A. Menarini Farmaceutica Internazionale SRL has provided an educational grant for the production of this e-learning programme and has had no editorial control or input. The views and content expressed within this programme are solely those of the authors.
PP-CA-UK-0157 Date of preparation: March 2020
Introduction
There are several sets of national and international guidelines for the treatment of chronic stable angina, a nomenclature which was altered in 2019 by the European Society of Cardiology (ESC) from stable coronary artery disease to chronic coronary syndromes.
The following section will focus mainly on the most recent UK guidelines (2011 but updated in 2016) from the National Institute for Health and Care Excellence (NICE)1, the European Society of Cardiology (ESC) Guidelines on Chronic Coronary Syndromes (2019),2 and the Scottish Intercollegiate Guideline Network (SIGN) (2018).3 Guidelines are also available from the American Heart Association/American College of Cardiology (AHA/ACC) but these will not be reviewed in this module.
Objectives for treatment
The goals of drug therapy for patients with stable angina pectoris are to:
- enhance quality of life through reduction of symptoms
- improve prognosis through reduction of risk of future myocardial infarction or death
- be well tolerated, and cause minimal side effects.
As emphasised by NICE,1 treatment and care should take into account angina patients’ individual needs and preferences. Good communication is essential, supported by evidence-based information, to allow patients to reach informed decisions about their care. If the patient agrees, families and carers should have the opportunity to be involved in decisions about treatment and care.
Cardiovascular disease (CVD) predominantly affects people older than 50 and age is the main determinant of risk. Apart from age and sex, three modifiable risk factors – smoking, raised blood pressure, and cholesterol – make a major contribution to CVD risk, particularly in combination. These account for 80% of all cases of premature coronary heart disease (CHD) (see Angina module 2: prevention). Aggressive risk factor modification is therefore vitally important for all angina patients. These are dealt with in detail in module 2 but are summarised later.
Secondary prevention treatment
A number of pharmacologic treatments are described as, ‘disease modifying’ in that they can improve outcomes in patients with stable CHD, and reduce risk of future cardiovascular events. The importance of angina treatment and risk factors for secondary prevention was highlighted in the 2019 update from the CLARIFY registry which showed that a previous myocardial infarction (MI) leads to a higher risk of future events, and residual angina leads to higher risk again. It also drew attention to the fact that blood pressure and cholesterol targets for secondary prevention are not being met in a large proportion of high-risk patients, and should be more aggressively treated.4
The following section reviews some of these ‘secondary prevention’ agents.
Antiplatelet and antithrombotic agents

All angina patients, unless there are contraindications, are recommended to take low-dose daily aspirin (figure 1). It is important to consider, however, any co-morbidities and bleeding risk. A meta-analysis5 of 16 secondary prevention trials in 17,000 individuals at high cardiovascular risk shows that aspirin therapy is associated with about a one fifth reduction in coronary events and in ischaemic stroke, the downside being a non-significant increase in haemorrhagic stroke. The absolute benefit seen in major coronary events in secondary prevention trials was an impressive 1% per year.
These findings are in contrast to primary prevention trials conducted in patients without prior CVD, where aspirin is of uncertain net value in preventing vascular events, as its effects are offset by an increase in major bleeds. This has been confirmed in a more recent meta-analysis6 of primary prevention trials showing that over six years, aspirin reduced total CVD events by about 10% but benefits were offset by major bleeding events. The message is therefore that:
- routine use of aspirin for primary prevention is not warranted and that;
- treatment decisions need to be decided on a case-by-case basis.
The optimal dose range in angina appears to be 75-150 mg/day and the lowest effective dose should be used in order to reduce gastrointestinal side effects. Evidence suggests that patients should remain on aspirin long-term; discontinuation may result in an increased risk of cardiovascular events.7 Non-adherence in patients is the most common reason for discontinuation of aspirin. Stopping aspirin increases the short-term risk of non-fatal MI or death from CHD by about 50% in primary care patients with a history of ischaemic events, according to one recent study.
The thienopyridine agent, clopidogrel 75 mg/day, might be considered as an alternative treatment in patients with aspirin intolerance at high risk of cardiovascular events.
Antithrombotic therapy in patients with stable angina and sinus rhythm may also be considered. The ESC guidelines2 give a class IIa recommendation to a second antiplatelet drug or low-dose rivaroxaban to patients at high risk (and IIb to moderate risk) in the absence of a high bleeding risk. In the presence of atrial fibrillation, long term anticoagulation is mandated regardless for stroke prevention, but the addition of an antiplatelet agent may be considered if ischaemic risk is judged to be high, and bleeding risk is not.
Lipid modifying agents
Using lipid modifying agents undoubtedly saves lives – up to 8,000 lives could be saved every three years by offering HMG-CoA reductase inhibitors – or statins – to anyone with a 10% risk of developing cardiovascular disease (CVD) within a decade, according to NICE. Offering statins to all eligible people could prevent up to 28,000 heart attacks and 16,000 strokes each year.
The most recent NICE clinical guideline (CG 181 published July 2014 and updated in September 2016) entitled ‘Cardiovascular disease: risk assessment and reduction, including lipid modification’8 updates and replaces the 2008 NICE clinical guideline 67 and the 2006 NICE technology appraisal guidance 94. It includes new and updated recommendations on risk assessment, lifestyle modifications and the use of lipid-lowering drugs.
Key recommendations in the NICE guidance include:
- identifying and assessing CVD risk using the QRISK2 assessment tool for the primary prevention of CVD in people up to the age of 84 years
- prioritising people for a full formal risk assessment if their estimated 10-year risk of CVD is 10% or more.
- taking a full lipid profile before starting lipid modification therapy for primary prevention. A fasting sample is not needed.
Blood cholesterol has a log–linear relationship to CHD risk and is a key modifiable risk factor. Blood cholesterol can be reduced by dietary change, physical activity and drugs.
Low-density lipoprotein cholesterol (LDL-C) remains the main target in most strategies of dyslipidaemia management. Statins are the treatment of choice and should be offered to patients with clinical evidence of cardiovascular disease.9
Findings from the Cholesterol Treatment Trialists Collaboration (CTTC) show the dose-dependent reduction in cardiovascular events with LDL-C lowering. Every 1.0 mmol/L reduction in LDL-C is associated with a corresponding 22% reduction in cardiovascular mortality and morbidity.10
The latest NICE guidance8 recommends starting statin treatment in people with cardiovascular disease with atorvastatin 80 mg. A lower dose of atorvastatin should be used if any of the following apply:
- potential drug interactions
- high risk of adverse effects
- patient preference.
It should be noted that the NICE guidance does not propose that statins should be used instead of the lifestyle adjustments that people at risk need to make, and encourages GPs to fully explore with their patients all the options promoted by the guidance, including lifestyle changes, blood pressure control, avoidance of diabetes and cholesterol (lipid) lowering.
With regard to other lipid-modifying agents, do NOT routinely offer fibrates, nicotinic acid (niacin), a bile acid sequestrant (anion exchange resin) or omega-3 fatty acid compounds for the prevention of CVD to any of the following:
- people being treated for primary or secondary prevention,
- people with CKD, type 1 or type 2 diabetes.
Tell patients that there is no evidence that omega-3 fatty acid compounds (fish oils) help to prevent CVD.
Do NOT offer the combination of a bile acid sequestrant (anion exchange resin), fibrate, nicotinic acid or omega-3 fatty acid compound with a statin for the primary or secondary prevention of CVD.
Do NOT offer coenzyme Q10 or vitamin D to increase adherence to statin treatment.
Targets
The 2019 ESC guidelines on dyslipidaemia management11 do not provide target total cholesterol (TC) levels and, instead, recommend TC as a component of the SCORE system and recommend the following LDL-C targets:
- individuals with a very high cardiovascular risk, a target of <1.4 mmol/L or a reduction of at least 50% of baseline LDL-C for both primary and secondary prevention
- individuals with a high cardiovascular risk, a target of <1.8 mmol/L or a reduction of at least 50% of baseline LDL-C e.g. markedly elevated single risk factor
- individuals with a moderate cardiovascular risk, a target of <2.6 mmol/L.
The SCORE system estimates the 10-year risk of a first fatal atherosclerotic event.11 Risk charts like SCORE are intended to determine risk estimation in apparently well individuals with no signs of clinical or pre-clinical disease. Patients who have angina or who have had an event, e.g. MI or stroke, automatically qualify for intensive risk factor evaluation and management. Also see risk estimation using the new Joint British Societies (JBS3) in module 2 of this programme.
Strategies aimed at elevating high-density lipoprotein cholesterol (HDL-C) levels have attracted interest but their effects in reducing the progression of coronary atherosclerosis and preventing future cardiovascular events in CHD patients are equivocal. While newer drugs to raise HDL-C are in development, lowering of LDL-C should remain the main therapeutic focus in patients with stable CHD. HDL-C is not recommended as a target for treatment.
No specific targets for HDL-C or triglycerides (TGs) have been determined in clinical trials, although low HDL-C and high TG levels are associated with excess events and increased cardiovascular mortality. A European Atherosclerosis Society Consensus Panel has considered interventions in patients with HDL-C <1.0 mmol/L and TG >1.7 mmol/L to facilitate cardiovascular risk reduction, particularly among those with cardiometabolic abnormalities.12
The ESC guidelines furthermore mandate the titration of statins to the highest tolerable dose in order to achieve the target LDL-C. They also recommend the addition of ezetimibe and thereafter PCSK9 inhibitors if LDL-C targets are not achieved on statin therapy alone. The role of bempedoic acid has not been addressed in the current guideline structure, and its routine use is not yet recommended.
Angiotensin-converting enzyme inhibitors
Aggressive blood pressure reduction to <130/85 mmHg is optimal for patients with stable CHD, especially in those with other co-morbidities including diabetes (and chronic kidney disease), where lower treatment levels, i.e. <130/80 mmHg are warranted.1-3
Angiotensin-converting enzyme (ACE) inhibitors, e.g. ramipril, perindopril, are widely used. It is appropriate to consider ACE inhibitors for the treatment of patients with stable angina and co-existing hypertension, diabetes, heart failure, asymptomatic left ventricular (LV) dysfunction and post-MI. In angina patients without co-existing indications for ACE-inhibitor treatment, the anticipated benefit of treatment (possible absolute risk reduction) should be weighed against costs and risks for side effects, and the dose and agent used of proven efficacy for this indication. Angiotensin receptor blocking (ARB) drugs may be used in patients in whom ACE inhibitors are poorly tolerated.
Lifestyle measures
Lifestyle interventions in people at high risk of or with CVD are addressed in detail in Angina module 2: prevention. They can be summarised as follows:
Physical activity
Physical activity within the patient’s limitations should be encouraged, as it may increase exercise tolerance, reduce symptoms and has favourable effects on weight, blood lipids, blood pressure (figure 2), glucose tolerance, and insulin sensitivity.
Psychosocial stress
Although the role of stress in CVD causation is controversial, there is no doubt that psychological factors are important in provoking angina. Furthermore, the diagnosis of angina often leads to excessive anxiety. Patient reassurance is essential. Patients may benefit from relaxation techniques and other methods of stress control.
Diet
People should be advised to eat a diet in which:
- total fat intake is 30% or less of total energy intake
- saturated fats are 10% or less of total energy intake
- dietary cholesterol is less than 300 mg/day
- saturated fats are replaced by monounsaturated and polyunsaturated fats.
Advise eating at least:
- five portions of fruit and vegetables per day
- two portions of fish per week, including a portion of oily fish.
Smoking cessation
Smokers should be advised to stop. To help people wanting to stop:
- offer support and advice
- offer referral to an intensive support service (for example, NHS Stop Smoking Services)
- if they do not, or cannot, take up the referral for intensive support, offer pharmacotherapy in line with locally approved ‘Smoking cessation services’.
Lipid modification and blood pressure control (see above)
Treatment with antianginals
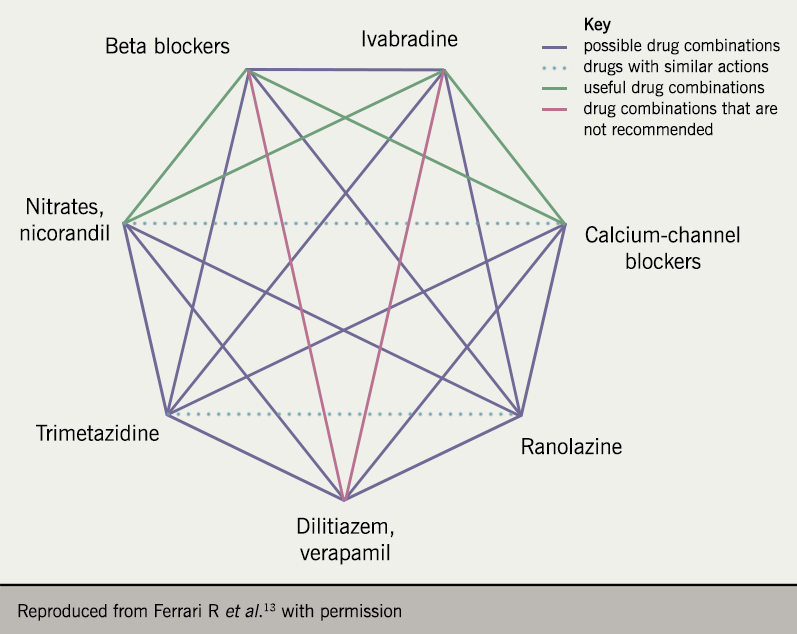
Symptoms of angina pectoris and myocardial ischaemia, are reduced by drugs that reduce myocardial oxygen demand and/or increase blood flow to the ischaemic territory. Nitrates, beta blockers and calcium antagonists are commonly seen and recommended as first-line pharmacotherapy. This section will review these agents in regards to monotherapy, as well as second-line drug classes, such as the potassium channel opener, nicorandil, the sinus node inhibitor, ivabradine, and the late sodium current inhibitor, ranolazine. These second-line more novel agents may have more robust evidence base compared to first-line agents, which have had more classical historical use. Treatment with multiple agents should also be considered to personalise treatment regimes to the patient – the so called ‘diamond approach’ of combination therapy (figure 3).13 Guidelines differ in regards to the approach to combination therapy, which we will review.
Note: while every effort is made to provide current and accurate advice, the authors cannot be responsible for inaccuracies or prescribing changes which may occur and we recommend that prescribers consult the summary of product characteristics (SPC) for any of the drugs reviewed.
Nitrates
Clinical indications
Short-acting, sublingual, or buccal (administered between the upper lip and the gum) glyceryl trinitrate GTN) tablets or spray should be used to prevent exertional anginal attacks and for immediate pain relief. All angina patients should be offered such ‘rescue’ nitrates.
Long-acting nitrates are available orally, or they may be delivered via an adhesive patch. Treatment with long-acting nitrates reduces the frequency and severity of anginal attacks, and may increase exercise tolerance. Nitrates do not reduce mortality or the incidence of MI in patients with stable angina.
Nitrates may, however, play an important role in pain relief in the early phase of acute MI. They are also potent platelet inhibitors and therefore should also be considered as first line agents in acute coronary syndromes.
Mode of action
Nitrates mimic the actions of endogenous nitric oxide (NO) by releasing NO or formation of NO in tissues, via an enzymatic process. They have a direct relaxant effect on vascular smooth muscle cells where they are metabolically converted to nitric oxide (NO) and dilatation of coronary vessels improves myocardial oxygen supply. They also dilate peripheral veins thereby reducing preload and afterload to reduce myocardial oxygen consumption.14
Pharmacological profile
The pharmacokinetics of GTN are not completely understood. The substance disappears rapidly from plasma largely by extrahepatic mechanisms. GTN is quickly and completely absorbed through the skin and all mucosal surfaces. It has a half-life of about 1–4 minutes. GTN is approximately 60% bound by plasma proteins, the dinitrate active metabolites are 30 to 6% bound.
Much metabolism, especially first-pass conversion of ingested drug, occurs in the liver. These dinitrates are less potent than the parent compound, but because their half-lives are 1-3 hours, they probably account for most of the prolonged activity of the drug.
Drug interactions: most cardiovascular drugs have the potential for multiple interactions. It is advisable to consult the Interactions Appendix of the British National Formulary (BNF).
A principal drug interaction of concern is that with nitrates and phosphodiesterase-5 inhibitor agents, sildenafil, vardenafil and tadalafil, used for treatment of erectile dysfunction. These should not be co-administered as the hypotensive effects of nitrates may be enhanced.
Clinical profile
Short-acting nitrates improve exercise tolerance when taken prophylactically. Both short- and long-acting preparations are widely used and, while sublingual tablets are rapidly effective for treating an acute attack, many patients find the spray more convenient. As nitrate levels are not sustained, tolerance to sublingual GTN does not occur but, because the effects are correspondingly short-lived, it is not possible to provide continuous prophylaxis. To circumvent this problem, a number of long-acting preparations have become available.
The limitation of this long-acting approach is nitrate tolerance,15 a phenomenon which appears to develop rapidly. Patients treated with long-acting nitrates should have a ‘nitrate free’ interval each day to preserve the therapeutic effects. This may be achieved with appropriate timing of doses of intermediate acting isosorbide dinitrate (ISDN) or with formulations of isosorbide mononitrate (ISMN) that provide a suitable plasma concentration profile.
Various oral nitrate preparations (table 1) are available.
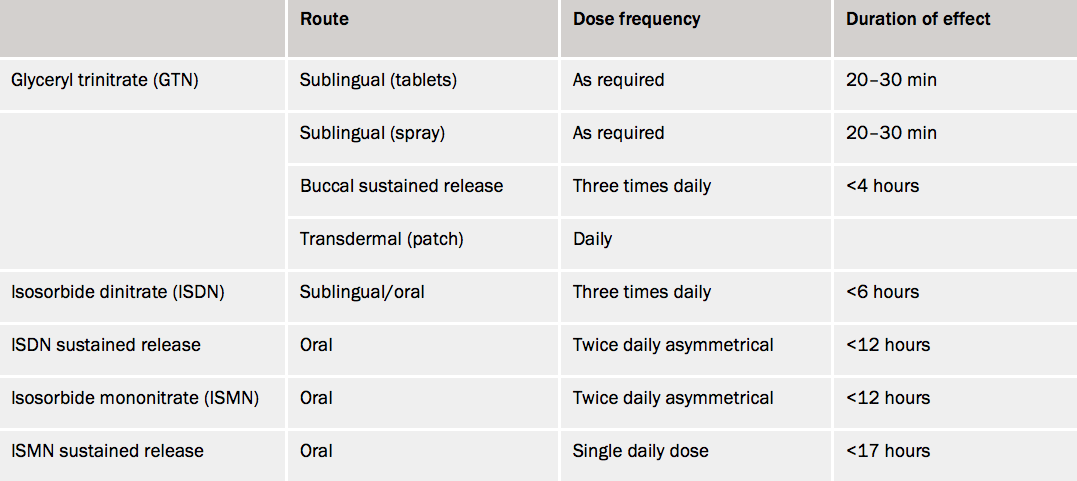
Oral isosorbide dinitrate (ISDN) is similar to GTN but with a longer duration of action of two to three hours. ISDN is typically taken orally as 20 or 40 mg twice daily. It undergoes extensive ‘first pass’ hepatic metabolism and its bioavailability is only about 25%, unlike its metabolite, isosorbide mononitrate. Isosorbide mononitrate (ISMN) is 100% bioavailable. ISMN is usually taken two to three times a day, e.g. 20 mg tds or 40 mg bd, although more expensive once-daily sustained-release preparations can be used. Patients on ISMN twice daily should take the second dose at 4 pm, and not at bedtime.
Transdermal GTN patches overcome some of the disadvantages of the now rarely used nitrate ointment. Because of more gradual absorption, the GTN patch may be particularly suitable for the patient who develops headaches with oral formulations. Tolerance to continuous oral or transdermal nitrates develops rapidly and, in order to overcome this, nitrate-free intervals or ‘drug holidays’ whereby patients remove the patch during part of the day or at night, or a modified delivery system designed to provide a low-nitrate period, have been recommended.
The worry about intermittent nitrate therapy is that rebound exacerbation of symptoms may occur and that the patient may be unprotected in the early hours of the morning, when there is a well-documented increase in the incidence of myocardial ischaemia, MI and sudden cardiac death.16 Another class of anti-anginal agent, therefore, such as a long-acting beta blocker or calcium antagonist, should be given in addition to nitrates.
When nitrates are used to treat episodes of angina it is important to advise patients to:
- repeat the dose after five minutes if the pain has not gone
- call an emergency ambulance if the pain has not gone five minutes after the second dose.
Safety, tolerability and side effects
The safety of nitrates has been established with long-term use. Only about 10% of patients are non-responders to nitrate therapy and a further 10% experience side effects requiring withdrawal of therapy.17
Side effects include postural hypotension, syncope and headache. It is therefore best to start with a low dose, increasing it once it is well tolerated. Reflex tachycardia may decrease coronary blood flow and worsen the angina. Headache may wear off but if this is intolerable alternative treatments will be required.
Occasionally, postural hypotension can occur and nitrates should not be co-administered with phosphodiesterase-5 inhibitors, such as sildenafil. Nitrate tolerance can develop rapidly with continuous use and may be lessened by using preparations which provide nitrate-low or nitrate-free intervals.
Molsidomine is a vasodilator which is nitrate-like with similar efficacy, but it is not available in many countries.
As with all drugs, nitrates have their benefits and limitations. These are some of the pros and cons of using nitrates in stable angina.
Pros:
- fast, effective symptom relief
- may be used before an anticipated attack
- long-acting nitrates may reduce frequency of attacks
- no serious side effects (though headache and flushing may occur)
- GTN is cheap.
Cons:
- sublingual nitrate only lasts for 20 minutes
- no prognostic benefit
- rapid development of tolerance is common
- “nitrate-free interval” required to avoid tolerance
- cannot be used in combination with drugs for erectile dysfunction.
Beta blockers
Clinical indications
Beta adrenergic receptor antagonists (beta blockers) are first-line therapy in angina. They are used for the prophylaxis of exertional angina rather than the alleviation of an acute attack.
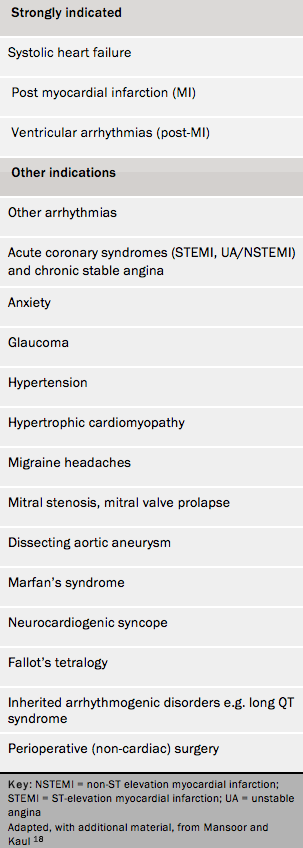
In addition to their traditional role in treating angina, hypertension and other cardiovascular diseases, beta blockers are used for many additional disorders (table 2). Beta blockers in general, and atenolol in particular, are no longer recommended as first-line therapy for the treatment of hypertension.
They are an important treatment for several supraventricular and ventricular tachyarrhythmias and they significantly reduce mortality in heart failure. There are no randomised trials showing improved survival with beta blockers in chronic stable angina, however, they do significantly reduce cardiovascular events in post-MI patients.15,18
Mode of action
Beta blockers have been available for about 50 years and were originally developed by British clinical pharmacologist and Nobel Prize winner, Sir James Black (figure 4). They are widely used for the treatment of angina. In developing the drug class it was hypothesised that the syndrome of angina was primarily due to stimulation of the heart via the sympathetic nervous system (SNS), and that a drug which inhibited this effect would be clinically useful.
If you are familiar with the concept of alpha and beta adrenergic receptors and their blockade you may wish to move straight to the ‘Principal actions’ section, below.
Adrenergic nerves and receptors
Activation of the SNS as a result of physical exertion or psychological stress, for example, leads to the release of neurotransmitter, catecholamines (commonly referred to as “fight or flight hormones”), such as norepinephrine and epinephrine (previously called noradrenaline and adrenaline) from the adrenal medulla and the post-ganglionic fibres of the SNS. These catecholamines act on alpha– and beta-receptors.
Norepinephrine activates primarily alpha receptors and epinephrine activates primarily beta receptors (although it may also activate alpha receptors).
Alpha receptor stimulation is associated with constriction of small blood vessels in the bronchial mucosa and relaxation of smooth muscles of the intestinal tract.
Beta receptor stimulation relaxes bronchial smooth muscles which causes bronchial dilatation. In addition, beta receptor stimulatory effects cause an increase in the rate and force of heart contractions.
There are drugs which block both alpha and beta receptors, but this programme concentrates on beta blockade.
There are three distinct types of beta receptor:
- beta-1 found mainly in cardiac muscle
- beta-2 present in cardiac muscle but more prominent in bronchial and peripheral vascular smooth muscle. Activation of these receptors results in vasodilatation and bronchodilatation
- beta-3 found in adipose tissue and the heart. Their activation may mediate catecholamine-induced thermogenesis and may reduce cardiac contractility.
Beta blockers act predominantly by competitively inhibiting catecholamines from binding to these beta receptors, particularly beta-1 cardiac receptors (figure 5).
The principal anti-anginal actions of beta blockers are:
- slowing the heart rate increase, in response to exercise and emotion
- reducing cardiac output; thus, myocardial oxygen demand is reduced and coronary blood flow is increased via increased filling time in diastole
- lowering blood pressure (which also conserves energy expenditure).
It is conventional to adjust the dose of beta blocker to reduce resting heart rate to 55–60 beats/minute (bpm), and a peak exercise heart rate of <110 bpm. Evidence suggests that high resting heart rates carry an adverse prognosis and that heart-rate lowering per se may be beneficial.19
Pharmacological profile
Some beta blockers are water soluble (hydrophilic), e.g. atenolol, and tend to have longer half-lives and are renally excreted. Lipid soluble (lipophilic) agents, e.g. metoprolol, are mainly hepatically metabolised and have shorter half-lives. Central nervous system (CNS) side effects, such as vivid dreams and depression, may be associated with lipophilic agents which pass the blood–brain barrier, and such problems may be substantially reduced or abolished by switching to more water-soluble drugs.
Drug interactions: Most cardiovascular drugs have potential for multiple interactions. It is advisable, therefore, to consult the Interactions Appendix of the British National Formulary (BNF).
Caution is needed, for example, if combining beta blockers with other agents which reduce heart rate such as diltiazem, verapamil, ivabradine, amiodarone, digoxin and also those which lower blood pressure.
What dose to use
The biological half-life of beta blockers exceeds the plasma half-life considerably18 so a drug such as propranolol, with a half-life of 3 hours is actually taken twice daily. Also, the higher the dose used, the longer the biological effect.
To achieve 24-hour efficacy a beta-1 blocker with a long half-life (e.g. bisoprolol) or a formulation providing an extended plasma concentration, e.g. metoprolol CR, may be used. For atenolol (half-life of 6–9 hours), twice-daily dosing may be better, but increasing the dose also extends the duration of action. Target doses for full anti anginal effects are:
- bisoprolol 10 mg od
- metoprolol CR 200 mg od
- atenolol 100 mg/day od (or 50 mg bid).
What is cardioselectivity?
The cardioselective agents, e.g. atenolol and bisoprolol, are so called because they preferentially bind to beta-1 receptors, overall, cause less frequent adverse effects such as bronchoconstriction or Raynaud’s phenomenon (i.e. cold extremeties), which are mediated via beta-2 receptor blockade.
However, beta-1 selectivity is only a relative characteristic because, at moderate-to-high doses, beta-2 receptor blockade also occurs even with so-called cardioselective agents. Beta blockers are therefore best avoided in patients who may be susceptible to asthma. Chronic obstructive pulmonary disease (COPD) is a relative contraindication to beta blockers. Cardioselective beta blockers should not be withheld from patients with COPD.
Partial agonism
For example, agents which have intrinsic sympathomimetic activity (ISA), also called partial agonist activity (PAA), have less effect on resting heart rate and not only block beta receptors but also stimulate them, depending on the prevailing level of sympathetic activity. These agents, e.g. pindolol, cause less bradycardia and may offer advantages to patients with peripheral vascular disease, although they tend to be less effective as an anti-anginal prophylaxis.
Clinical profile
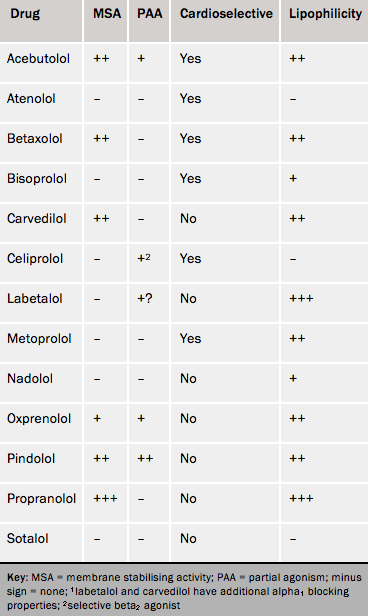
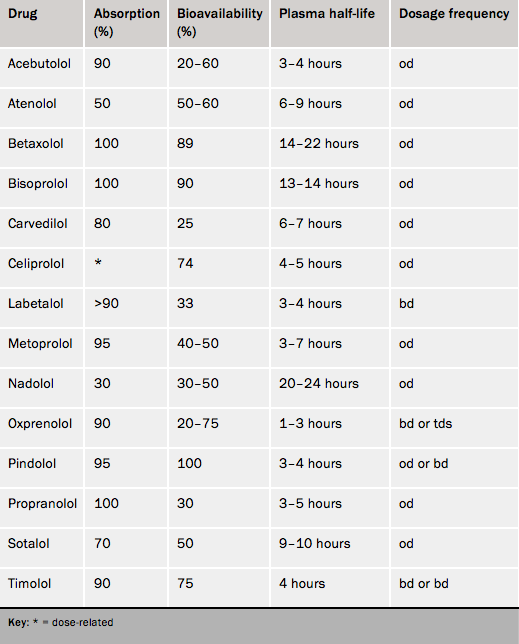
There are over 30 widely available beta blockers and, although they share general beta receptor blocking properties, they have pharmacological differences (tables 3 A and B), so a number of additional considerations need be taken into account when selecting drug treatment of the individual patient.20 We will focus on the beta blockers used widely to treat angina, such as atenolol, bisoprolol, metoprolol, etc. Notably, the highly selective beta blocker, nebivolol, which was widely used for treating hypertension and also heart failure, is available generically in the UK.
Beta blockers in stable angina
Beta blockers are acknowledged as first-line therapy in effort-induced chronic stable angina in all patients in the absence of contraindications.1–3 An improvement in exercise tolerance has been shown with beta blocker use in patients with coronary artery disease (CAD). In contrast, however, beta blockers can cause a reduction in exercise endurance in young healthy subjects and trained sportsmen.21
Safety, tolerability and side effects
About 15–20% of patients will not tolerate long-term use of beta blockers.19 The most common adverse effects include fatigue, dizziness, weakness, depression and, particularly with the lipid soluble agents, CNS problems such as vivid dreams and insomnia. Care must be taken to observe the contraindications to beta blockade, such as heart block or asthma.
While beta blockers reduce cardiac contractility because they reduce sympathetic activity (which is increased as a compensatory response) in patients with impaired left ventricular function, they have a beneficial role when introduced gradually in heart failure.
Beta blockers can adversely affect blood glucose levels and lipid profiles, which may be of particular importance in the diabetic patient; some agents may also mask or prolong hypoglycaemia.
Beta blockers should be avoided in cases of prinzmetal vasospastic angina as they may worsen coronary spasm.22
It should be noted that sudden withdrawal of beta blockers may occasionally result in rebound angina, presumably as a consequence of receptor upregulation and thereby enhanced sympathetically-mediated responses.
Here are some of the main advantages and disadvantages of using beta blockers for patients with stable angina.
Pros
- survival benefit shown in post-MI patients
- improve outcome in heart failure
- provide symptom relief and improve exercise tolerance.
Cons
- important contraindications and cautions
- use limited by side effects and up to 20% of patients cannot tolerate them
- rebound angina if withdrawn suddenly
- may cause metabolic e.g. lipid, glucose disturbances.
Calcium antagonists
Clinical indications
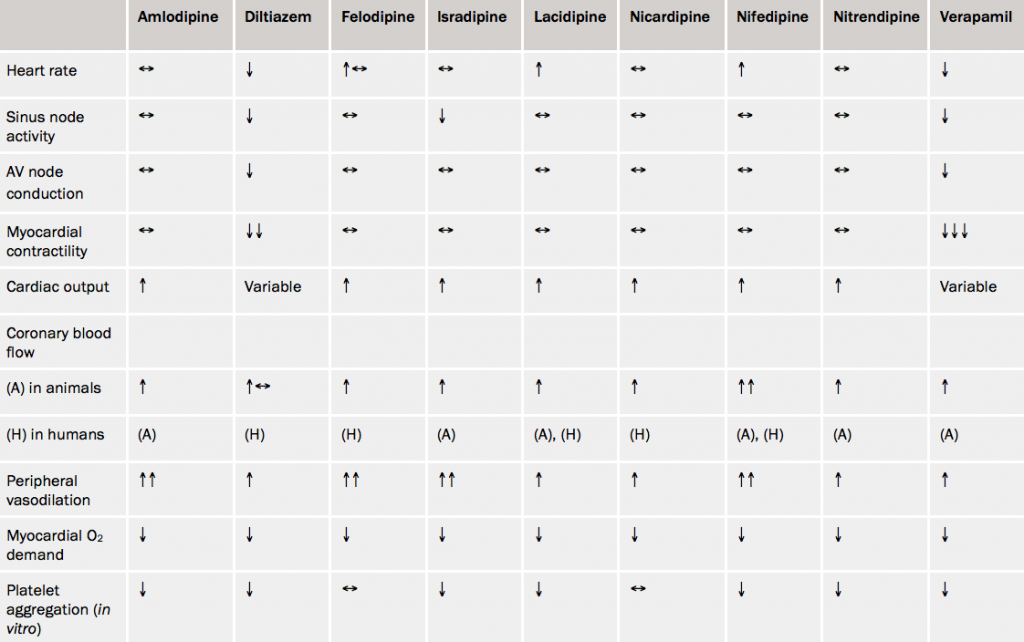
Calcium antagonists are widely used for angina prophylaxis and relief of coronary spasm including Prinzmetal angina, as well as for treatment of hypertension, and may additionally have use in treatment of coronary microcirculatory dysfunction.23 There are three main classes, the prototype drugs are verapamil, diltiazem and nifedipine, and all these agents and their analogues are effective anti-anginals.
Other uses for calcium antagonists include the treatment of coronary spasm, supraventricular arrhythmias, where verapamil and diltiazem are useful in controlling the ventricular rate. Calcium antagonists may also improve symptoms of dyspnoea, angina and dizziness in patients with hypertrophic cardiomyopathy.
Mode of action
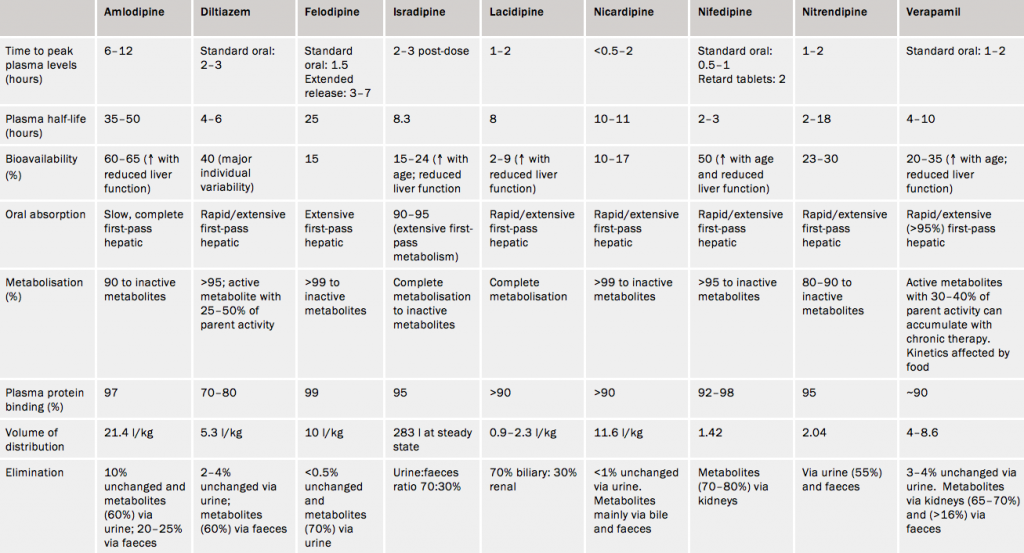
Calcium antagonists are structurally and pharmacologically heterogeneous compounds (tables 4 and 5) which share a common property – to inhibit entry of calcium into cells (thus they are sometimes called, calcium channel blockers) resulting in vasodilatation of vascular smooth muscle and relaxation of myocardial muscle.24
Their anti-anginal effects appear to be mediated through a reduction of myocardial oxygen demand secondary to decreased afterload and myocardial contractility.
In classifying agents that inhibit the movement and binding of calcium, the World Health Organisation (WHO) has identified two predominant types of calcium antagonist which are, selective for L-type (long-lasting, large-current, or slow), voltage-dependent calcium channels, and those that are nonselective.
Selective calcium antagonists include three discrete chemical types:
- phenylalkylamines (e.g. verapamil – relatively myocardium selective)
- benzothiazepines (e.g. diltiazem – intermediate selectivity)
- 1,4-dihydropyridines (e.g. nifedipine, amlodipine – high vascular selectivity).
The selective calcium antagonists share a similar mechanism of action in that they inhibit the influx of extracellular calcium through the L-type channel, resulting in relaxation of vascular smooth muscle, and increase in arterial diameter, causing reduction in vascular resistance, and blood pressure lowering. Decrease in cellular calcium also leads to a reduction in cardiac contractility, thereby reducing cardiac oxygen demand (anti-anginal). There are other calcium channels, notably the T-type (Transient) channel, which are short lasting and function at a lower voltage, which can be reviewed elsewhere.25
Non-selective agents include, bepridil, but further discussion of these is not relevant for this module. Because calcium antagonists are a heterogeneous group of compounds, with marked differences in chemical structure, binding sites, and tissue selectivity, they possess different clinical activity and therapeutic indications.
Pharmacological profile
The pharmacokinetics of the three original calcium antagonists and five newer dihydropyridines are compared in table 4. Calcium antagonists are metabolised in the liver and undergo extensive and variable first-pass metabolism, which limits the amounts reaching the systemic circulation. Since individual metabolism is unpredictable, dose titration of these drugs is essential for optimal response.
The extensive metabolism leads to a short duration of action in (with obvious exceptions such as amlodipine which has less first-pass metabolism) conventional formulations. Several agents are available in slow- or sustained-release formulations allowing for once- or twice-daily treatment. Calcium antagonists do not adversely affect lipids or serum glucose and are therefore more suitable than beta blockers in diabetic patients with angina, for example.
Drug interactions: Most cardiovascular drugs have potential for multiple interactions. It is advisable therefore to consult the Interactions Appendix of the British National Formulary (BNF).
Calcium antagonists may be involved in significant interactions with other drugs. For example, cimetidine inhibits the metabolism of nifedipine and accentuates its clinical effect. Nifedipine, verapamil and diltiazem have also been shown to elevate serum digoxin concentrations.
Clinical profile
Although chemically and structurally distinct, the pharmacologic profiles of verapamil and diltiazem (which are sometimes referred to as non-dihydropyridines) are much closer to one another than either is to the dihydropyridines.
Diltiazem and verapamil reduce heart rate (i.e. negatively chronotropic) and blood pressure, and are negatively inotropic, reducing cardiac output (thus contraindicated in heart failure). They may also impair atrioventricular conduction. Calcium antagonists may retard the atherosclerotic process, but more complete clinical data are required to establish if they have any role other than symptom control in angina.
What doses to use
Diltiazem, is prescribed typically at doses of 60 mg three-times daily or longer acting preparations 120 mg once daily, and verapamil 80–120 mg three-times daily; or longer acting formulations, 240 mg twice daily, are generally as effective as beta blockers in monotherapy or they may have additive anti-ischaemic effects if combined. Verapamil should not be combined with beta blockers and caution should be used when combining beta blocker and diltiazem. Care is needed not to reduce the heart rate excessively. Adverse effects include flushing, dizziness, constipation and fluid retention.
Dihydropyridine calcium antagonists
These include nifedipine and amlodipine, which have minimal effect on heart rate or cardiac contractility. Short-acting preparations of nifedipine are inappropriate to use as they cause reflex tachycardia. Amlodipine has a uniquely long half-life of about 45 hours, and a slow onset of action, and amlodipine 5–10 mg once daily can also be used to treat stable angina, and also angina in patients with stable heart failure. Similarly, felodipine 5–10 mg is also available for the treatment of angina and hypertension.
Safety, tolerability and side effects
There has been considerable controversy about the safety of calcium antagonists and the suggestion of an increased risk of MI with usage.26 But the controversy overlooked the clear differences between various formulations. With the older short-acting formulation of nifedipine, for example, there is a rapid onset of action and pronounced dose-related fluctuations in the vasodilator effect which provokes stimulation of the sympathetic nervous system and a resulting reflex tachycardia (increasing oxygen demand). Long-acting agents, such as amlodipine, or the newer formulations of nifedipine, have a protracted and more consistent duration of action with little or no reflex tachycardia. Meta-analysis of the use of nifedipine monotherapy or short-acting nifedpine in combination with other antianginal therapy indicated that there may be an increase in cardiovascular events, specifically anginal attacks.27
Rate-limiting calcium channel antagonists, such as dilatiazem and verapamil, are contra-indicated in patients with systolic heart failure, bradycardia and high degree atrioventricular block.28
The PRAISE (Prospective Randomized Amlodipine Survival Evaluation) study29 showed that amlodipine did not increase mortality and can be used to treat angina in this setting. There is no role, however, for dihydropyridine calcium antagonists in the treatment of unstable angina.
Side effects including flushing, nausea, headache, dizziness, tachycardia, palpitations, gastrointestinal disturbances, are common with most calcium antagonists and other drugs which lower blood pressure. Diltiazem and verapamil may cause bradycardia and flushing or rash; while constipation is sometimes seen with verapamil.
Calcium antagonists appear to cause few metabolic disturbances and seem to be ‘lipid friendly’. Similarly, they may be used in patients with diabetes and peripheral vascular disease, but caution is again needed when treating patients with heart failure (see below). Long-acting, once-daily agents may improve patient compliance.
Here are some of the pros and cons of using calcium channel blockers in the treatment of stable angina.
Pros
- generally well tolerated
- metabolically ‘neutral’
- useful in coronary spasm
- an alternative for patients who cannot tolerate beta blockers.
Cons
- no evidence of survival benefit
- avoid co-administration of verapamil and diltiazem with beta blockers and ivabradine
- side effects include severe peripheral oedema and flushing.
Potassium channel openers
Clinical indications
There is really only one widely used clinical agent in this family, nicorandil. It is a hybrid compound which is a potassium channel opener with a nitrate moiety (figure 6). It is prescribed for the prophylaxis and treatment of angina. Nicorandil may suffice as monotherapy in some, perhaps more mildly symptomatic, patients. It may be less useful for those who are headache-prone. It can be used safely as an ‘add on’ to beta blockers and calcium antagonists (but this is an unlicensed indication in the UK) and may be beneficial in patients who cannot tolerate either of these other agents, particularly those who feel lethargic on beta blockade, or have profound ankle oedema with a calcium antagonist.
Nicorandil may also have a role in individuals with obstructive airways disease, dyslipidaemia or diabetes, where a beta blocker is inappropriate. Similarly, it may be useful in patients who are difficult to manage with drugs which increase heart rate or, possibly, where nitrate tolerance is a problem.
Mode of action
The adenosine triphosphate-sensitive (ATP-sensitive) potassium channel (sometimes called the ATPK-sensitive channel) was discovered in 1983 and the first potassium channel opener nicorandil was licensed in Europe for treatment of angina in 1994. The compound consists of a nicotinamide vitamin group and an organic nitrate, and has a novel haemodynamic and vasodilator profile. Potassium channels, particularly those regulated by intracellular ATP, are ubiquitous in the heart and blood vessels and are important modulators of cardiovascular function.
On opening potassium channels there is an increased efflux of potassium ions from the cell, and the resting membrane potential is shifted towards more negative values (hyperpolarisation); this leads to an inhibition of calcium influx or indirect calcium antagonism, causing a fall in intracellular calcium concentration, relaxation of vascular smooth muscle cells and vasodilatation.
Nicorandil has dual mechanism of action on both preload and afterload, producing dose-related improvement in haemodynamics. Angiographic studies have shown that the drug dilates both stenotic and non-stenotic coronary arteries. Unlike classical nitrates there appears to be an absence of haemodynamic tolerance to nicorandil.
Nicorandil may mimic ischaemic preconditioning, which is a powerful protection against myocardial necrosis.
Nicorandil causes reduction in total peripheral resistance and blood pressure but at therapeutic concentrations this effect is small and transient and does not lead to first-dose hypotension. However, it may be appropriate when starting therapy to advise patients who are susceptible to nitrate-induced or non-specific headache to initiate treatment with low doses.
Pharmacological profile
Nicorandil is metabolised in liver and excreted as metabolites in urine. It has 75–80% bioavailability and absorption is not affected by food. Nicorandil has a half-life of one hour and is used as a twice-daily drug.
Drug interactions: most cardiovascular drugs have potential for multiple interactions. It is advisable therefore to consult the Interactions Appendix of the British National Formulary (BNF).
There are few major interactions with nicorandil but the same restrictions apply as for nitrates with avoiding co-administration of phosphodiesterase inhibitors.
Clinical profile
Nicorandil has similar anti-anginal activity and produces similar improvements in exercise capacity as nitrates, beta blockers and calcium antagonists.30 The Impact Of Nicorandil in Angina (IONA) study31 showed that nicorandil 20 mg twice daily, in addition to standard anti-anginal therapy, improved outcomes in terms of reducing events related to acute coronary disease and the associated requirement for admission to hospital.
The drug may therefore have a useful role in the treatment of angina which is unresponsive to initial medical treatment. Nicorandil is not approved for use in patients with low systolic blood pressure, acute pulmonary oedema or ventricular failure.
What doses to use
Nicorandil is a twice-daily drug (half-life of one hour). It should be initially started at a dose of 10 mg bd or 5 mg bd if the patient is prone to headache. The usual dose is nicorandil 20 mg bd, and it can be used to a maximum dose of 30 mg bd.
Safety, tolerability and side effects
Headache is the problem most commonly encountered, usually occurring early on commencement of treatment and disappearing with chronic dosing. The problem can be minimised by reducing the initial dose of 5 mg twice daily. Adverse events usually occur early in the course of treatment and, after 30 days, fewer than 10% of patients report adverse events. Nicorandil is generally well tolerated and dizziness, flushing and oedema are rarely observed. Further rare side effects include oral (aphthous stomatitis) and anal ulceration but this is reversible on cessation of the drug. Due to the safety concerns in regards to gastrointestinal ulceration, the MHRA issued a document in 2016 concerning the the use of nicorandil, restricting it to second-line therapy, that it should be used only if beta blockers or calcium channel blockers are contra-indicated or if the patient is intolerant of first line therapy.32
The potassium channel opener nicorandil has certain advantages and disadvantages.
Pros
- no haemodynamic tolerance
- side effect of headache usually disappears with long-term use
- IONA trial showed that drug improves outcomes
- useful in both prophylaxis and treatment.
Cons
- headache is a common side effect when starting treatment
- limiting side effect is hypotension
- contraindicated in cardiogenic shock
- cannot co-administer with phosphodiesterase inhibitors
- risk of gastrointestinal ulceration limits its use to second line.
Sinus node inhibitors
Ivabradine, the first clinically available sinus node inhibitor, is licensed for the symptomatic treatment of chronic stable angina pectoris in adult patients with normal sinus rhythm and ≥70 bpm.
It is indicated in adults with a contraindication or intolerance for beta blockers; or in combination with beta blockers in patients inadequately controlled with an optimal beta blocker dose.
Ivabradine is also licensed for the treatment of chronic heart failure (New York Heart Association [NYHA] II to IV class) with systolic dysfunction, when patients are in sinus rhythm and whose heart rate is ≥75 bpm, in combination with standard therapy including beta-blocker therapy or when beta-blocker therapy is contraindicated or not tolerated.
Ivabradine should be used as per:
- the recommended starting dose is 5 mg twice daily
- the maximum maintenance dose of 7.5 mg twice daily should not be exceeded
- down-titrate the dose if resting heart rate decreases persistently below 50 bpm or if the patient experiences symptoms of bradycardia. The dose can be down-titrated to 2.5 mg twice daily if necessary
- stop ivabradine treatment if the resting heart rate remains below 50 bpm or symptoms of bradycardia persist
- only start ivabradine if the resting heart rate is at least 70 bpm
- do not prescribe ivabradine with other medications that cause bradycardia, such as verapamil, dilatiazem or strong CYP3A4 inhibitors
- monitor patients regularly for atrial fibrillation and if this occurs, carefully reconsider whether the benefits of continuing ivabradine treatment outweigh the risks
- consider stopping ivabradine if there is no or only limited symptomatic improvement after three months.
The European Medicines Agency (EMA) has made recommendations on the use of ivabradine in the treatment of angina,33 based on the review of the final data from SIGNIFY (Study Assessing the morbidity–mortality Benefits of the If Inhibitor Ivabradine in Patients with Coronary Artery Disease).34 This was a randomised placebo-controlled study conducted in 19,102 CAD patients without clinical heart failure. It showed that in a subgroup of patients there was a small but significant increase in the combined risk of cardiovascular death or non-fatal heart attack with ivabradine compared with placebo (3.4% vs. 2.9% yearly incidence rates). The data also indicated a higher risk of bradycardia with ivabradine compared with placebo (17.9% vs. 2.1%).
Patients in SIGNIFY were started on a higher than recommended dose of ivabradine and received up to 10 mg twice a day, which is higher than the currently authorised maximum daily dose (7.5 mg twice a day).
Mode of action
Heart rate is controlled by inhibiting the If current, which plays a central role as a pacemaker in the sinoatrial node, a zone of impulse-generating tissue located in the right atrium of the heart. Ivabradine acts on the If ion current (figure 7). The ‘f‘ is for ‘funny’, so named because it had unusual properties compared with other ion currents, and which is highly expressed in the sinoatrial node. If is a mixed sodium-potassium (Na+–K+) inward current activated by hyperpolarisation and modulated by the autonomic nervous system.
Ivabradine selectively inhibits the pacemaker If current in a dose-dependent manner. Blocking this channel reduces pacemaker activity (reducing the slope of the diastolic depolarisation curve), slowing the heart rate thus reducing myocardial oxygen demand, while simultaneously improving supply.
Ivabradine is a pure bradycardic agent which, at a maximum dose of 7.5 mg twice daily, decreases heart rate comparably to a beta blocker, but has no effect on systemic blood pressure. It is not negatively inotropic but has effective anti-ischaemic and anti-anginal activity, which is superior to placebo and as effective as atenolol and amlodipine.35
Ivabradine is not effective in the treatment or prevention of cardiac arrhythmias and it is likely it loses its efficacy when a tachyarrhythmia occurs (e.g. ventricular or supraventricular tachycardia). It is therefore not recommended in patients with atrial fibrillation or other cardiac arrhythmias that interfere with sinus node function.
At usual recommended doses, heart rate reduction is approximately 10 bpm at rest and during exercise. This leads to a reduction in cardiac workload and myocardial oxygen consumption.
Ivabradine does not influence intracardiac conduction, contractility (no negative inotropic effect) or ventricular repolarisation: in clinical electrophysiology studies, ivabradine had no effect on atrioventricular or intraventricular conduction times or corrected QT intervals.
Pharmacological profile
Ivabradine has a half-life of about two hours, with an effective half-life of 11 hours, which is consistent with twice-daily dosing. It is extensively metabolised by the liver through cytochrome CYP450 3A4. Co-administration of inhibitors of this system, such as ketoconazole which increases levels of ivabradine, are contraindicated.
Plasma levels of ivabradine are increased by grapefruit juice. There is no accumulation of ivabradine at steady state which is achieved within 24 hours. It is rapidly eliminated equally in urine and faeces.
Drug interactions: Most cardiovascular drugs have potential for multiple interactions. It is advisable therefore to consult the Interactions Appendix of the British National Formulary (BNF).
Diltiazem and verapamil may increase plasma levels of ivabradine and should not be co-administered (see above). Similarly, antiarrhythmic and antipsychotic drugs may increase risk of ventricular arrhythmias.
Clinical profile
Ivabradine produces dose-dependent improvements in exercise tolerance and time to development of ischaemia during exercise. Randomised clinical studies in patients with stable angina show that ivabradine effectively reduces heart rate, improves exercise capacity and reduces the number of angina attacks.
It has superior anti-anginal and anti-ischaemic activity to placebo and is non-inferior to atenolol and amlodipine. Ivabradine offers a novel alternative to heart-rate lowering in patients who do not tolerate beta blockade or rate-limiting calcium antagonists. Similarly, since it does not affect blood pressure, it may be an option in patients who experience hypotension.
The landmark, BEAUTIFUL trial36 showed that ivabradine did not improve outcomes in all stable IHD patients with left ventricular dysfunction but could be used to improve outcomes (admission to hospital for fatal and non-fatal MI and coronary revascularisation) in a sub-group of patients with heart rates >70 bpm.
In the more recent SHIFT trial,37 ivabradine significantly reduced hospitalisations for worsening heart failure and heart failure deaths. Ivabradine can now be used in the treatment of chronic heart failure NYHA classes II to IV with systolic dysfunction, in patients in sinus rhythm whose heart rate is ≥75 bpm, in combination with standard therapy including beta blocker, or when beta blockers are contraindicated or poorly tolerated.
Some details of the most recent trial in angina patients, SIGNIFY, are described above.
What dose to use
For treatment of angina the usual recommended starting dose of ivabradine is 5 mg twice daily. After three to four weeks of treatment, the dose may be increased to 7.5 mg twice daily depending on the therapeutic response. If, during treatment, heart rate decreases persistently below 50 bpm at rest or the patient experiences symptoms related to bradycardia, such as dizziness, fatigue or hypotension, the dose must be titrated downward including the possible dose of 2.5 mg twice daily (one half 5 mg tablet twice daily). Treatment must be discontinued if heart rate below 50 bpm or symptoms of bradycardia persist.
Safety, tolerability and side effects
Ivabradine is generally well tolerated but transient and mild visual disturbances, described as ‘luminous phenomena (phosphenes)’ may occur generally during the early phase of dosing. Other side effects include bradycardia, heart block and ventricular extrasystoles. It is important therefore to avoid use of ivabradine in patients with resting heart rates below 60 bpm or in those with heart block or arrhythmias.
Ivabradine is contraindicated in sick sinus syndrome, and cannot be used concominantly with inhibitors of CYP3A4 such as azole antifungals (such as ketoconazole), macrolide antibiotics, nefazodone and the anti-HIV drugs nelfinavir and ritonavir.
Some of the advantages and disadvantages of the use of ivabradine in stable angina:
Pros:
- specific inhibition of heart rate without effects on myocardial contractility
- effective in angina prevention and in improving duration of exercise
- only known side effect to date is reversible visual symptoms.
Cons:
- more expensive than other heart-rate lowering agents
- due to its effect on sinus node activity, should only be used in patients in sinus rhythm
- should be avoided with rate limiting calcium channel blockers.
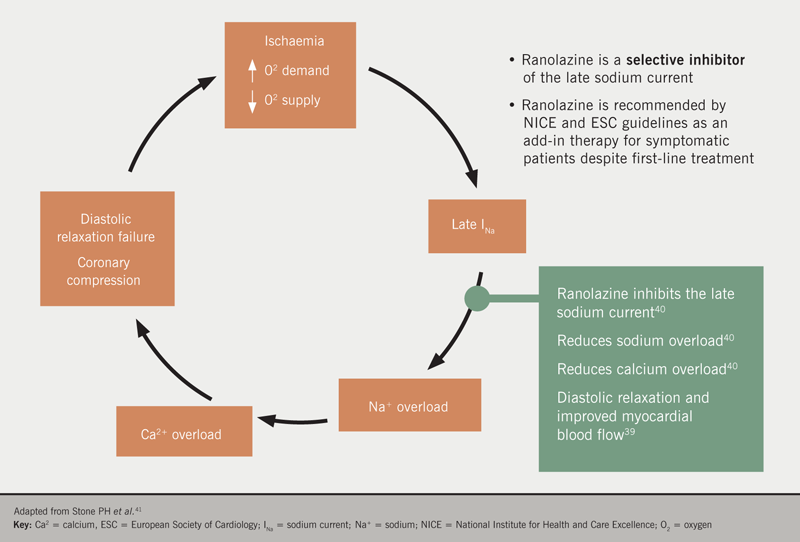
Late sodium current inhibitors
Ranolazine is another more novel anti-anginal drug which inhibits the late sodium current but which does not significantly affect heart rate or blood pressure. It received approval for use in the EU in 2008. The approved indication is as add-on therapy for the symptomatic treatment of patients with stable angina pectoris who are inadequately controlled or intolerant to first-line anti-anginal therapies such as beta blockers and/or calcium antagonists.
Mode of action
Ranolazine, is a metabolically active agent which selectively inhibits the late sodium current (figure 8), which is activated during ischaemia, and which leads to calcium overload and other effects.38–43 Ranolazine reduces these ionic imbalances and the reduction in calcium overload is expected to enhance myocardial relaxation and thereby decrease left ventricular diastolic stiffness, and improve angina symptoms.
Haemodynamic effects are minimal (decreases in mean heart rate <2 bpm and in mean systolic blood pressure <3 mmHg) in patients treated with ranolazine alone or in combination with other anti-anginal agents in controlled studies.
Dose- and plasma concentration-dependent increases in the QTc interval (about 6 msec at 1,000 mg bd), have been observed in patients treated with ranolazine. These effects are thought to relate to inhibition of the fast-rectifying potassium current and are not clinically relevant. There has been no evidence of torsade de pointes (ventricular tachycardia) or treatment discontinuation due to QTc prolongation in clinical trials.
Pharmacological profile
The mean bioavailability after oral administration of ranolazine varies from roughly 30 to 55%, and peak plasma concentrations are reached two to five hours after administration. Steady state is usually achieved within three days of twice-daily dosing. Trough concentrations occur about 12 hours after administration. Approximately 62% of ranolazine is bound to plasma proteins. It is eliminated primarily by metabolism (less than 5% is excreted unchanged). The terminal half-life at steady state after oral administration of ranolazine is about seven hours. Ranolazine undergoes rapid and extensive metabolism. In liver, ranolazine is metabolised principally by CYP3A4, but also by CYP2D6.
Clinical profile
Ranolazine has been subject to conflicting evidence. A systematic review of 17 randomised control trials (including 9,975 patients)44 assessing ranolazine as either monotherapy, or as an adjunct in comparison to placebo or alternative antianginals, demonstrated that only three studies concluded that ranolazine was associated with a reduction in anginal episodes. A further systematic review of seven studies (n=3,317)45 reported that six trials showed a benefit with reduction in anginal episodes versus placebo, along with a reduced consumption of sublingual GTN.
Ranolazine was initially evaluated in three trials in patients with stable angina (MARISA, CARISA and ERICA (see summary data46) where it was shown to significantly improve exercise performance compared to placebo and to decreased angina attacks when added to atenolol, amlodipine or diltiazem.
In one large trial of patients with non-ST elevation acute coronary syndromes (MERLIN-TIMI 36),47 ranolazine did not significantly decrease risk of occurrence of the primary end point (cardiovascular death, MI or recurrent ischaemia) compared to placebo. But it did reduce the risk of recurrent ischaemia, additional anti-anginal medication was needed less often; and there was an improvement in angina frequency. Ranolazine does not have a licence for treatment of non-ST elevation acute coronary syndromes.
Beyond the scope of its use as an antianginal, there may be some benefit using ranolazine as an anti-arrhythmic. In the MERLIN-TIMI 36 study, there was a significantly lower incidence of arrhythmias among patients treated with ranolazine (74%) compared to placebo (83%), including ventricular tachycardia (5% vs. 8%). There has been no evidence of torsade de pointes or treatment discontinuation due to QTc prolongation in clinical trials.
A further adjunctive beneficial side effect of ranolazine is its favourable effect on glucose, which was observed with a reduction in HbA1c in the MERLIN-TIMI 36 trial. Additionally, subjects without diabetes in the ranolazine group were significantly less likely to develop new hyperglycaemia, increases in HbA1c or elevated fasting glucose levels.
The anti-diabetic effects of ranolazine were confirmed in the TERISA (Type 2 Diabetes Evaluation of Ranolazine in Subjects With Chronic Stable Angina) trial.48 The trial involved 949 type 2 diabetes patients who were randomised to a target dose of ranolazine 1,000 mg twice a day or placebo, for eight weeks. These patients had a high burden of angina, with around six to seven episodes per week, and were already receiving treatment with one or two anti-anginals. The primary endpoint – average weekly angina frequency between weeks 2 and 8 – was significantly improved in the ranolazine group (3.8 episodes vs. 4.3 per week; p=0.008). Weekly use of rescue sublingual nitroglycerin was also lower among the ranolazine-treated patients (1.7 vs. 2.1 doses per week; p=0.003).
What dose to use
Ranolazine at a maximal dose of 750 mg twice daily is indicated as add-on therapy for angina patients who are inadequately controlled or intolerant to first-line anti-anginals such as beta blockers or calcium antagonists.
The recommended initial dose in adults is 375 mg twice daily. After 2–4 weeks, the dose should be titrated to 500 mg twice daily. According to the patient’s response, it can be further titrated to the recommended maximum dose of 750 mg twice daily.
If the patient experiences treatment-related adverse effects, such as dizziness, nausea or vomiting, down-titration to 500 mg or 375 mg twice daily may be carried out. If symptoms do not resolve, then treatment should be discontinued.
Safety, tolerability and side effects
Ranolazine appears generally well tolerated. It has the potential to prolong the QT interval, but no increase in significant arrhythmias has been observed and the compound appears to provide a safe addition (being mindful to avoid certain co-administrations, e.g. CYP 3A4 such as ketoconazole) to traditional antianginal agents.
It is contraindicated in persons with pre-existing QT-prolongation. The most commonly reported adverse effects are dizziness, headache, nausea, vomiting and constipation. Increasing the dose of ranolazine from 1,000 mg twice daily to 1,500 mg twice daily (unlicensed) can be in a disproportionate increase in the most frequent adverse effects, including asthenia, nausea, vomiting, dizziness, headache and constipation.
Here are some of the advantages and disadvantages of the use of ranolazine in stable angina.
Pros:
- well tolerated and effective anti-anginal
- metabolically neutral and potentially beneficial in diabetics with stable angina
- potential anti-arrhythmic properties.
Cons:
- caution required with pre-existing QT prolongation
- more expensive than conventional agents
- conflicting evidence base.
Angina treatment guidelines
NICE, the ESC and SIGN have published comprehensive guidance which incorporates findings from significant clinical trials of medical management of angina.1–3 They also all emphasise that it is important for patients to receive optimal medical treatment (OMT) being defined as the use of up to two anti-anginal drugs plus secondary prevention measures (figure 9).
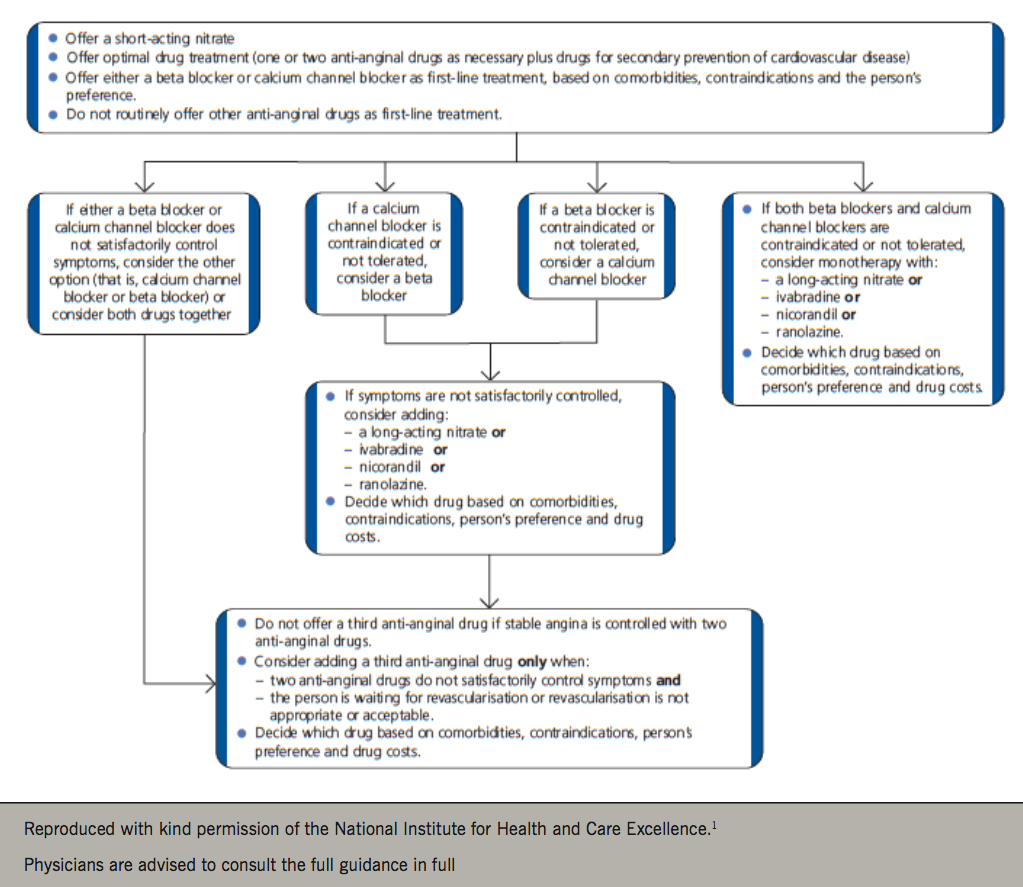
Consideration to adding a third anti-anginal drug should only be given when:
- the person’s symptoms are not controlled with two anti-anginal drugs and
- the person is either waiting for revascularisation or revascularisation is not considered appropriate or acceptable.
First-line angina/ischaemic relief
To treat ongoing anginal episodes, short-acting nitrates – taken either sublingually or in tablet form – are recommended by ESC, NICE and SIGN. Symptoms of angina can effectively be relieved by the use of short-acting nitrates. The individual should be carefully instructed in their use and also of the potential benefit of using nitrates before any planned exercise or activity. Short-acting nitrates should be prescribed for all those with stable angina.
The NICE1 and ESC2 guidelines recommend, as first-line treatment, either beta blockers or calcium antagonists to prevent anginal episodes. The decision to choose, either a beta blocker or a calcium antagonist, should be based on the individual’s co-morbidities, contraindications and preference. If they are unable to tolerate either of these therapies, then consideration should be given to switching to the other. The recommendation is not to routinely offer antianginal drugs other than beta blocker or calcium channel blocker as first-line treatment in stable angina.
If symptoms are not satisfactorily controlled on either option alone, then the recommendation is to consider using a combination of a beta blocker and a calcium antagonist, although the latter should be a dihydropyridine-type (amlodipine, slow-release nifedipine or felodipine). The ESC guidelines2 advocate the use of combination beta blocker and calcium channel blocker as a potential first-line therapy should blood pressure and heart rate allow.
Second-line angina/ischaemic relief
For those individuals in whom symptoms persist, despite optimisation of their beta blocker or calcium channel blocker dose, consideration should be given to introducing additional antianginal therapy. These may also be considered as initial monotherapy when the patient is intolerant of either a beta blocker or calcium channel blocker, or if blood pressure and heart rate limit the use of first-line agents. These therapeutic options include:
- long-acting nitrates
- ivabradine
- nicorandil
- ranolazine.
The decision on which drug to use should be based on co-morbidities, contraindications and patient preference.44
When counselling patients with regards to the relative merits of medical treatment, it is helpful to advise them that the aim of anti-anginal treatment is to prevent episodes of angina, and the aim of secondary prevention is to prevent cardiovascular events, including MI and stroke.
All anti-anginal therapies should be titrated, depending on the patient’s symptoms, to their optimal tolerated doses (within the licensed dose range) before considering adding additional therapy.49
Combination therapy
By taking a careful history it may be possible to estimate the pathophysiological basis of angina in individual patients: coronary spasm, fixed obstructive coronary disease and obstructive coronary disease with a dynamic component. On this basis, an appropriate drug therapy can be selected.
In patients with minimal symptoms, intermittent sublingual GTN may suffice. In patients with moderate symptoms it is worth starting therapy with either a cardioselective beta blocker, calcium antagonist or long-acting nitrate. No one drug has shown consistently greater efficacy in relieving chest pain or reducing exercise-induced ischaemia.
Across a broad range of commonly assessed outcomes, there is little difference in the magnitude of clinical efficacy of different anti-anginal classes, used in an add-on role.50
If intermittent long-acting nitrates are used, it is probably wise to do so in association with a beta blocker or calcium antagonist to provide 24-hour anti-anginal and anti-ischaemic ‘cover’. In general, optimal dose regimens of a given class of drug are preferable to routine use of combination therapy. Many patients respond to monotherapy when drugs are used at the appropriate doses; when monotherapy fails it is often advisable to add-in another class of anti-anginal medication.
In most patients with severe symptoms combined therapy is more effective than the use of a single drug and usually the side effects are no greater and may even be reduced. However, triple therapy, although widely used, is of doubtful benefit to the patient.51
The addition of further antianginal medications should be guided by symptoms and by haemodynamic status. Heart rate, blood pressure and the presence of left ventricular dysfunction may limit or guide the addition of further therapies. Other possible combinations are shown in figure 3.13
Other anti-anginals
Trimetazidine, another metabolically acting drug52 is available in many European countries but not the UK. It preserves energy balance during myocardial ischaemia and stimulates glucose oxidation and acts as a partial fatty acid oxidation inhibitor.
Also, there is current interest in the use of the uric acid lowering agent, allopurinol, for the treatment of angina. Allopurinol, a xanthine oxidase inhibitor, at high doses appears to markedly prolong the time to chest pain and to ST-segment depression during exercise in patients with stable angina.53 The compound continues to be investigational in treatment of angina.
Failure of medical therapy
When drug therapy fails to control the symptoms of angina, a number of alternative therapies are available including acupuncture, transcutaneous electrical nerve stimulation (TENS) and enhanced external counterpulsation (EECP).54 NICE states that these cannot be recommended in the treatment of refractory angina. Similarly, there is an increasing consensus that unproven therapies, such as chelation therapy should be discouraged.
Revascularisation and anatomical assessment
All guidelines suggest that diagnostic invasive coronary angiography may be offered to guide the treatment strategy for patients whose symptoms are not controlled with optimal medical treatment. This can include percutaneous coronary intervention (PCI) or planned coronary artery bypass grafting (CABG).
The SIGN3 and ESC2 guidelines suggest that computerised tomography-coronary angiography should be considered for the investigation of patients with chest pain in whom the diagnosis of stable angina is suspected but not clear from the history alone. In the presence of significant coronary artery disease, tailored medical therapy or invasive coronary angiography may then be offered in order to proceed to PCI.
The controversial and debate-inspiring ORBITA study by Al-Lamee et al.55 highlighted the importance of optimal medical therapy. In single vessel CAD, which has been determined to be physiologically significant by fractional flow reserve/instantaneous wave free ratio, PCI does not improve anginal symptoms beyond the improvement derived from optimum antianginal medical therapy. More complex multi-vessel coronary artery disease was not addressed in this study.
A detailed review comparing revascularisation or optimal medical treatment in stable angina patients can be found in module 7 in the recently reported ISCHEMIA (International Study of Comparative Health Effectiveness With Medical and Invasive Approaches) study.56
Key learning messages
- Antianginal treatment should be tailored to the individual patient
- It is important to improve prognosis as well as minimise or abolish symptoms
- No one antianginal has shown consistently greater efficacy in relieving chest pain or reducing exercise-induced ischaemia
- The dosing of one drug should be optimised before adding another
- Combinations of drugs may have additive antianginal effects
- Invasive and non invasive anatomical assessment plays a role in both interventional treatment and risk stratification
close window and return to take test
References
- National Institute for Health and Care Excellence (NICE). Stable angina: management. Clinical guideline [CG126]. London: NICE, 2011 (updated 2016). www.nice.org.uk/guidance/CG126
- Knuuti J, Wijns W, Saraste A et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes: The Task Force for the diagnosis and management of chronic coronary syndromes of the European Society of Cardiology. Eur Heart J 2019 (published online 31st August 2019). https://doi.org/10.1093/eurheartj/ehz425
- Scottish Intercollegiate Guidelines Network (SIGN). Guideline 151. Management of stable angina. A national clinical guideline. Edinburgh: SIGN, April 2018. https://www.sign.ac.uk/sign-151-stable-angina.html
- Sorbets E, Fox KM, Elbez Y et al. Long-term outcomes of chronic coronary syndrome: worldwide insights from the international CLARIFY registry. Eur Heart J 2019 (published online 3rd September 2019). http://dx.doi.org/10.1093/eurheartj/ehz660
- Antithrombotic Trialists (ATT) Collaboration. Aspirin in primary and secondary prevention of vascular disease: collaborative meta-analysis of individual participant data from randomised trials. Lancet 2009;373:1849–60. http://dx.doi.org/10.1016/S0140-6736(09)60503-1
- Seshasai SRK, Wijesuriya S, Sivakumaran R et al. Effect of aspirin on vascular and nonvascular outcomes. Arch Intern Med 2012 (published online January 9, 2012). http://dx.doi.org/10.1001/archinternmed.2011.628
- Garcia LA, Cea-Soriano L, Martin-Merino E, Johansson S. Discontinuation of low dose aspirin and risk of myocardial infarction: a case-control study in UK primary care. BMJ 2011;343:d4094. http://dx.doi.org/10.1136/bmj.d4094
- National Institute for Health and Care Excellence. NICE clinical guideline [CG181] Cardiovascular disease: risk assessment and reduction, including lipid modification. London: NICE, 2014 (updated 2016). https://www.nice.org.uk/guidance/cg181
- Cooper A, Nherera L, Calvert N et al. Clinical guidelines and evidence review for lipid modification: cardiovascular risk assessment and the primary and secondary prevention of cardiovascular disease. London: National Collaborating Centre for Primary Care and Royal College of General Practitioners, 2008 (revised March 2010).
- Cholesterol Treatment Trialists Collaboration (CCTC). Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet 2010;376:1670–81. http://dx.doi.org/10.1016/S0140-6736(10)61350-5
- Mach F, Baigent C, Catapano AL et al. 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk: The Task Force for the management of dyslipidaemias of the European Society of Cardiology (ESC) and European Atherosclerosis Society (EAS). Eur Heart J 2019 (published online 31st August 2019) http://dx.doi.org/10.1093/eurheartj/ehz455
- Chapman MJ, Ginsberg HN, Amarenco P et al. Triglyceride-rich lipoproteins and high-density lipoprotein cholesterol in patients at high risk of cardiovascular disease: evidence and guidance for management. Eur Heart J 2011;32:1345–61. http://dx.doi.org/10.1093/eurheartj/ehr112
- Ferrari R, Camici PG, Crea F et al. A ‘diamond’ approach to personalized treatment of angina. Nature Rev Cardiol 2018;15:120–32. https://doi.org/10.1038/nrcardio.2017.131
- Thadani U, Opie LH (eds). Nitrates updated. Current use in angina, ischemia, infarction and failure. Boston: Kluwer Academic Publishers. 1997. ISBN 0-7923-3466-3
- Crea F, Camici PG, De Caterina R, Lanza GA. Chronic ischaemic heart disease, Chapter 17, pages 597–664. In: Camm AJ, Luscher TF, Serruys PW (eds). ESC textbook of cardiovascular medicine. 2nd Edition. Oxford: Oxford University Press, 2009.
- Purcell H, Mulcahy D. Circadian rhythms and the onset of myocardial infarction: clinical implications. J Cardiovasc Risk 1995;2:510–14. http://dx.doi.org/10.1097/00043798-199512000-00005
- Thadani U. Medical therapy of stable angina pectoris, pages 73–87. In: Abrams J (ed). Cardiology clinics. Angina pectoris. London: WB Saunders, 1991.
- Mansoor AH, Kaul U. Beta-blockers in clinical medicine. JAPI 2009;57:7–12. http://www.japi.org/special_issue_2009/article_02.pdf
- Borer JS. Heart rate: from risk marker to risk factor. Eur Heart J 2008;10(suppl F):F2–F6. http://dx.doi.org/10.1093/eurheartj/sun019
- Cruikshank JM, Pritchard BN. Beta blockers in clinical practice, 2nd ed. Edinburgh: Churchill Livingstone, 1994.
- Bangalore S, Messerli FH, Kostis JB, Pepine CJ. Cardiovascular protection using beta-blockers. J Am Coll Cardiol 2007;50:563–72. http://dx.doi.org/10.1016/j.jacc.2007.04.060
- Tilmant PY, Lablanche JM, Thieuleux FA, Dupuis BA, Bertrand ME. Detrimental effect of propranolol in patients with coronary arterial spasm countered by combination with diltiazem. Am J Cardiol 1983;52(3):230–3. http://dx.doi.org/10.1016/0002-9149(83)90113-3
- Armstrong R, De Maria GL, Scarsini R et al. Assessing and managing coronary microcirculation dysfunction in acute ST-segment elevation myocardial infarction. Expert Rev Cardiovasc Ther 2019;17:111–26. https://dx.doi.org/10.1080/14779072.2019.1561279
- Purcell H, Waller DG, Fox K. Calcium antagonists in cardiovascular disease. Br J Clin Pract 1989;43:369–79.
- Opie LH. Clinical use of calcium channel antagonist drugs. Boston: Kluwer Academic Publishers, 1990. ISBN 0-7923-0872-7.
- Psaty BM, Heckbert SR, Koepsell TD et al. The risk of myocardial infarction associated with antihypertensive drug therapies. JAMA 1995;274:620–5. http://dx.doi.org/10.1001/jama.1995.03530080036038
- Stason WB1, Schmid CH, Niedzwiecki D et al. Safety of nifedipine in angina pectoris: a meta-analysis. Hypertension 1999;33:24-31. https://doi.org/10.1161/01.HYP.33.1.24
- British Heart Foundation. Drug cabinet: calcium channel blockers. Heart Matters https://www.bhf.org.uk/informationsupport/heart-matters-magazine/medical/drug-cabinet/calcium-channel-blockers (last accessed 24th September 2019).
- Packer M, O’Connor CM, Ghali JK et al. Effect of amlodipine on morbidity and mortality in severe chronic heart failure. N Engl J Med 1996;335:1107–14. http://dx.doi.org/10.1056/NEJM199610103351504
- Purcell H, Patel DJ, Mulcahy D, Fox K. Nicorandil, chapter 178, pages 1638–45. In: Messerli FH (ed). Cardiovascular drug therapy, 2nd edition. Philadelphia: WB Saunders, 1996.
- The IONA Study Group. Effect of nicorandil on coronary events in patients with stable angina: the Impact Of Nicorandil in Angina (IONA) randomised trial. Lancet 2002;359:1269–75. http://dx.doi.org/10.1016/S0140-6736(02)08265-X
- Medicines and Healthcare products Regulatory Agency (MHRA). Drug safety update. Nicorandil (Ikorel): now second-line treatment for angina – risk of ulcer complications. MHRA, January 2016. www.gov.uk/drug-safety-update/nicorandil-ikorel-now-second-line-treatment-for-angina-risk-of-ulcer-complications (last accessed 24th September 2019)
- European Medicines Agency (EMA). EMA recommends measures to reduce risk of heart problems with Corlentor/Procoralan (ivabradine). EMA, November 2014. http://www.ema.europa.eu/ema/index.jsp?curl=pages/news_and_events/news/2014/11/news_detail_002217.jsp&mid=WC0b01ac058004d5c1 (last accessed 24th September 2019)
- Fox K, Ford I, Steg PG, Tardif J, Tendera M, Ferrari R. Ivabradine in stable coronary artery disease without clinical heart failure. N Engl J Med 2014;371:1091–9. http://dx.doi.org/10.1056/NEJMoa1406430
- Sulfi S, Timmis AD. Ivabradine – the first selective sinus node If channel inhibitor in the treatment of stable angina. Int J Clin Prac 2006;60:222–8. http://dx.doi.org/10.1111/j.1742-1241.2006.00817.x
- Fox K, Ford I, Steg PG, Tendera M, Ferrari R, on behalf of the BEAUTIFUL Investigators. Ivabradine for patients with stable coronary artery disease and left-ventricular systolic dysfunction (BEAUTIFUL): a randomised, double-blind, placebo-controlled trial. Lancet 2008;37:807–16. http://dx.doi.org/10.1016/S0140-6736(08)61170-8
- Swedberg K, Komajda M, Bohm M et al. Ivabradine and outcomes in chronic heart failure (SHIFT): a randomised placebo-controlled trial. Lancet 2010;376:875–85. http://dx.doi.org/10.1016/S0140-6736(10)61198-1
- Khan K, Jones M. Ranolazine in the management of chronic stable angina. Br J Cardiol 2011;18:179. https://bjcardio.co.uk/2011/08/ranolazine-in-the-management-of-chronic-stable-angina/
- Chaitman BR, Pepine CJ, Parker JO, et al. Effects of ranolazine with atenolol, amlodipine, or diltiazem on exercise tolerance and angina frequency in patients with severe chronic angina: a randomized controlled trial. JAMA 2004;291:309-16. https://dx.doi.org/10.1001/jama.291.3.309
- Belardinellia L, Antzelevitch C, Fraserc H. Inhibition of late (sustained/persistent) sodium current: a potential drug target to reduce intracellular sodium-dependent calcium overload and its detrimental effects on cardiomyocyte function. Eur Heart J Suppl 2004;6(Suppl 1):i3-i7. https://dx.doi.org/10.1093/eurheartj/6.suppl_i.i3
- Stone PH. Ranolazine: new paradigm for management of myocardial ischemia, myocardial dysfunction, and arrhythmias. Cardiol Clin 2008;26:603-14. https://dx.doi.org/10.1016/j.ccl.2008.06.02
- Chaitman BR. Ranolazine for the treatment of chronic angina and potential use in other cardiovascular conditions. Circulation 2006;113:2462-72. https://dx.doi.org/10.1161/CIRCULATIONAHA.105.597500
- Hasenfuss G, Maier LS. Mechanism of action of the new anti-ischemia drug ranolazine. Clin Res Cardiol 2008;97:222-6. https://dx.doi.org/10.1007/s00392-007-0612-y
- Salazar CA, Basilio Flores JE, Veramendi Espinoza LE et al. Ranolazine for stable angina pectoris. Cochrane Database Syst Rev 2017; Issue 2: Article no. CD011747. https://dx.doi.org/10.1002/14651858.CD011747.pub2 (last accessed 24th September 2019).
- Banon D, Filion KB, Budlovsky T et al. The usefulness of ranolazine for the treatment of refractory chronic stable angina pectoris as determined from a systematic review of randomized controlled trials. Am J Cardiol 2014;113:1075–82. https://dx.doi.org/10.1016/j.amjcard.2013.11.070
- Nash DT, Nash SD. Ranolazine for chronic stable angina. Lancet 2008;372:1335–41. http://dx.doi.org/10.1016/S0140-6736(08)61554-8
- Morrow DA, Scirica BM, Karwatowska-Prokopczuk E et al. for the MERLIN-TIMI 36 Trial Investigators. Effects of Ranolazine on Recurrent Cardiovascular Events in Patients with Non-ST-Elevation Acute Coronary Syndromes. The MERLIN-TIMI 36 Randomized Trial. JAMA 2007;297:1775–83. http://dx.doi.org/10.1001/jama.297.16.1775
- Kosiborod M, Arnold SV, Spertus JA, et al. Evaluation of ranolazine in patients with type 2 diabetes mellitus and chronic stable angina: results from the TERISA randomized clinical trial (type 2 diabetes evaluation of ranolazine in subjects with chronic stable angina). J Am Coll Cardiol 2013;61:2038–45. http://dx.doi.org/10.1016/j.jacc.2013.02.011
- Purcell H, De Silva R. Treating stable angina – is there a NICE way towards an international consensus? Int J Clin Pract 2012;66:614–8. http://dx.doi.org/10.1111/j.1742-1241.2012.02941.x
- Belsey J, Savelieva I, Mugelli A, Camm AJ. Relative efficacy of antianginal drugs used as add-on therapy in patients with stable angina: A systematic review and meta-analysis. Eur J Prevent Cardiol 2014. http://dx.doi.org/10.1177/2047487314533217
- Akhras F, Jackson G. Efficacy of nifedipine and isosorbide mononitrate in combination with atenolol in stable angina. Lancet 1991;338:1036–9. http://dx.doi.org/10.1016/0140-6736(91)91900-F
- Onay-Besikci A, Ozkan SA. Trimetazidine revisited: A comprehensive review of the pharmacological effects and analytical techniques for the determination of trimetazidine. Cardiovasac Ther 2008;26:147–65. http://dx.doi.org/10.1111/j.1527-3466.2008.00043.x
- Narasimharajapura SR, Ireland S, Georg J, Belch JJF, Lang CC, Struthers AD. Mechanistic insights into the therapeutic use of high-dose allopurinol in angina pectoris. J Am Coll Cardiol 2011;58:820–8. http://dx.doi.org/10.1016/j.jacc.2010.12.052
- Mulcahy D, Knight K, Stables R. Fox K. Lasers, burns, cuts, tingles and pumps: a consideration of alternative treatments for intractable angina. Br Heart J 1994;71:406–7. http://dx.doi.org/10.1136/hrt.71.5.406
- Al-Lamee R, Thompson D, Dehbi HM et al. Percutaneous coronary intervention in stable angina (ORBITA): a double-blind, randomised controlled trial. Lancet 2018;391:31–40. https://dx.doi.org/10.1016/S0140-6736(17)32714-9
- Hochman JS. International Study of Comparative Health Effectiveness With Medical and Invasive Approaches: primary report of clinical outcomes. Presented at: AHA 2019. November 16, 2019. Philadelphia, PA. https://www.tctmd.com/news/ischemia-invasive-strategy-no-better-meds-cv-events (last accessed 21st November 2019).


All rights reserved. No part of this programme may be reproduced, stored in a retrieval system, or transmitted in any form or by any means, electronic, mechanical, photocopying, recording or otherwise, without the prior permission of the publishers, Medinews (Cardiology) Limited.
It shall not, by way of trade or otherwise, be lent, re-sold, hired or otherwise circulated without the publisher’s prior consent.
Medical knowledge is constantly changing. As new information becomes available, changes in treatment, procedures, equipment and the use of drugs becomes necessary. The editors/authors/contributors and the publishers have taken care to ensure that the information given in this text is accurate and up to date. Readers are strongly advised to confirm that the information, especially with regard to drug usage, complies with the latest legislation and standards of practice.
Healthcare professionals should consult up-to-date Prescribing Information and the full Summary of Product Characteristics available from the manufacturers before prescribing any product. Medinews (Cardiology) Limited cannot accept responsibility for any errors in prescribing which may occur.