There is a high prevalence of frailty in people with heart failure, chronic kidney disease, type 2 diabetes, and multiple long-term conditions. These groups are eligible for treatment with sodium-glucose cotransporter 2 inhibitors (SGLT2i), with numerous large-scale trials demonstrating favourable clinical outcomes spanning disease states. There are now increasing data that the benefits of these agents are consistent across all frailty severities and are well tolerated. However, real-world data suggest a hesitancy in SGLT2i use in those with frailty. As a result, SGLT2i are either not initiated or discontinued inappropriately in people with frailty who are likely to derive benefit from these agents. This review critically evaluates current evidence and clinical guidelines for SGLT2i use in people with frailty, addressing safety concerns and offering practical recommendations for clinical practice.
Introduction
Sodium-glucose cotransporter 2 inhibitors
Sodium-glucose cotransporter 2 inhibitors (SGLT2i) demonstrate prognostic benefits independent of glycaemic control, most notably in the EMPA-REG OUTCOME trial.1 Subsequent large-scale randomised-controlled trials (RCTs) of SGLT2i consistently demonstrated improved cardiovascular outcomes irrespective of type 2 diabetes (T2D), extending to populations with heart failure (HF) and chronic kidney disease (CKD). This has culminated in the inclusion of SGLT2i in most T2D, HF and CKD guidelines.2
Frailty
There is no universally accepted definition of frailty, which may be understood to be an age-related and non-specific vulnerability to stressors associated with the accumulation of deficits (described by Rockwood) or a syndrome of “exhaustion, weakness, slowness, physical inactivity, and weight loss” (described by Fried).3,4 Frailty is strongly associated with disability, hospitalisation, and mortality. Often, disease management in individuals with frailty is considered a trade-off between disease and treatment burden,4–6 with emphasis on improving quality of life and preserving functional status. There is a recognised need to deprescribe and minimise polypharmacy – especially anticholinergic drugs – in people with frailty,4–7 which may provoke hesitancy to initiate new treatments. A person-centred approach to treatment is vital.4,7 Beneficial treatments should not be discontinued solely due to frailty,4,6 especially considering the close interplay of frailty with hospitalisations, inflammation, and symptom burden.3–6 Individuals with frailty are under-represented in clinical trials, especially those with advanced frailty or at the extremes of age.4,7 In clinical practice, assessments such as the Clinical Frailty Scale are commonly used; whereas in clinical trials, indices such as the Rockwood Frailty Index or Claims-Based Frailty Index may be employed, limiting direct comparison.6 For these reasons, careful consideration is necessary when prescribing in frailty.
Current guidance and clinical practice
Guidance for clinicians regarding the use of SGLT2i for people with frailty can be conflicting. Some recommend avoidance in moderate frailty,7 while others recommend consideration, even in severe frailty,5 given their cardioprotective features and low risk of hypoglycaemia.8 A recent observational study of >100,000 patients in Denmark found that patients with moderate or severe frailty were less likely to be initiated on a SGLT2i, even when controlling for comorbidity and polypharmacy.9 Those with moderate frailty were 14% less likely to be prescribed a SGLT2i, and those with severe frailty were 35% less likely, compared with non-frail individuals. A longitudinal cohort study, including 35,999 participants, of which 31.6% were frail, found that frailty severity was associated with significantly lower rates of initiation of HF therapy, including SGLT2i.10 This is perhaps unsurprising given the lack of consensus in major guidelines recommending SGLT2i use in frailty. Table 1 outlines these recommendations by a few notable societies.11–16
Table 1. Recommendations for sodium-glucose cotransporter 2 inhibitor (SGLT2i) use in people living with frailty
| Organisation; year of publication | Recommendations for SGLT2i in frailty/older adults |
| European Society of Cardiology, 2021 (including focused update 2023)11,12 | Notes that elderly and frail people are at particular risk of dehydration, hypotension, and pre-renal renal failure when SGLT2i are used No specific guidelines regarding when to initiate/continue/discontinue SGLT2i in individuals with frailty |
| American Heart Association/American College of Cardiology, 202213 | No specific guidelines regarding the use of SGLT2i in individuals with frailty |
| Kidney Disease Improving Global Outcomes, 202414 | No specific guidance regarding the use of SGLT2i in people with frailty |
| UK Kidney Association, 202315 | Suggests the following regarding the use of SGLT2i in individuals with frailty:
|
| American Diabetes Association/European Association for the Study of Diabetes, 202216 | Notes that in adults over 65 years of age, the cardiovascular and renal benefits of SGLT2i were consistent as with other populations Notes that individuals with frailty often have a higher medication burden and derive lesser benefit from strict glycaemic management, and recommends to consider deprescribing |
HF and frailty: scope to break the cycle?
Frailty and HF
Frailty is an established risk factor for HF, independent of age and comorbidity.17 Prevalence of frailty in those with HF is thought to be up to 75%.6 Heart failure with preserved ejection fraction (HFpEF) is more common than heart failure with reduced ejection fraction (HFrEF) in people with frailty. In a recent study of 645 individuals, participants with HFpEF (as opposed to HFrEF) were more likely to have both frailty (55.2% vs. 46.8%) and three or more comorbidities (77.9% vs. 65.6%).18 Frailty can be considered an independent risk factor for HFrEF, wherein SGLT2i forms the foundation of care showing prognostic benefits,19,20 alongside the non-steroidal mineralocorticoid receptor antagonist (MRA) finarenone.21 HFpEF remains a diagnostic challenge and continues to be underdiagnosed;22 especially in frailty, wherein HF often presents atypically or with non-specific symptoms.23
The efficacy of SGLT2i in patients with HF and frailty
Recent data emphasise that SGLT2i are both safe and effective in the management of HF in individuals with frailty (table 2).23–26 Post-hoc analyses of DELIVER and DAPA-HF trials,23,24 demonstrate the safety and efficacy of dapagliflozin in patients living with frailty in separate HF phenotypes. Post-hoc analysis of the EMPEROR-preserved trial evaluated the use of empagliflozin in patients with HFpEF and frailty.25 In all these studies, they defined frailty as per Rockwood’s accumulation of deficits,4 and used the Rockwood cumulative deficit frailty index (FI). Patients were categorised into three frailty subgroups according to the number of frailty deficits present: FI class 1, 2, and 3.
Table 2. Key studies evaluating the use of SGLT2i in frailty23–26
| Author, year | Study description | N, mean age | Frailty consideration | Main findings |
| Butt et al., 2022 | A post-hoc analysis of DAPA-HF trial in patients aged ≥65 years (safety and efficacy of dapagliflozin in HFrEF) | 4,742, 66.3 | Rockwood cumulative deficit frailty index |
|
| Butt et al., 2022 | A prespecified analysis of DELIVER trial in patients aged ≥65 years (safety and efficacy of dapagliflozin in preserved ejection fraction [LVEF >40%]) | 6,258, 72 | Rockwood cumulative deficit frailty index |
|
| Coats et al., 2024 | A post-hoc analysis of EMPEROR-preserved trial in patients living with frailty (safety and efficacy of empagliflozin in HFpEF) | 5,988, 71 | Rockwood cumulative deficit frailty index |
|
| Alfadas et al., 2024 | A systematic review and meta-analysis (safety and efficacy of SGLT2i in frail patients in the management of T2D and heart failure) |
|
||
| Desai et al., 2022 | A real-world study comparing the effect of empagliflozin with DPP4i or GLP-1 RA in older adults >66 years with T2D | 57,962 | Claims-based frailty index |
|
| Vart et al., 2023 | A post-hoc analysis of DAPA-CKD trial across various frailty categories | 433, 61.7 | Rockwood cumulative deficit frailty index |
|
| Mayne et al., 2024 | A post-hoc analysis of EMPA-KIDNEY trial across various frailty categories | 6,609, 63.8 | Predicted risk score of hospitalisation as a surrogate marker for frailty |
|
| Key: CI = confidence interval; CV = cardiovascular; DPP4i = dipeptidyl peptidase-4 inhibitor; eGFR = estimated glomerular filtration rate; FI = frailty index; GLP1-RA = glucagon-like peptide-1 receptor agonist; HF= heart failure; HFpEF = heart failure with preserved ejection fraction; HFrEF = heart failure with reduced ejection fraction; HR = hazard ratio; LVEF = left ventricular ejection fraction; RR = relative risk; SGLT2i = sodium-glucose cotransporter 2 inhibitor; T2D = type 2 diabetes; UTI = urinary tract infection | ||||
In the post-hoc analysis of the DAPA-HF trial, where 4,742 patients with HFrEF were randomised to dapagliflozin or placebo, 50% were found to be frail, and greater frailty was associated with worse outcomes, including HF hospitalisations and death. Dapagliflozin was found to reduce the risk of worsening HF, cardiovascular death and all-cause mortality, irrespective of frailty status. The number needed to treat (NNT) to prevent one cardiovascular event per 100 person-years were 31, 25, and 15 in FI classes 1, 2 and 3, respectively.24 Comparatively, the NNT for the use of aspirin in secondary prevention of serious vascular events in the general population (not specifically in persons with frailty) is around 50–100.27
In the subgroup analysis of the DELIVER trial – following 6,258 patients with HFpEF and heart failure with mildly reduced ejection fraction (HFmrEF) – dapagliflozin reduced cardiovascular events, as well as all-cause death, improved quality of life and reduced both hospital admissions and worsening of HF for all patients, regardless of frailty status. The hazard ratio (HR) of primary composite end points (worsening HF and cardiovascular death) with dapagliflozin compared with placebo was 0.82 (95% confidence interval [CI] 0.73 to 0.92), and NNT among FI of class 1, 2 and 3, were 40, 31 and 19, respectively, to prevent one primary end point over a median follow-up of 2.3 years.23 Crucially, improvements occurred early after initiation of treatment. The increase in absolute benefit for the frailest cohorts observed in both analyses is likely due to the risk of poor outcomes associated with frailty. Dapagliflozin was well tolerated in both studies – although there was a higher rate of discontinuation in individuals with a higher frailty index, discontinuation rates of SGLT2i were no different to placebo.
In the post-hoc analysis of the EMPEROR-preserved trial, including 5,988 patients with HFpEF, participants were classified into non-frail, mild, moderate and severe frailty classes. They confirmed the high prevalence of frailty among people with HFpEF – 74.9% were found to have mild to severe frailty. Cardiovascular death, or HF hospitalisations were reduced in the empagliflozin treatment group by a HR of 0.59 (95%CI 0.42 to 0.83), 0.79 (95%CI 0.61 to 1.01), 0.77 (95%CI 0.61 to 0.96) and 0.90 (95%CI 0.69 to 1.16) in the non-frail, mild frailty, moderate frailty and severe frailty groups, respectively.25
Lastly, a meta-analysis focused specifically on people with frailty, HF, and diabetes, found that SGLT2i were consistently associated with lower rates of all-cause mortality and cardiovascular events.26
T2D, CKD and frailty
There is a growing appreciation of a close interplay between metabolic, renal and cardiovascular disease leading to a clinically identifiable condition termed as the cardiovascular–kidney–metabolic syndrome.28 In this condition, the American Heart Association (AHA) has recommended the use of SGLT2i.29 T2D and CKD are risk factors for the development of both sarcopenia and frailty, and associated with worse outcomes and faster functional decline in people living with frailty.30,31
Efficacy of SGLT2i in T2D and frailty
A propensity-matched real-world study compared older adults with T2D (inclusion >66 years; mean 72 years of age) and a mean claims-based frailty index (CFI) of 0.18 (mild frailty) who were treated with empagliflozin as compared with dipeptidyl peptidase-4 inhibitors (DPP4i) or glucagon-like peptide-1 receptor agonist (GLP-1RA). Empagliflozin was associated with a reduced risk of total cardiovascular hospitalisations – relative risk (RR) 0.80 (95%CI 0.69 to 0.93) versus sitagliptin, and 0.88 (95%CI 0.77 to 1.00) versus GLP-1RA over a mean follow-up of 6.3 months.32 Similarly, a cohort study compared the efficacy of SGLT2i to DPP4i in 38,272 people >65 years old with T2D and frailty (CFI >0.25). In this cohort, SGLT2i were associated with 27.24 fewer cardiovascular events per 1,000 person-years (p<0.001). There were also 11.59 fewer primary safety outcomes per 1,000 person-years in the SGLT2i group as compared with the DPP4i group. HR of cardiovascular events for those on SGLT2i (rather than DPP4i) was also comparable between frail and non-frail cohorts – at 0.79 (95%CI 0.70 to 0.89) and 0.72 (95%CI 0.69 to 0.75), respectively.33
Efficacy of SGLT2i in CKD and frailty
Post-hoc analyses of DAPA-CKD34 and EMPA-KIDNEY35 studied the efficacy of SGLT2i in CKD with or without diabetes in participants living with frailty (table 2). Post-hoc analysis of DAPA-CKD found 73% of the participants were classified as having moderate to severe frailty (Rockwood FI). In those assigned dapagliflozin rather than placebo, HR for primary outcome (estimated glomerular filtration rate [eGFR] decline ≥50%, end-stage kidney disease, or kidney or cardiovascular death) was 0.50 (95%CI 0.33 to 0.76), 0.62 (95%CI 0.45 to 0.85) and 0.64 (95%CI 0.49 to 0.83) for non-frail, moderate frailty and severe frailty, respectively. Serious adverse effects (AEs) were less frequent in those assigned dapagliflozin as compared with placebo across the full spectrum of frailty.34
Post-hoc analysis of the EMPA-KIDNEY trial was conducted, where a score of predicted risk of hospitalisation during follow-up was used as a surrogate measure for frailty. Trial participants were divided into groups where this predicted risk was ≤20%; >20% to ≤35%; >35% to ≤45%; and >45%. For those who were assigned empagliflozin as opposed to placebo, HR for primary outcome (kidney disease progression or cardiovascular death) was 0.77 (95%CI 0.56 to 1.06), 0.65 (95%CI 0.53 to 0.80), 0.66 (95%CI 0.49 to 0.90), and 0.79 (95%CI 0.62 to 1.00) for each level of frailty, respectively. There was no significant difference in safety outcomes when comparing empagliflozin with placebo at any level of frailty. Moreover, the absolute risk reduction with empagliflozin was greater with increasing frailty. Per 1,000 person-years treated with empagliflozin, there was an estimated 14, 25 and 35 fewer primary outcomes in frailty groups ≤20%, >20% to ≤35%, and >35%, respectively.35
SGLT2i adverse effects in older adults
This section evaluates the current evidence regarding the safety concerns with SGLT2i among older adults. AEs should not be considered in isolation, nor concerns dismissed; as ever, a holistic and person-centred approach with shared decision-making is necessary – the purpose of this section is to facilitate this process and highlight relevant evidence.
Urinary tract and genital mycotic infections
SGLT2i-induced glycosuria is thought to provide an ideal environment for the growth of colonised bacteria. One study compared the urinary tract infection (UTI) risk between SGLT2i and placebo or active comparator in 86 RCTs, including 50,880 patients. The risk of UTI was not increased when compared with placebo (relative risk [RR] 1.03, 95%CI 0.96 to 1.11, I2 0%) or active comparator (RR 1.08, 95%CI 0.93 to 1.25, I2 22%). However, patients on SGLT2i were three times more likely to develop genital mycotic infections (RR 3.37).36 It is noteworthy that these data do not pertain specifically to individuals with frailty. The risk of UTIs is, therefore, an important clinical consideration, but seems to be over-emphasised in clinical practice. Caution should be exercised in patients that have a history of recurrent UTIs or with indwelling catheters, as these groups were excluded from many of the included studies.
Weight loss and sarcopenia
Weight loss is a hallmark of frailty and thought to directly contribute to sarcopenia. Weakness and fatigue, through loss of muscle mass in addition to body fat, has been shown to be associated with the development of frailty.37 A meta-analysis has shown that SGLT2i can lead to 1.5–2 kg weight reduction when compared with placebo,38 primarily through water and adipose tissue loss.
The EMPA-ELDERLY trial evaluated empagliflozin on muscle mass and strength, in addition to glucose control, in older adults with T2D. This study randomised 129 people with a mean age of 74.1 years, and the parameters observed were skeletal muscle index, grip strength, and quality of life assessments. The study found that empagliflozin improved glycaemic control and reduced body weight without compromising muscle mass or strength.39 This study included individuals of Japanese ethnicity with a body mass index (BMI) ≥22 kg/m2 and excluded individuals with co-existent sarcopenia.
Euglycaemic diabetic ketoacidosis
Euglycaemic ketoacidosis is defined as diabetic ketoacidosis (DKA) without marked hyperglycaemia, for which SGLT2i are a known cause. Reassuringly, most of the landmark clinical trials, such as DAPA-CKD, EMPA-KIDNEY and DAPA-HF, suggest that ketoacidosis is unlikely – particularly in individuals that do not have concurrent diabetes.40–42
A systematic review looking at possible precipitating factors for euglycaemic ketoacidosis in patients treated with SGLT2i, recommended caution in individuals with uncontrolled insulin-treated T2D and in type 1 diabetes (T1D).43 Among the 56 publications, 34 incidences of euglycaemic DKA were identified: 26 of these incidences were associated with canagliflozin, five with dapagliflozin, and one with empagliflozin. Other recommendations were to avoid excessive alcohol intake and avoidance of low carbohydrate diets. SGLT2i should also be withheld during periods of fasting and peri-operatively.44
Current American College of Cardiology guidelines advise stopping SGLT2i 3–4 days before non-cardiac surgery to reduce metabolic acidosis risk, while the American Diabetes Association recommends withholding them during severe illness in patients with ketonaemia.45,46 This is of particular concern in individuals with frailty, for whom acute illness is both more common and more severe. Improving awareness and adherence to these guidelines may reduce complications. However, this practice is increasingly debated. A recent case-control study in critically ill patients with T2D found no increased risk of ketoacidosis or adverse outcomes associated with SGLT2i use in intensive care.47 This discrepancy highlights the need for further research.
Volume depletion and orthostatic hypotension
In the DAPA-HF trial, investigating 4,500 participants, volume depletion occurred in 1.2% of the dapagliflozin group, and in 1.7% of subjects in the placebo group. There was no difference in renal AEs compared with placebo.42 These data were consistent among more frail individuals (FI class 3) seen in the DELIVER trial, as 3.2% of patients experienced volume depletion in the treatment group against 2.9% in the placebo group.23 Furthermore, a meta-analysis including 12,000 participants with T2D was unable to find a correlation between SGLT2i and increased risk of orthostatic hypotension. The results were consistent among patients >60 years of age.48
Given the burden of falls in older adults, deprescribing medications that lead to orthostatic hypotension is often warranted. However, it is important to deprescribe carefully; deprescribing diuretics and antihypertensives may be considered, rather than SGLT2i.
Falls and fractures
The relationship between falls and frailty is bidirectional; therefore, it remains an important consideration when using SGLT2i in older adults. Despite theoretical concerns, real-world and comparative data show no increased risk and may even suggest a reduced fall risk. A seven-year-long retrospective pharmacovigilance study compared individuals treated with SGLT2i against those treated with DPP4i and found 34% lower rates of falls with SGLT2i, and no differences in risks of fractures. It is important to note that, as a retrospective pharmacovigilance study, it could be associated with underreporting. Results were consistent across all age categories.49 A case-control study in Germany compared the fracture risk associated with different antidiabetic medications. In this real-world study, SGLT2i, compared with DPP4i, did not show an increased risk, even when analysed particularly among participants aged 75 years or above.50
Table 3 summarises the evidence presented above, and outlines expert clinical considerations and recommendations aimed at mitigating potential risks.
Table 3. Potential SGLT2i adverse effects and clinical considerations in frailty
| Adverse effect | Considerations in frailty |
| Risk of genital mycotic infection | Counsel to maintain adequate hydration, good genital hygiene Important to educate carers for those living with severe frailty |
| Risk of UTI | Avoid in patients with recurrent UTIs or those with indwelling long-term catheters |
| Sarcopenia | Caution in frail patients with severely low BMI. Emphasis on improving nutrition and cardiac rehabilitation should be done in this cohort |
| Euglycaemic ketoacidosis | Avoid in T1D, T3cD, ketosis-prone T2D Education regarding adherance to ‘sick-day rules’ Avoidance of low-carbohydrate diets Educate carers for those living with severe frailty |
| Orthostatic hypotension | Consider deprescribing other antihypertensives if clinically relevant Consider reducing dose of concurrent loop diuretics Consider reducing dose of concurrent ACEi/MRA |
| Falls/fractures | No evidence of increased risks with SGLT2i |
| Key: ACEi = angiotensin-converting enzyme inhibitor; BMI = body mass index; MRA = mineralocorticoid receptor antagonist; SGLT2i = sodium-glucose cotransporter 2 inhibitor; T1D = type 1 diabetes; T2D = type 2 diabetes; T3cD = type 3c diabetes; UTI = urinary tract infection | |
Conclusion and recommendations
HF, CKD and T2D are intimately interlinked, and over-represented in the frail population. Due to the high-risk nature of this population, the absolute benefit of evidence-based therapy can be most pronounced – it is, therefore, vital that individuals are not denied treatment due to frailty status alone. There is a growing body of evidence suggesting that SGLT2i have greater benefit than risk in individuals with frailty (figure 1). Trials have demonstrated significantly lower all-cause mortality and hospitalisations when SGLT2i are given for CKD or HF in individuals with frailty, and additionally low rates of discontinuation or serious AEs. Recent data suggest that concerns regarding the safety of SGLT2i in individuals with frailty have been overstated or can be mitigated, although further research into the effects of these drugs on skeletal muscle strength and risks of ketoacidosis in critical illness is warranted. As with all clinical trials, the most severely frail and the very oldest patients are likely to have been excluded, and particular care is required when prescribing for these individuals. Furthermore, the evidence presented here often relates to post-hoc analysis – there exists a need to conduct frailty-focused RCTs in future. Frail people are often exposed to polypharmacy and medications with a high anticholinergic burden; prescribing in frailty can be a difficult balance to strike. A person-centred approach with careful risk–benefit analysis and a shared decision-making process is paramount. SGLT2i are generally well tolerated, rapidly provide symptomatic benefit, and do not exhibit anticholinergic effects. Considering this, clinicians may wish to prioritise initiating or continuing these drugs in people with frailty.
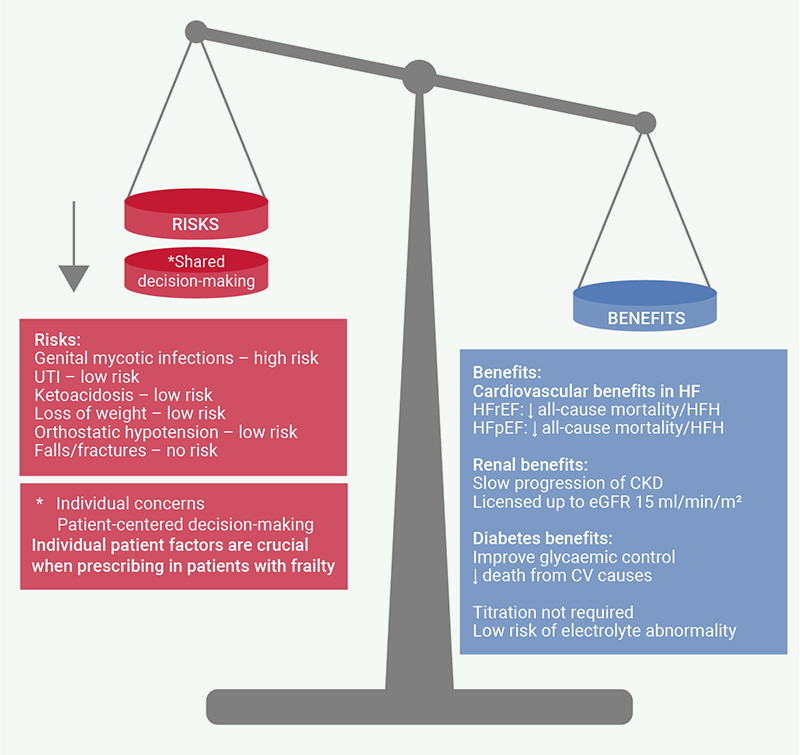
| Key: CKD = chronic kidney disease; CV = cardiovascular; eGFR = estimated glomerular filtration rate; HFH = heart failure hospitalisations; HFpEF = heart failure with preserved ejection fraction; HFrEF = heart failure with reduced ejection fraction; UTI = urinary tract infection |
Key messages
- Heart failure (HF), type 2 diabetes (T2D), chronic kidney disease (CKD) and frailty are significantly interlinked, and frailty is consistently associated with worse outcomes.
- Sodium-glucose cotransporter 2 inhibitors (SGLT2i) have demonstrated consistent benefit across the full spectrum of frailty.
- SGLT2i are generally well tolerated, including in individuals with frailty, and adverse effects can often be mitigated
Conflicts of interest
None declared.
Funding
None.
References
1. Zinman B, Wanner C, Lachin JM et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med 2015;373:2117–28. https://doi.org/10.1056/NEJMoa1504720
2. van der Aart-van der Beek AB, de Boer RA, Heerspink HJL. Kidney and heart failure outcomes associated with SGLT2 inhibitor use. Nat Rev Nephrol 2022;18:294–306. https://doi.org/10.1038/s41581-022-00535-6
3. Rockwood K, Mitnitski A. Frailty in relation to the accumulation of deficits. J Gerontol A Biol Sci Med Sci 2007;62:722–7. https://doi.org/10.1093/gerona/62.7.722
4. Kim DH, Rockwood K. Frailty in older adults. N Engl J Med 2024;391:538–48. https://doi.org/10.1056/NEJMra2301292
5. Strain WD, Down S, Brown P, Puttanna A, Sinclair A. Diabetes and frailty: an expert consensus statement on the management of older adults with type 2 diabetes. Diabetes Ther 2021;12:1227–47. https://doi.org/10.1007/s13300-021-01035-9
6. Pandey A, Kitzman D, Reeves G. Frailty is intertwined with heart failure: mechanisms, prevalence, prognosis, assessment, and management. JACC Heart Fail 2019;7:1001–11. https://doi.org/10.1016/j.jchf.2019.10.005
7. Bourdel-Marchasson I, Maggi S, Abdelhafiz A et al. Essential steps in primary care management of older people with type 2 diabetes: an executive summary on behalf of the European geriatric medicine society (EuGMS) and the European diabetes working party for older people (EDWPOP) collaboration. Aging Clin Exp Res 2023;35:2279–91. https://doi.org/10.1007/s40520-023-02519-3
8. Brown E, Heerspink HJL, Cuthbertson DJ, Wilding JPH. SGLT2i and GLP-1 receptor agonists: established and emerging indications. Lancet 2021;398:262–76. https://doi.org/10.1016/S0140-6736(21)00536-5
9. Malik ME, Butt JH, Strange JE et al. Initiation of SGLT2 inhibitors and GLP-1 receptor agonists according to level of frailty in people with type 2 diabetes and cardiovascular disease in Denmark: a cross-sectional, nationwide study. Lancet Healthy Longev 2023;4:e552–e560. https://doi.org/10.1016/S2666-7568(23)00164-2
10. Zahir Anjum D, Strange JE, Fosbøl E et al. Use of medical therapy and risk of clinical events according to frailty in heart failure patients – a real‐life cohort study. Eur J Heart Fail 2024;26:1717–26. https://doi.org/10.1002/ejhf.3249
11. McDonagh TA, Metra M, Adamo M et al. 2021 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J 2021;42:3599–726. https://doi.org/10.1093/eurheartj/ehab670
12. McDonagh TA, Metra M, Adamo M et al. 2023 focused update of the 2021 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J 2023;44:3627–39. https://doi.org/10.1093/eurheartj/ehad195
13. Heidenreich PA, Bozkurt B, Aguilar D et al. 2022 AHA/ACC/HFSA guideline for the management of heart failure: a report of the American College of Cardiology/American Heart Association joint committee on clinical practice guidelines. Circulation 2022;145:e895–e1032. https://doi.org/10.1161/CIR.0000000000001063
14. Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2024 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int 2024;105:S117–S314. https://doi.org/10.1016/j.kint.2023.10.018
15. Roddick AJ, Wonnacott A, Webb D et al. UK Kidney Association clinical practice guideline: sodium-glucose co-transporter-2 (SGLT-2) inhibition in adults with kidney disease 2023 update. BMC Nephrol 2023;24:310. https://doi.org/10.1186/s12882-023-03339-3
16. Davies MJ, Aroda VR, Collins BS et al. Management of hyperglycemia in type 2 diabetes, 2022. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care 2022;45:2753–86. https://doi.org/10.2337/dci22-0034
17. Butrous H, Hummel SL. Heart failure in older adults. Can J Cardiol 2016;32:1140–7. https://doi.org/10.1016/j.cjca.2016.05.005
18. Hamada T, Kubo T, Kawai K et al. Clinical characteristics and frailty status in heart failure with preserved vs. reduced ejection fraction. ESC Heart Fail 2022;9:1853–63. https://doi.org/10.1002/ehf2.13885
19. Solomon SD, McMurray JJV, Claggett B et al. Dapagliflozin in heart failure with mildly reduced or preserved ejection fraction. N Engl J Med 2022;387:1089–98. https://doi.org/10.1056/NEJMoa2206286
20. Anker SD, Butler J, Filippatos G et al. Empagliflozin in heart failure with a preserved ejection fraction. N Engl J Med 2021;385:1451–61. https://doi.org/10.1056/NEJMoa2107038
21. Solomon SD, McMurray JJV, Vaduganathan M et al. Finerenone in heart failure with mildly reduced or preserved ejection fraction. N Engl J Med 2024;391:1475–85. https://doi.org/10.1056/NEJMoa2407107
22. Heckman GA, McKelvie RS, Rockwood K. Individualizing the care of older heart failure patients. Curr Opin Cardiol 2018;33:208–16. https://doi.org/10.1097/HCO.0000000000000489
23. Butt JH, Jhund PS, Belohlávek J et al. Efficacy and safety of dapagliflozin according to frailty in patients with heart failure: a prespecified analysis of the DELIVER trial. Circulation 2022;146:1210–24. https://doi.org/10.1161/CIRCULATIONAHA.122.061754
24. Butt JH, Dewan P, Merkely B et al. Efficacy and safety of dapagliflozin according to frailty in heart failure with reduced ejection fraction. Ann Intern Med 2022;175:820–30. https://doi.org/10.7326/M21-4776
25. Coats AJS, Butler J, Tsutsui H et al. Efficacy of empagliflozin in heart failure with preserved ejection fraction according to frailty status in EMPEROR‐Preserved. J Cachexia Sarcopenia Muscle 2024;15:412–24. https://doi.org/10.1002/jcsm.13393
26. Aldafas R, Crabtree T, Alkharaiji M, Vinogradova Y, Idris I. Sodium-glucose cotransporter-2 inhibitors (SGLT2) in frail or older people with type 2 diabetes and heart failure: a systematic review and meta-analysis. Age Ageing 2024;53:afad254. https://doi.org/10.1093/ageing/afad254
27. Antithrombotic Trialists’ (ATT) Collaboration, Baigent C, Blackwell L et al. Aspirin in the primary and secondary prevention of vascular disease: collaborative meta-analysis of individual participant data from randomised trials. Lancet 2009;373:1849–60. https://doi.org/10.1016/S0140-6736(09)60503-1
28. Ndumele CE, Neeland IJ, Tuttle KR et al. A synopsis of the evidence for the science and clinical management of cardiovascular-kidney-metabolic (CKM) syndrome: a scientific statement from the American Heart Association. Circulation 2023;148:1636–64. https://doi.org/10.1161/CIR.0000000000001186
29. Ndumele CE, Rangaswami J, Chow SL et al. Cardiovascular-kidney-metabolic health: a presidential advisory from the American Heart Association. Circulation 2023;148:1606–35. https://doi.org/10.1038/s41574-021-00512-2
30. Bellary S, Kyrou I, Brown JE, Bailey CJ. Type 2 diabetes mellitus in older adults: clinical considerations and management. Nat Rev Endocrinol 2021;17:534–48. https://doi.org/10.1038/s41574-021-00512-2
31. Carrero JJ, Stenvinkel P, Cuppari L et al. Etiology of the protein-energy wasting syndrome in chronic kidney disease: a consensus statement from the International Society of Renal Nutrition and Metabolism (ISRNM). J Ren Nutr 2013;23:77–90. https://doi.org/10.1053/j.jrn.2013.01.001
32. Desai RJ, Glynn RJ, Everett BM et al. Comparative effectiveness of empagliflozin in reducing the burden of recurrent cardiovascular hospitalizations among older adults with diabetes in routine clinical care. Am Heart J 2022;254:203–15. https://doi.org/10.1016/j.ahj.2022.09.008
33. Kutz A, Kim DH, Wexler DJ, Liu J, Schneeweiss S, Glynn RJ, Patorno E. Comparative cardiovascular effectiveness and safety of SGLT-2 inhibitors, GLP-1 receptor agonists, and DPP-4 inhibitors according to frailty in type 2 diabetes. Diabetes Care 2023;46:2004–14. https://doi.org/10.2337/dc23-0671
34. Vart P, Butt JH, Jongs N et al. Efficacy and safety of dapagliflozin in patients with chronic kidney disease across the spectrum of frailty. J Gerontol A Biol Sci Med Sci 2024;79:glad181. https://doi.org/10.1093/gerona/glad181
35. Mayne KJ, Sardell RJ, Staplin N et al. Frailty, multimorbidity and polypharmacy: exploratory analyses of the effects of empagliflozin from the EMPA-KIDNEY trial. Clin J Am Soc Nephrol 2024;19:1119–29. https://doi.org/10.2215/CJN.0000000000000498
36. Puckrin R, Saltiel MP, Reynier P, Azoulay L, Yu OHY, Filion KB. SGLT-2 inhibitors and the risk of infections: a systematic review and meta-analysis of randomized controlled trials. Acta Diabetol 2018;55:503–14. https://doi.org/10.1007/s00592-018-1116-0
37. Fougère B, Morley JE. Weight loss is a major cause of frailty. J Nutr Health Aging 2017;21:933–5. https://doi.org/10.1007/s12603-017-0971-7
38. Liu XY, Zhang N, Chen R, Zhao JG, Yu P. Efficacy and safety of sodium-glucose cotransporter 2 inhibitors in type 2 diabetes: a meta-analysis of randomized controlled trials for 1 to 2 years. J Diabetes Complications 2015;29:1295–303. https://doi.org/10.1016/j.jdiacomp.2015.07.011
39. Yabe D, Shiki K, Homma G, Meinicke T, Ogura Y, Seino Y. Efficacy and safety of the sodium‐glucose co‐transporter‐2 inhibitor empagliflozin in elderly Japanese adults (≥65 years) with type 2 diabetes: a randomized, double‐blind, placebo‐controlled, 52‐week clinical trial (EMPA‐ELDERLY). Diabetes Obes Metab 2023;25:3538–48. https://doi.org/10.1111/dom.15249
40. Heerspink HJL, Stefánsson BV, Correa-Rotter R et al. Dapagliflozin in patients with chronic kidney disease. N Engl J Med 2020;383:1436–46. https://doi.org/10.1056/NEJMoa2024816
41. The EMPA-KIDNEY Collaborative Group; Herrington WG, Staplin N et al. Empagliflozin in patients with chronic kidney disease. N Engl J Med 2023;388:117–27. https://doi.org/10.1056/NEJMoa2204233
42. McMurray JJV, Solomon SD, Inzucchi SE et al. Dapagliflozin in patients with heart failure and reduced ejection fraction. N Engl J Med 2019;381:1995–2008. https://doi.org/10.1056/NEJMoa1911303
43. Burke KR, Schumacher CA, Harpe SE. SGLT2 inhibitors: a systematic review of diabetic ketoacidosis and related risk factors in the primary literature. Pharmacotherapy 2017;37:187–94. https://doi.org/10.1002/phar.1881
44. Rosenstock J, Ferrannini E. Euglycemic diabetic ketoacidosis: a predictable, detectable, and preventable safety concern with SGLT2i. Diabetes Care 2015;38:1638–42. https://doi.org/10.2337/dc15-1380
45. Thompson A, Fleischmann KE, Smilowitz NR et al. 2024 AHA/ACC/ACS/ASNC/HRS/SCA/SCCT/SCMR/SVM guideline for perioperative cardiovascular management for noncardiac surgery. J Am Coll Cardiol 2024;150:e351–e442. https://doi.org/10.1161/CIR.0000000000001285
46. ElSayed NA, Aleppo G, Aroda VR et al. 16. Diabetes care in the hospital: standards of care in diabetes – 2024. Diabetes Care 2024;47(suppl 1):S295–S306. https://doi.org/10.2337/dc24-S016
47. Mårtensson J, Cutuli SL, Osawa EA et al. Sodium glucose co-transporter-2 inhibitors in intensive care unit patients with type 2 diabetes: a pilot case control study. Crit Care 2023;27:189. https://doi.org/10.1186/s13054-023-04481-y
48. Rong X, Li X, Gou Q, Liu K, Chen X. Risk of orthostatic hypotension associated with sodium-glucose cotransporter-2 inhibitor treatment: a meta-analysis of randomized controlled trials. Diab Vasc Dis Res 2020;17:147916412095362. https://doi.org/10.1177/1479164120953625
49. Mascolo A, Rafaniello C, di Mauro G et al. Sodium-glucose cotransporter-2 (SGLT2) inhibitors and the reporting of falls and fractures: an european pharmacovigilance analysis. Front Pharmacol 2023;14:1245642. https://doi.org/10.3389/fphar.2023.1245642
50. Schmedt N, Andersohn F, Walker J, Garbe E. Sodium-glucose co-transporter-2 inhibitors and the risk of fractures of the upper or lower limbs in patients with type 2 diabetes: a nested case-control study. Diabetes Obes Metab 2019;21:52–60. https://doi.org/10.1111/dom.13480

