiovascular events in either testing group or training group (testing group: χ2=0, p=0.99, phi=0.24; training group: χ2=2.1, p=0.15, phi=–0.42). Among patients with poor functional capacity, cardiovascular events were found in half of those (n=1) who were assessed during exercise testing with the SWT, and in all of those (n=7) who were assessed during exercise training.
Discussion
The present study provides information regarding the safety of exercise training during a community-based, phase IV cardiac rehabilitation programme and a standard functional exercise test.
Risk of cardiovascular events during exercise testing and training
According to cardiologist-verified ECG data only minor, non-fatal cardiovascular events were observed during both the SWT (with an event rate of 0.64 per patient per test) and in exercise training (with an event rate of 0.73 per patient per session).No major cardiovascular events were detected during exercise testing or training. These findings agree with those of Hollenberg et al.20 who found no complex arrhythmias or symptomatic ischaemia during exercise in participants without suspected cardiac disease.
Contrary to previous studies,4,5,8 we showed that exercise training is associated with more events than exercise testing. This may be explained by use of the SWT, whereas previous studies have used treadmill tests, which may provoke greater stresses on the cardiovascular system than the incremental SWT.21-23The most serious (major) event, silent ischaemia, was observed more often during the SWT than during exercise training. This may indicate that the SWT induces fewer, yet, potentially more serious cardiovascular events than exercise training. This is likely due to differences in work rate and duration of exercise. For example, during the SWT the patients eventually achieve exercise at a higher intensity than during circuit-based exercise training, resulting in greater physiological stresses and, thus, major events. The nature of the observed cardiovascular events is discussed below.
Myocardial ischaemia
The most clinically important event was silent (asymptomatic-painless) myocardial ischaemia indicated by ST segment depression. This event is likely associated with untreated coronary artery stenosis8 or, more likely, exercise-induced transient oxygen deprivation.6 Silent exercise-induced ST segment depression should not be regarded as a severe cardiac event. Its survival rate is similar to silent non-ST ischaemic event, and significantly greater than symptomatic ST changes, in cardiac patients.24
A significant association was found between silent myocardial ischaemia and sudden cardiac death in men without a history of coronary heart disease with a high number of risk factors.25 However, this result cannot be used in the present study due to different criteria for subject selection. This highly significant relationship between silent ischaemia and cardiac death might be associated with the large number of false-positive diagnostic exercise tests,25 as the presence of false-positive or negative exercise tests are higher in asymptomatic individuals (Baye’s rule).26,27 Thus, the presence of silent myocardial ischaemia does not necessarily signify a severe cardiovascular condition.
Cardiac arrhythmias
There was a relatively high prevalence of ventricular and atrial arrhythmias, during exercise but these were not related to any major cardiac event. Particularly, non-complex ventricular ectopic activity, atrial ectopics and atrial fibrillation were detected in 70% of patients. Atrial ectopic beats and fibrillation commonly occur in 15–40% of patients after coronary artery bypass graft surgery,28-30 10–11% of patients after a percutaneous coronary intervention,31,32 5–10% after a myocardial infarction,33 37–50% after a valve replacement25,34,35 and 11–24% after a cardiac transplantation.36,37 The above data were collected at rest, our study showed similar frequencies, as atrial arrhythmias were detected in 45% of patients during the SWT and 23% of patients during exercise training. The high percentage of atrial arrhythmias may be explained by higher participation rates of post-vascularisation patients and the fact that they were stress-induced arrhythmias.38
Ventricular ectopic beats were observed in 36.4% of patients during the SWT and in 68.2% during exercise training. The prognostic value of exercise-induced ventricular ectopic beats remains unclear.39,40 There is some evidence that they can be independent predictors of cardiac mortality in patients without a pre-existing diagnosis of coronary heart disease.41 These are common cardiovascular events during exercise for cardiac patients.40 Exercise can increase catecholamines by more than 10-fold, decrease serum pH and increase serum potassium by twofold.41 These changes can all predispose patients to arrhythmias, which are common and usually well tolerated during exercise.
Exercise-induced arrhythmias can also occur during the recovery period after exercise, when catecholamine levels usually continue to increase.42,43 No major cardiovascular events were reported post-exercise in our study. A small number of minor cardiovascular events were recorded during the cool-down phase. Most events (76.5%) were detected during the main component of the session; however, none of them were identified as a major event. The relative safety of the exercise training experienced by the present study group is possibly the result of a well-supervised exercise programme, which was risk stratified and gave special consideration to warm-up and cool-down elements.44
If any of these physiological changes occur during rest, there is an increased risk of cardiac arrest, particularly for patients with diagnosed coronary heart disease.42,45 The current data collection protocol did not include a resting ECG for comparison with the exercise data. No episodes of transient ventricular fibrillation were observed, however, supporting the case that the isolated ventricular ectopic beats were physiologically induced by exercise and not a contraindication for exercise.39
The presence of ventricular ectopic beats can also relate to elevatedlevelsof exercise stress. During treadmill exercise testing
the reproducibility of exercise-induced ventricular ectopic beats is low and a second exercise stress test was recommended.46This finding was not explained by the authors as a learning effect, but as a decrease in myocardial oxygen consumption and a product of the pressure-rate, which has not been substantiated. In the present study, only one trial of the SWT was used, since a previous study showed no learning effect.12 Moreover, the target of this study was to investigate events during exercise in a realistic cardiac rehabilitation setting.
Is it possible to predict the risk of cardiovascular events considering the functional capacity level?
Low exercise capacity (less than 5 metabolic equivalents [METs] or 450 m in SWT) is used in risk stratification for entry into cardiac rehabilitation programmes, exercise prescription and prognosis of mortality.19,47 Patients who walked <450 m in the SWT had no greater risk of cardiovascular events during exercise than patients who walked >450 m.
The lack of significant association between event rate and functional capacity may be due to the small sample and the lack of major events observed. The cut-off value of 450 m may also be inappropriate for this population. It was originally used to categorise cardiac patients as high and low performers, but was developed for use in heart failure patients. This group differ from the present cohort as they did not regularly exercise,19,47 due to severe cardiorespiratory problems, exercise intolerance and fatigue.48
Study limitations
The lack of resting ECG data means we cannot confirm whether the isolated ventricular ectopic beats recorded here are exercise-induced or related to other cardiovascular pathologies. The presence of isolated ventricular ectopic beats is not a contraindication to exercise, whether there was evidence that these arrhythmias are exercise-induced or not, the recommended management of a patient with such events would be similar.
The present sample size was relatively small, principally because ECG monitoring is not frequently used in the routine evaluation of low-risk, asymptomatic, cardiac rehabilitation patients. We had, therefore, to introduce this to our clinic and the sample actually represents all non-paced patients who were attending at the time of the study. This sample represents well a typical participation in a long-term cardiac rehabilitation programme.
Conclusions
During exercise testing or training, complex arrhythmias or symptomatic ST segment depression is not induced in cardiac patients enrolled regularly for more than two months in supervised cardiac rehabilitation maintenance programmes. Moreover, the overall minor cardiovascular event incidence of 0.7 per patient exercise session seems to be a safe rate in long-term cardiac rehabilitation settings. The minor cardiovascular events, such as arrhythmias and silent ST depression, are not to be ignored, but are a reason to suggest additional cardiac assessment and risk modifications.
This study provides important safety information for cardiac rehabilitation. It is suggested, first, that the routine evaluation of low-risk cardiac rehabilitation patients with a functional capacity higher than 5 METs (or >450 m performance in SWT), does not require ECG monitoring during exercise and they can safely exercise in community-based cardiac rehabilitation settings. Second, exercise prescription is safe for long-term cardiac rehabilitation patients, if the contraindications to testing and training are followed.
Further investigations should determine whether the presence of isolated ventricular arrhythmias is related to exercise; they should then examine the prognostic value of these minor events over a longer-term follow-up period.
Funding
No funding received for this work.
Conflict of interest
None declared.
Key messages
- Poor functional capacity is not shown to be associated with the risk of a cardiac event during exercise
- Supervised exercise testing and training are accompanied only by minor cardiovascular events and they can be carried out safely in community-based cardiac rehabilitation settings
- Although, exercise can sometimes trigger symptoms in cardiac patients, exercise prescription is safe for long-term supervised cardiac rehabilitation patients, if the contraindications to testing and training are followed
- When clinicians evaluate low-risk cardiac rehabilitation patients, there is no need for ECG monitoring during exercise in community-based cardiac rehabilitation settings
- Patients can safely exercise in a community-based cardiac rehabilitation setting
References
- Saha M, Redwood SR, Marber MS. Exercise training with ischaemia: is warming up the key? Eur Heart J 2007;28:1543–4. http://dx.doi.org/10.1093/eurheartj/ehm187
- O’Connor GT, Buring JE, Yusuf S et al. An overview of randomized trials of rehabilitation with exercise after myocardial infarction. Circulation 1989;80:234–44. http://dx.doi.org/10.1161/01.CIR.80.2.234
- Jolliffe JA, Rees K, Taylor RS et al. Exercise-based rehabilitation for coronary heart disease. Cochrane Database Syst Rev 2000;(4):CD001800. http://dx.doi.org/10.1002/14651858.CD001800
- Haskell WL. Cardiovascular complications during exercise training of cardiac patients. Circulation 1978;57:920–4. http://dx.doi.org/10.1161/01.CIR.57.5.920
- Van Camp SP, Peterson RA. Cardiovascular complications of outpatient cardiac rehabilitation programs. JAMA 1986;256:1160–3. http://dx.doi.org/10.1001/jama.1986.03380090100025
- Franklin BA, Bonzheim K, Gordon S, Timmis GC. Safety of medically supervised outpatient cardiac rehabilitation exercise therapy: a 16-year follow-up. Chest 1998;114:902–06. http://dx.doi.org/10.1378/chest.114.3.902
- Scheinowitz M, Harpaz D. Safety of cardiac rehabilitation in a medically supervised, community-based program. Cardiology 2005;103:113–17. http://dx.doi.org/10.1159/000083433
- Pavy B, Iliou MC, Meurin P et al. Safety of exercise training for cardiac patients: results of the French registry of complications during cardiac rehabilitation. Arch Intern Med 2006;166:2329–34. http://dx.doi.org/10.1001/archinte.166.21.2329
- American College of Sports Medicine. ACSM’s guidelines for exercise testing and prescription. 6th edition. Philadelphia: Lippincott Williams & Wilkins, 2000.
- Fletcher GF, Balady GJ, Amsterdam EA et al. Exercise standards for testing and training: a statement for healthcare professionals from the American Heart Association. Circulation 2001;104:1694–740. http://dx.doi.org/10.1161/hc3901.095960
- Gibbons L, Blair SN, Kohl HW, Cooper K. The safety of maximal exercise testing. Circulation 1989;80:846–52. http://dx.doi.org/10.1161/01.CIR.80.4.846
- Pepera G, McAllister J, Sandercock G. Long-term reliability of the incremental shuttle walking test in clinically stable cardiovascular disease patients. Physiotherapy 2010;96:222–7. http://dx.doi.org/10.1016/j.physio.2009.11.010
- Singh SJ, Morgan MD, Scott S et al. Development of a shuttle walking test of disability in patients with chronic airways obstruction. Thorax 1992;47:1019–24. http://dx.doi.org/10.1136/thx.47.12.1019
- Tobin D, Thow MK. The 10 m shuttle walk test with Holter monitoring: an objective outcome measure for cardiac rehabilitation. Coronary Health Care 1999;3:3–17. http://dx.doi.org/10.1016/S1362-3265(99)80028-5
- Scottish Intercollegiate Guidelines Network (SIGN). Cardiac rehabilitation. A national clinical guideline. Edinburgh: SIGN, 2002. Available from: http://www.sign.ac.uk/pdf/sign57.pdf [accessed 10/08/2011].
- British Association for Cardiac Rehabilitation. Exercise instructor. Training module. 4th edition. Leeds: Human Kinetics Europe, 2006.
- World Medical Association Inc. Declaration of Helsinki. Ethical principles for medical research involving human subjects. J Indian Med Assoc 2009;107:403–05.
- Borg G. Borg’s perceived exertion and pain scales. Champaign: Human Kinetics, 1998.
- Lewis ME, Newall C, Townend JN et al. Incremental shuttle walk test in the assessment of patients for heart transplantation. Heart 2001;86:183–7. http://dx.doi.org/10.1136/heart.86.2.183
- Hollenberg M, Ngo LH, Turner D, Tager IB. Treadmill exercise testing in an epidemiologic study of elderly subjects. J Gerontol A Biol Sci Med Sci 1998;53:B259–B267. http://dx.doi.org/10.1093/gerona/53A.4.B259
- Singh SJ, Morgan MD, Hardman AE et al. Comparison of oxygen uptake during a conventional treadmill test and the shuttle walking test in chronic airflow limitation. Eur Respir J 1994;7:2016–20.
- Zwierska I, Nawaz S, Walker RD et al. Treadmill versus shuttle walk tests of walking ability in intermittent claudication. Med Sci Sports Exerc 2004;36:1835–40. http://dx.doi.org/10.1249/01.MSS.0000145471.73711.66
- Fowler SJ, Singh SJ, Revill S. Reproducibility and validity of the incremental shuttle walking test in patients following coronary artery bypass surgery. Physiotherapy 2005;91:22–7. http://dx.doi.org/10.1016/j.physio.2004.08.009
- Mark DB, Hlatky MA, Califf RM et al. Painless exercise ST deviation on the treadmill: long-term prognosis. J Am Coll Cardiol 1989;14:885–92. http://dx.doi.org/10.1016/0735-1097(89)90459-2
- Laukkanen JA, Makikallio TH, Rauramaa R, Kurl S. Asymptomatic ST-segment depression during exercise testing and the risk of sudden cardiac death in middle-aged men: a population-based follow-up study. Eur Heart J 2009;30:558–65. http://dx.doi.org/10.1093/eurheartj/ehn584
- Miranda CP, Lehmann KG, Lachterman B et al. Comparison of silent and symptomatic ischemia during exercise testing in men. Ann Intern Med 1991;114:649–56.
- Lakka TA, Venalainen JM, Rauramaa R et al. Relation of leisure-time physical activity and cardiorespiratory fitness to the risk of acute myocardial infarction. N Engl J Med 1994;330:1549–54. http://dx.doi.org/10.1056/NEJM199406023302201
- Mathew JP, Fontes ML, Tudor IC et al. A multicenter risk index for atrial fibrillation after cardiac surgery. JAMA 2004;291:1720–9. http://dx.doi.org/10.1001/jama.291.14.1720
- Villareal RP, Hariharan R, Liu BC et al. Postoperative atrial fibrillation and mortality after coronary artery bypass surgery. J Am Coll Cardiol 2004;43:742–8. http://dx.doi.org/10.1016/j.jacc.2003.11.023
- Dogan SM, Buyukates M, Kandemir O et al. Predictors of atrial fibrillation after coronary artery bypass surgery. Coron Artery Dis 2007;18:327–31. http://dx.doi.org/10.1097/MCA.0b013e3281689a2c
- Celik T, Iyisoy A, Kursaklioglu H et al. Effects of primary percutaneous coronary intervention on P wave dispersion. Ann Noninvasive Electrocardiol 2005;10:342–7. http://dx.doi.org/10.1111/j.1542-474X.2005.00647.x
- Gorenek B, Parspur A, Timuralp B et al. Atrial fibrillation after percutaneous coronary intervention: predictive importance of clinical, angiographic features and P-wave dispersion. Cardiology 2007;107:203–08. http://dx.doi.org/10.1159/000095418
- Bhatia GS, Lip GY. Atrial fibrillation post-myocardial infarction: frequency, consequences, and management. Curr Heart Fail Rep 2004;1:149–55. http://dx.doi.org/10.1007/s11897-004-0002-y
- Asher CR, Miller DP, Grimm RA et al. Analysis of risk factors for development of atrial fibrillation early after cardiac valvular surgery. Am J Cardiol 1998;82:892–5. http://dx.doi.org/10.1016/S0002-9149(98)00498-6
- Banach M, Goch A, Misztal M et al. Predictors of paroxysmal atrial fibrillation in patients undergoing aortic valve replacement. J Thorac Cardiovasc Surg 2007;134:1569–76. http://dx.doi.org/10.1016/j.jtcvs.2007.08.032
- Creswell LL, Schuessler RB, Rosenbloom M, Cox JL. Hazards of postoperative atrial arrhythmias. Ann Thorac Surg 1993;56:539–49. http://dx.doi.org/10.1016/0003-4975(93)90894-N
- Pavri BB, O’Nunain SS, Newell JB et al. Prevalence and prognostic significance of atrial arrhythmias after orthotopic cardiac transplantation. J Am Coll Cardiol 1995;25:1673–80. http://dx.doi.org/10.1016/0735-1097(95)00047-8
- Bunch TJ, Chandrasekaran K, Gersh BJ et al. The prognostic significance of exercise-induced atrial arrhythmias. J Am Coll Cardiol 2004;43:1236–40. http://dx.doi.org/10.1016/j.jacc.2003.10.054
- Gibbons RJ, Balady GJ, Bricker JT et al. ACC/AHA 2002 guideline update for exercise testing: summary article: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Update the 1997 Exercise Testing Guidelines). Circulation 2002;106:1883–92. http://dx.doi.org/10.1161/01.CIR.0000034670.06526.15
- Dewey FE, Kapoor JR, Williams RS et al. Ventricular arrhythmias during clinical treadmill testing and prognosis. Arch Intern Med 2008;168:225–34. http://dx.doi.org/10.1001/archinte.168.2.225
- Beckerman J, Wu T, Jones S, Froelicher VF. Exercise test-induced arrhythmias. Prog Cardiovasc Dis 2005;47:285–305. http://dx.doi.org/10.1016/j.pcad.2005.02.011
- Dimsdale JE, Hartley LH, Guiney T et al. Postexercise peril. Plasma catecholamines and exercise. JAMA 1984;251:630–2. http://dx.doi.org/10.1001/jama.1984.03340290044018
- Fleg JL, Tzankoff SP, Lakatta EG. Age-related augmentation of plasma catecholamines during dynamic exercise in healthy males. J Appl Physiol 1985;59:1033–9.
- Balady GJ, Williams MA, Ades PA et al. Core components of cardiac rehabilitation/secondary prevention programs: 2007 update: a scientific statement from the American Heart Association Exercise, Cardiac Rehabilitation, and Prevention Committee, the Council on Clinical Cardiology; the Councils on Cardiovascular Nursing, Epidemiology and Prevention, and Nutrition, Physical Activity, and Metabolism; and the American Association of Cardiovascular and Pulmonary Rehabilitation. Circulation 2007;115:2675–82. http://dx.doi.org/10.1161/CIRCULATIONAHA.106.180945
- Futterman LG, Lemberg L. The clinical significance of exercise-induced ventricular arrhythmias. Am J Crit Care 2006;15:431–5.
- Sheps DS, Ernst JC, Briese FR et al. Decreased frequency of exercise-induced ventricular ectopic activity in the second of two consecutive treadmill tests. Circulation 1977;55:892–5. http://dx.doi.org/10.1161/01.CIR.55.6.892
- Morales FJ, Montemayor T, Martinez A. Shuttle versus six-minute walk test in the prediction of outcome in chronic heart failure. Int J Cardiol 2000;76:101–05. http://dx.doi.org/10.1016/S0167-5273(00)00393-4
- Troosters T, Gosselink R, Decramer M. Chronic obstructive pulmonary disease and chronic heart failure: two muscle diseases? J Cardiopulm Rehabil 2004;24:137–45. http://dx.doi.org/10.1097/00008483-200405000-00001

