Pulmonary hypertension (PH) is common in patients with severe aortic stenosis (AS). Severe PH has been associated with mortality up to two years following transcatheter aortic valve implantation (TAVI). Data on longer-term outcomes in TAVI patients with severe PH are limited. We aimed to compare all-cause mortality at five years post-TAVI in patients with and without severe PH and to identify any patient factors associated with reduced long-term survival.
TAVI patients meeting our inclusion criteria between January 2013 and October 2017 at a specialist cardiac centre in the UK were retrospectively analysed. Severe PH was defined as a systolic pulmonary arterial pressure >40 mmHg, estimated on transthoracic echocardiography. Data on patient demographics, comorbidities and mortality were obtained from routinely collected registry data. Kaplan-Meier and Cox-proportional hazards analyses were performed.
The median ages of the patients were 84 and 83 years in the group without severe PH and the group with severe PH, respectively. Severe PH was present in 95 out of 219 patients (43%). Patients with severe PH had higher levels of disability, left ventricular impairment and serum creatinine. On multi-variate analysis, the presence of severe PH was not associated with an increased mortality (adjusted hazard ratio [HR]=1.23, p=0.29). Peripheral vascular disease (PVD) was associated with a significantly increased risk of death at five years’ follow-up (adjusted HR=2.24, p=0.002). A lower body mass index (BMI) was also independently associated with reduced survival (adjusted HR=0.96, for an increase of 1 kg/m2, p=0.038).
In conclusion, our data show that severe PH in patients with AS did not affect five-year survival post-TAVI. Reduced BMI and PVD were significantly associated with long-term all-cause mortality in patients undergoing TAVI.
Introduction
Degenerative aortic stenosis (AS) is the most common acquired valvulopathy worldwide, affecting 2% to 3% of individuals aged ≥65 years, with its prevalence expected to increase due to an ageing population.1 Patient morbidity and mortality increase sharply with worsening clinical signs and symptoms, requiring prompt therapeutic management.2 Transcatheter aortic valve implantation (TAVI) represents an alternative minimally-invasive therapeutic option to surgical valve replacement in patients who are intermediate-to-high risk.3,4 Severe pulmonary hypertension (PH) is commonly found in patients with symptomatic severe AS undergoing TAVI. In this patient group, it has a reported prevalence of between 28% and 56%, with observed variation between studies attributed to differences in the techniques used to measure haemodynamic pulmonary artery parameters and the threshold by which severe PH is defined.5
In patients with severe AS, PH is frequently attributed to left heart disease (or postcapillary PH), often secondary to left ventricular systolic or diastolic dysfunction.6 To date, most studies have shown that in the short-to-intermediate term, the presence of severe PH is associated with increased morbidity and mortality up to two years following TAVI.7 Data on the long-term follow-up of TAVI patients with severe PH, however, are less well characterised. This is of importance, as in patients with AS and postcapillary PH, a reduction in systolic pulmonary artery pressure (sPAP) and, in some cases, subsequent reversal of PH, following TAVI has previously been observed and found to be associated with improved outcomes.8 The long-term effects of preprocedural severe PH are, therefore, important to understand in guiding further insights into whether its presence at baseline has any extended effects on clinical outcomes, and whether successful TAVI intervention alone is sufficient in this patient group.
The primary aim of this study was, therefore, to assess the impact of severe PH on all-cause mortality at five-year follow-up in patients who were undergoing TAVI for severe AS, accounting for the influence of other potential covariates on patient survival.
Method
Study design
This was a retrospective cohort study of TAVI procedures carried out at a single tertiary cardiac centre in the UK. Data were obtained through a registry of routinely collected hospital data, audited in all patients undergoing TAVI. Patients who underwent TAVI from the start of the registry (in January 2013) up to October 2017 were included and followed-up for five years. Data on all-cause mortality were obtained through the Office of National Statistics linked records. Severe PH was defined as the presence of an estimated sPAP on transthoracic echocardiography of greater than 40 mmHg. Patients without any recorded sPAP measurements, or with aortic valve pathology other than AS, were excluded. In patients who had multiple TAVI procedures within the prespecified time-period, only the index procedure was considered.
Patient demographic data, including age, sex, body mass index (BMI), ethnicity, New York Heart Association (NYHA) scores and the presence of any comorbidities of interest (derived from previous medical records), were anonymised prior to analysis. Investigations, such as serum creatinine, echocardiogram and any prior invasive coronary angiographic findings, were also recorded. Activities of daily living (ADL) were assessed by a healthcare professional on admission, prior to patients’ TAVI procedure, and a corresponding value on the Katz index of ADL was subsequently assigned. TAVI access route was also noted.
Statistical analysis
Statistical analysis was performed on Stata V.18 (Statcorp LLC). Patients were assigned to either of two groups according to the presence of severe PH. Baseline data were compared between the two groups. Chi-squared or Fisher’s exact test (if n≤4) were used for categorical variables. Continuous variables were assessed for normality using the Shapiro-Wilk test. Non-parametric and ordinal data were compared statistically using the Mann-Whitney U test.
A Kaplan-Meier survival plot was formulated using all-cause mortality data with a median survival reported in patients with and without severe PH. Hazard ratios (HR) were calculated using the Cox-proportional hazards (CPH) model. Both univariate and multi-variate analyses (adjusting for any covariates with univariate p<0.1) were performed. Missing data were reported and addressed through the exclusion of patients from any corresponding CPH analysis.
Results
A total of 219 patients meeting our inclusion criteria underwent TAVI. Severe PH was present in 95 patients (43%) compared with 124 patients (57%) without.
Baseline characteristics
Patients’ demographics and comorbidities are summarised in table 1. All continuous data in this analysis were non-parametric and are expressed as median (interquartile range [IQR]). Ordinal data (namely Katz index of ADL) are expressed as median (range). Categorical data are expressed as n with a corresponding percentage. The median age was 84 and 83 years for the groups without and with severe PH, respectively. Patients with severe PH had a significantly higher baseline creatinine (median creatinine in the severe PH group was 106 µmol/L compared with 94 µmol/L in patients without severe PH, p=0.014) and an increased range of patients with a greater degree of dependence in their ADL (Katz ADL range of 1–6 in severe PH group compared with 3–6 in patients without, p=0.017). A significantly greater incidence of left ventricular (LV) impairment (ejection fraction [EF] <50%) was also found in the severe PH group (45.3% compared with 30.3% in patients without severe PH, p=0.024). The two patient groups were otherwise well-matched.
Table 1. Summary of patient demographic data and clinical baseline characteristics
| Patients without severe PH (N=124) |
Patients with severe PH (N=95) |
p value | |
| Median age (IQR), years | 84 (80–87) | 83 (78–87) | 0.27 |
| Male, n (%) | 63 (48) | 53 (56) | 0.23 |
| Ethnicity, n (%) Caucasian Asian |
124 (96) 5 (4) |
92 (97) 3 (3) |
0.73 |
| Median BMI (IQR), kg/m2 | 26.1 (23.7–29.4) | 26.5 (23.5–31.1) | 0.54 |
| History of diabetes, n (%) | 27 (22) | 28 (29) | 0.19 |
| Comorbid pulmonary disease, n (%) | 48 (39) | 40 (42) | 0.61 |
| History of neurological disease, n (%) | 27 (22) | 15 (16) | 0.27 |
| History of PVD, n (%) Missing=1 |
19 (15) | 8 (8) | 0.12 |
| Current or previous history of smoking, n (%) Missing=2 |
65 (53) | 57 (61) | 0.25 |
| Previous MI, n (%) | 25 (20) | 24 (25) | 0.37 |
| Median creatinine (IQR), µmol/L | 94 (76–109) | 106 (77–138) | 0.014 |
| NYHA 3 or more, n (%) | 98 (79) | 79 (83) | 0.44 |
| Median Katz index score (range) | 6 (3–6) | 6 (1–6) | 0.017 |
| Median AV peak gradient (IQR), mmHg Missing=5 |
65 (48–83) | 64 (49–85) | 0.97 |
| Median AV mean gradient (IQR), mmHg Missing=9 |
35 (26–46) | 37 (25–50) | 0.70 |
| Median AV area (IQR), cm2 Missing=2 |
0.7 (0.5–0.8) | 0.6 (0.5–0.73) | 0.17 |
| Median aortic annular diameter (IQR), mm Missing=7 |
24 (22–25) | 23 (22–25) | 0.37 |
| LV impairment (EF<50%), n (%) Missing=2 |
37 (30) | 43 (45) | 0.024 |
| Any coronary artery disease, n (%) Missing=2 |
63 (52) | 51 (54) | 0.77 |
| Left main stem disease present, n (%) Missing=2 |
4 (3) | 3 (3) | 1.00 (Fisher’s exact test) |
| Percutaneous transfemoral approach, n (%) | 104 (84) | 83 (87) | 0.47 |
| Key: AV = aortic valve; BMI = body mass index; EF = ejection fraction; IQR = interquartile range; LV = left ventricular; MI = myocardial infarction; NYHA = New York Heart Association; PH = pulmonary hypertension; PVD = peripheral vascular disease | |||
Survival analysis
Figure 1 shows the Kaplan-Meier plot for all-cause mortality post-TAVI in patients grouped according to the presence of severe PH over five years. All-cause mortality was observed in 59% of patients with severe PH, with a median survival of 4.12 years. Patients without severe PH had a mortality rate of 54% with a median survival of 4.51 years.
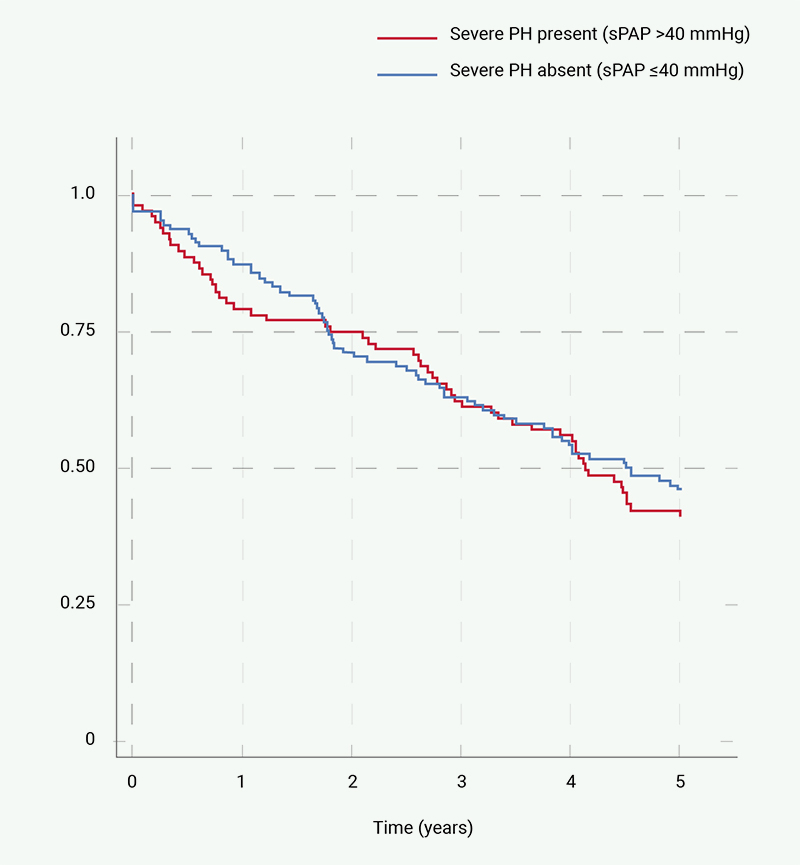
| Key: PH = pulmonary hypertension; sPAP = systolic pulmonary artery pressure |
Table 2 summarises the univariate CPH analyses conducted in this study. This included the presence of severe PH, as well as all other demographic, clinical and echocardiographic data. Patients with severe PH had a 12% increased risk of death over five years, however, this was not statistically significant (p=0.53). BMI, diabetes, peripheral vascular disease (PVD), Katz index, aortic valve (AV) peak gradient, AV mean gradient, aortic annular diameter and alternative TAVI approaches (other than a percutaneous transfemoral approach) were included in a multi-variate analysis, as univariate CPH indicated possible association with p<0.1.
Table 2. Summary of univariate survival analyses conducted using Cox proportional hazards test
| Hazard ratio | p value | 95%CI | |
| Severe pulmonary hypertension | 1.12 | 0.53 | 0.78 to 1.60 |
| Asian ethnicity compared with Caucasians | 0.33 | 0.12 | 0.08 to 1.34 |
| BMI (increase of unit of 1 kg/m2) | 0.97 | 0.079 | 0.93 to 1.0038 |
| Diabetes | 1.39 | 0.096 | 0.94 to 2.05 |
| History of pulmonary disease | 1.25 | 0.21 | 0.88 to 1.79 |
| History of neurological disease | 0.893 | 0.63 | 0.56 to 1.42 |
| History of PVD | 2.71 | <0.0001 | 1.7 to 4.31 |
| Current or previous history of smoking | 1.21 | 0.30 | 0.84 to 1.74 |
| Previous MI | 1.11 | 0.63 | 0.73 to 1.69 |
| Creatinine (increase of unit of 1) | 1.0004 | 0.75 | 0.998 to 1.003 |
| NYHA of 3 or more | 0.92 | 0.73 | 0.59 to 1.44 |
| Katz index (increase of unit of 1) | 0.87 | 0.099 | 0.74 to 1.03 |
| AV peak gradient (increase of unit of 1) | 0.99 | 0.092 | 0.99 to 1.001 |
| AV mean gradient (increase of unit of 1) | 0.99 | 0.087 | 0.98 to 1.001 |
| AV area (increase of unit of 1) | 1.004 | 0.99 | 0.37 to 2.72 |
| Aortic annular diameter (increase of unit of 1) | 1.11 | 0.006 | 1.03 to 1.20 |
| LV impairment (EF<50%) | 0.85 | 0.40 | 0.59 to 1.24 |
| Any coronary artery disease | 0.97 | 0.85 | 0.68 to 1.38 |
| Left main stem disease | 1.0015 | 1.00 | 0.37 to 2.71 |
| Alternative TAVI approach to percutaneous transfemoral | 2.32 | <0.0001 | 1.48 to 3.62 |
| Key: AV = aortic valve; BMI = body mass index; CI = confidence interval; EF = ejection fraction; LV = left ventricular; MI = myocardial infarction; NYHA = New York Heart Association; PVD = peripheral vascular disease; TAVI = transcatheter aortic valve implantation | |||
Table 3 summarises this multi-variate analysis, adjusting for covariates of interest. Here, severe PH was associated with a 23% increased adjusted risk of death and, again, this was not statistically significant (p=0.29). An increase of 1 kg/m2 in BMI was associated with a 4% reduced adjusted risk of death over five years (p=0.038). The presence of PVD was strongly associated with a 124% increased adjusted risk of death (p=0.002). When adjusted for other covariates, diabetes, TAVI access route, Katz ADL and aortic valve/annular parameters did not reach the threshold for statistical significance in our multi-variate survival analysis.
Table 3. Summary of the multi-variate survival analysis conducting using Cox proportional hazards test (adjusted for variables with a p value <0.1)
| Variable of interest | Adjusted hazard ratio | p value | 95%CI |
| Severe pulmonary hypertension | 1.23 | 0.29 | 0.836 to 1.81 |
| BMI (increase of unit of 1 kg/m2) | 0.96 | 0.038 | 0.919 to 0.998 |
| Diabetes | 1.48 | 0.059 | 0.985 to 2.24 |
| History of PVD | 2.24 | 0.002 | 1.34 to 3.76 |
| Katz index (increase of unit of 1) | 0.86 | 0.14 | 0.701 to 1.05 |
| AV peak gradient (increase of unit of 1) | 0.998 | 0.86 | 0.976 to 1.02 |
| AV mean gradient (increase of unit of 1) | 0.996 | 0.84 | 0.961 to 1.03 |
| Aortic annular diameter (increase of unit of 1) | 1.09 | 0.057 | 0.998 to 1.18 |
| Alternative TAVI approach to percutaneous transfemoral | 1.70 | 0.054 | 0.991 to 2.91 |
| Key: AV = aortic valve; BMI = body mass index; CI = confidence interval; PVD = peripheral vascular disease; TAVI = transcatheter aortic valve implantation | |||
Discussion
To our knowledge, this is the first study to report survival at five years post-TAVI in AS patients with severe PH that has been determined noninvasively using transthoracic echocardiography. Our main statistical finding was that severe PH had no significant effect on all-cause mortality.
Characteristics of patients with severe PH
We report a prevalence of severe PH in our real-world TAVI cohort of 43%. We found that patients with severe PH were more likely to have a higher baseline creatinine, a greater incidence of LV impairment, and to exhibit a greater degree of dependence in their ADL, compared with patients without severe PH. Our finding of an increased incidence of LV impairment in patients with severe PH suggests that, as expected, most of the PH observed in our cohort was likely to involve an element of left heart disease. This has previously been observed in studies performing invasive right heart catheterisation in patients prior to TAVI, whereby either isolated postcapillary or combined pre- and postcapillary PH constituted 84–88% of TAVI patients with PH.7
The increased level of serum creatinine at baseline observed in our cohort of severe PH patients is likely to reflect the association between PH and renal dysfunction. This has been well-characterised outside of the TAVI population, with mechanisms relating to increased venous congestion, decreased cardiac output and neurohormonal activation previously proposed.9 Our findings of severe PH patients having a greater overall degree of disability, despite otherwise being generally well-matched in terms of comorbidities, to patients without severe PH is also of importance. This suggests that in severe AS patients referred for TAVI, the burden of disease is more likely to translate to increased dependence in ADL if concomitant severe PH is also present. It remains unclear whether improvements in sPAP or even reversal of PH post-TAVI translate directly to outcomes measuring dependence in ADL, however, this would be useful to consider given that both disability and frailty are highly prevalent in the TAVI population.10
PH and survival post-TAVI
We found no significant difference in mortality between patients with and without severe PH at five years’ follow-up. Many larger cohort studies, however, have previously shown that severe PH is associated with a significant increase in all-cause mortality up to two years post-TAVI.7 Marked variation notably exists between observational studies in how severe PH is both measured and defined. The most relevant to our analysis were the findings from the FRANCE-2 registry, as similar criteria were used to define PH. They found that PH significantly increased the odds of death at one-year following TAVI.11 There are several reasons that may explain the variation between our results.
The FRANCE-2 registry was a larger multi-centre cohort of 2,435 patients, with PH patients further subclassified according to the presence of a sPAP between 41–59 mmHg and >59 mmHg. This difference in sample size, specifically, is of potential importance in instances where the absolute differences in reported outcomes between groups are modest. In both our analysis and the FRANCE-2 registry, similar increases in the incidence of death in patients with severe PH of 5% and 6%, respectively, were found. In these situations, larger cohorts are more likely to be able to determine the presence of statistical significance. This is supported by the findings of several other smaller observational studies reporting severe PH outcomes at one to two years post-TAVI, and finding no statistically significant difference in all-cause mortality.12,13
There are also important differences to consider in patient follow-up times and the impact this may have on outcomes. Multiple studies have shown reductions in sPAP in patients with severe PH following TAVI. Alushi et al. performed serial sPAP measurements (using transthoracic echocardiography) in 617 AS patients undergoing TAVI, demonstrating a reduction in sPAP both at discharge and at one-year postprocedure. This correlated with a lower risk of death from all causes in the short-to-medium term.14 It is, therefore, plausible that with the longer-term follow-up used in our analysis, the impact of high sPAP measurements at baseline decreased with time in surviving TAVI patients due to the potential ongoing effects of improvements in pulmonary arterial haemodynamics.8
It is also important to note that patients with varying subcategories of PH behave differently over the long term. Sultan et al. performed a similar length five-year follow-up in TAVI patients with PH defined using invasive right-heart catheterisation (mean pulmonary artery pressure ≥25 mmHg). They found no significant difference in survival between patients without PH and patients with both isolated postcapillary PH and precapillary PH.15 Although categorisation of PH according to mean pulmonary capillary wedge pressure was not possible in our analysis, the generalisability of any outcomes in observational studies using invasive right heart catheterisation to diagnose PH are more limited, as this is not routinely performed in the clinical setting for patients undergoing TAVI, given it is an additional procedure that carries inherent risk. This analysis, therefore, provides additional insight, being the first study to assess the impact of severe PH estimated using more readily available echocardiographic data over a similar long-term follow-up period. Although our analysis should be taken in the context of its limitations, the finding of no significant increase in mortality in TAVI patients with severe PH after a five-year follow-up provides some reassurance regarding decision-making for suitability of TAVI in patients with both severe AS and PH, while placing importance on the identification and optimisation of other potentially important patient factors, such as BMI and PVD, that have a greater degree of association with long-term all-cause mortality, even in our relatively small-sized registry.
Lastly, the cohort included in our analysis was obtained from 2013 to 2017. This was later than many of the initial short-to-medium term observational studies looking at the effects of severe PH on all-cause mortality. Since 2002, there has been an evolution in TAVI methods and device technologies, which have resulted in improved patient outcomes.16,17
Patient factors associated with reduced survival
We report that BMI and PVD are significant factors to consider in patients with severe AS undergoing TAVI, showing significant independent associations with all-cause mortality, even after adjusting for other covariates. The impact of a low BMI on mortality outcomes in TAVI patients has been well characterised. In the CENTER collaboration, which was a large cohort of 12,381 TAVI patients, a low BMI (<18 kg/m2) was a strong predictor of survival up to one-year post-TAVI, even when compared with overweight and obese patients.18 Our analysis with a cohort of similar age and comorbidity burden, supports this finding and demonstrates that the effects of being underweight prior to TAVI extend to five-year survival outcomes. A lower BMI is often associated with increased risk of frailty in older patients.19 The use of comorbidity burden scores like the Charlson comorbidity index,20 validated frailty tools, such as the Rockwood clinical frailty scale,21 and the integration of comprehensive geriatric assessment are likely to improve frailty identification in this older population and further aid decision-making. This highlights the role of the geriatrician in the multi-disciplinary team (MDT) managing older patients undergoing TAVI. Geriatricians play a role in several key areas, including pre-operative assessment, optimising physical, functional and social issues, MDT decision-making, ethical considerations and individualised postoperative care and rehabilitation.22
A history of PVD being associated with a worse survival outcome in our cohort is also of interest. Retrospective analysis of US hospital admission data has shown that comorbid PVD in 189,216 TAVI patients was significantly associated with mortality and adverse clinical events at two-year follow-up, with a possible relation to alternative TAVI access routes initially suggested.23 The findings of our multi-variate analysis, however, provide further insight by showing that PVD remains a significant predictor of mortality even when adjusting for patients who undergo alternative TAVI access routes to a percutaneous transfemoral approach. This suggests that the increased risk observed in TAVI patients with a history of PVD are likely secondary to an increased overall cardiovascular risk burden, resulting in a greater likelihood of adverse events. Information on the cause of death of patients with PVD was not available in this analysis, and further research is required to ascertain reasons for mortality in this particularly vulnerable patient group.
Limitations
Our study has several limitations, being a retrospective observational analysis of a small single-centre registry. Given the primary aim of this study was to determine outcomes in TAVI patients with severe PH, patients without a recorded estimation of sPAP on echocardiography in our registry needed to be excluded. This introduces unavoidable selection bias into our analysis, with reasons for missing sPAP values not always clear. Repeat sPAP measurements were also not performed, and this limits the extent to which our findings of no significant increase in mortality in severe PH patients can be confidently attributed to any potential improvements in sPAP. Further studies employing serial sPAP measurements in the long-term assessment of TAVI patients postprocedure would, therefore, be of benefit. Furthermore, information on the underlying aetiology of the severe PH observed in our cohort was not available, as this is often attributed to left heart disease in the routine clinical care of TAVI patients. This, nonetheless, could have been an important confounding variable to consider and adjust for in our analyses. Additionally, in high-risk TAVI patients the importance of improved symptomology and quality of life outcomes cannot be understated. Since these were not captured in our registry, we acknowledge that our findings may not fully represent the complete picture of TAVI outcomes in patients with severe PH.
Conclusion
We found that the presence of severe PH in patients undergoing TAVI did not significantly increase risk of all-cause mortality at five years’ follow-up. Patients with severe PH were more likely to have a greater degree of renal impairment, disability and left ventricular dysfunction compared with patients without severe PH. Our overall findings suggest careful consideration be given to suitability for TAVI in patients who are underweight and have a history of PVD.
Key messages
- Pulmonary hypertension is a common finding in patients with severe aortic stenosis
- Pulmonary hypertension did not significantly increase the risk of all-cause mortality at five-year follow-up in patients undergoing transcatheter aortic valve implantation (TAVI)
- Comorbidities, such as low body mass index (BMI) and peripheral vascular disease, are associated with increased long-term all-cause mortality in patients with severe aortic stenosis undergoing TAVI
Conflicts of interest
JK is a consultant, proctor and holds a research contract with Boston Scientific, Edwards Lifesciences, Medtronic, Merill, Picardia and Abbott Vascular. HM, KM, RA, SSB, ER: none declared.
Funding
None.
Study approval
Approval was obtained locally for this study by the University Hospitals of Leicester Audit and Quality Improvement Programme (AQIP Reference 13786).
Data availability
Data can be made available upon reasonable request by contacting the Glenfield Hospital Audit Team. Data are stored on a secure NHS database within the University Hospitals of Leicester.
Acknowledgement
The authors would like to thank the Glenfield Hospital Audit Team for their collaboration in providing the registry data used for this analysis.
References
1. Carabello BA. Clinical practice. Aortic stenosis. N Engl J Med 2002;346:677–82. https://doi.org/10.1056/NEJMcp010846
2. Lung B, Baron G, Butchart EG et al. A prospective survey of patients with valvular heart disease in Europe: the Euro Heart survey on valvular heart disease. Eur Heart J 2003;24:1231–43. https://doi.org/10.1016/S0195-668X(03)00201-X
3. Smith CR, Leon MB, Mack MJ et al. Transcatheter versus surgical aortic-valve replacement in high-risk patients. N Engl J Med 2011;364:2187–98. https://doi.org/10.1056/NEJMoa1103510
4. Reardon MJ, Van Mieghem NM, Popma JJ et al. Surgical or transcatheter aortic-valve replacement in intermediate-risk patients. N Engl J Med 2017;376:1321–31. https://doi.org/10.1056/NEJMoa1700456
5. Tang M, Liu X, Lin C et al. Meta-analysis of outcomes and evolution of pulmonary hypertension before and after transcatheter aortic valve implantation. Am J Cardiol 2017;119:91–9. https://doi.org/10.1016/j.amjcard.2016.09.015
6. Cam A, Goel SS, Agarwal S et al. Prognostic implications of pulmonary hypertension in patients with severe aortic stenosis. J Thorac Cardiovasc Surg 2011;142:800–08. https://doi.org/10.1016/j.jtcvs.2010.12.024
7. Khalil S, Tabowei G, Kaur M et al. Effect of pulmonary hypertension on survival outcomes in patients with transcatheter aortic valve replacement: a systematic review and meta-analysis. Cureus 2024;16:e58540. https://doi.org/10.7759/cureus.58540
8. Sinning J-M, Hammerstingl C, Chin D et al. Decrease of pulmonary hypertension impacts on prognosis after transcatheter aortic valve replacement. EuroIntervention 2014;9:1042–9. https://doi.org/10.4244/EIJV9I9A177
9. Nickel NP, O’Leary JM, Brittain EL et al. Kidney dysfunction in patients with pulmonary arterial hypertension. Pulm Circ 2017;7:38–54. https://doi.org/10.1086/690018
10. Bertschi D, Moser A, Stortecky S et al. Evolution of basic activities of daily living function in older patients one year after transcatheter aortic valve implantation. J Am Geriatr Soc 2021;69:500–05. https://doi.org/10.1111/jgs.16927
11. Luçon A, Oger E, Bedossa M et al. Prognostic implications of pulmonary hypertension in patients with severe aortic stenosis undergoing transcatheter aortic valve implantation. Circ Cardiovasc Interv 2014;7:240–7. https://doi.org/10.1161/CIRCINTERVENTIONS.113.000482
12. Medvedofsky D, Klempfner R, Fefer P et al. The significance of pulmonary arterial hypertension pre- and post-transfemoral aortic valve implantation for severe aortic stenosis. J Cardiol 2015;65:337–42. https://doi.org/10.1016/j.jjcc.2014.06.008
13. Piazza N, Kalesan B, van Mieghem N et al. A 3-center comparison of 1-year mortality outcomes between transcatheter aortic valve implantation and surgical aortic valve replacement on the basis of propensity score matching among intermediate-risk surgical patients. JACC Cardiovasc Interv 2013;6:443–51. https://doi.org/10.1016/j.jcin.2013.01.136
14. Alushi B, Beckhoff F, Leistner D et al. Pulmonary hypertension in patients with severe aortic stenosis: prognostic impact after transcatheter aortic valve replacement: pulmonary hypertension in patients undergoing TAVR. JACC Cardiovasc Imaging 2019;12:591–601. https://doi.org/10.1016/j.jcmg.2018.02.015
15. Sultan I, Fukui M, Bianco V et al. Impact of combined pre and postcapillary pulmonary hypertension on survival after transcatheter aortic valve implantation. Am J Cardiol 2020;131:60–6. https://doi.org/10.1016/j.amjcard.2020.06.037
16. Giordano A, Corcione N, Ferraro P et al. Comparative one-month safety and effectiveness of five leading new-generation devices for transcatheter aortic valve implantation. Sci Rep 2019;9:17098. https://doi.org/10.1038/s41598-019-53081-w
17. Giordano A, Biondi-Zoccai G, Frati G. Transcatheter Aortic Valve Implantation: Clinical, Interventional and Surgical Perspectives. Switzerland: Springer International Publishing, 2019. https://doi.org/10.1007/978-3-030-05912-5
18. van Nieuwkerk AC, Santos RB, Sartori S et al. Impact of body mass index on outcomes in patients undergoing transfemoral transcatheter aortic valve implantation. JTCVS Open 2021;6:26–36. https://doi.org/10.1016/j.xjon.2021.03.012
19. Yuan L, Chang M, Wang J. Abdominal obesity, body mass index and the risk of frailty in community-dwelling older adults: a systematic review and meta-analysis. Age Ageing 2021;50:1118–28. https://doi.org/10.1093/ageing/afab039
20. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 1987;40:373–83. https://doi.org/10.1016/0021-9681(87)90171-8
21. Church S, Rogers E, Rockwood K, Theou O. A scoping review of the clinical frailty scale. BMC Geriatrics 2020;20:393. https://doi.org/10.1186/s12877-020-01801-7
22. Brunetti E, Lucà F, Presta R et al. A comprehensive geriatric workup and frailty assessment in older patients with severe aortic stenosis. J Clin Med 2024;13:4169. https://doi.org/10.3390/jcm13144169
23. Ullah W, Satti DI, Sana MK et al. Trends and outcomes of transcatheter aortic valve implantation in patients with peripheral arterial disease: insights from the national readmissions database. Curr Probl Cardiol 2023;48:101605. https://doi.org/10.1016/j.cpcardiol.2023.101605

