Infective endocarditis (IE) is a rare, life-threatening infection of the cardiac endocardium caused by bacterial seeding. On the other hand, atopic dermatitis (AD) is a common inflammatory skin condition that disrupts the epidermal barrier, increasing susceptibility to Staphylococcus aureus colonisation and recurrent bacteraemia. This provides a biologically plausible bridge to IE, which we explore in this current literature review.
MEDLINE, Embase and CENTRAL were searched from inception to 7 April 2025. Sixteen studies were included – 15 case reports and one retrospective cohort study – which were grouped into three categories: well-controlled AD with no predisposing risk factors, AD with predisposing risk factors, and uncontrolled AD or alternative AD management.
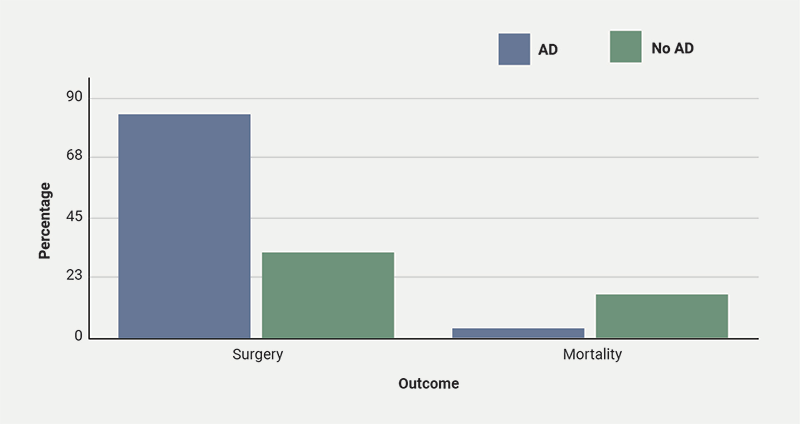
This review is made up of 24 patients with IE secondary to AD. Of the 16 studies, nine originated from Japan. Mean age was 29.6 years (range 15–42 years), which is significantly younger than classic IE cohorts (mean 60.0 years). Staph. aureus accounted for 23/24 patients, and the mitral valve was affected in 18/24 cases. In terms of patient outcomes, all but one patient survived, but valve surgery was necessary in 20/24 patients.
In conclusion, AD-related IE shows a distinct pattern: younger patients, Staph. aureus dominance and mitral valve involvement requiring mitral valve surgery in the majority. This potential link appears to be under-recognised, hence, greater cross-speciality awareness and rigorous AD control for those at high risk is recommended. The heavy Japanese skew in the literature highlights the need for broader case documentation in other regions to confirm this link across different patient demographics.
Introduction
Atopic dermatitis (AD) is a chronic inflammatory skin condition characterised by dry, inflamed, and pruritic skin due to skin barrier dysfunction.1 This often results in painful, open skin lesions that serve as a breeding ground for infection. This condition creates a vicious cycle as an impaired skin barrier leads to itchy skin, which triggers scratching behaviour, leading to further damage to the skin.2 AD is notably common, with a lifetime prevalence of 15–30% in children and 2–10% in adults.3 First-line management in primary care consists of regular emollients, avoidance of certain triggers and short courses of topical corticosteroids. However, patients whose disease remains uncontrolled, may be referred to secondary care for escalation to phototherapy or systemic medications, such as methotrexate and ciclosporin.4
Table 1. Summary of predisposing factors for infective endocarditis (IE)
| Predisposing factors |
| Congenital heart disease |
| Valvular heart disease |
| Prosthetic valve |
| Bicuspid aortic valve |
| Previous IE |
| Previous IE |
| Long-term IV catheter |
| Immunocompromised |
| IV drug use |
| Poor dentition |
| Adapted from Baddour et al.8 Key: IE = infective endocarditis; IV = intravenous |
On the other hand, infective endocarditis (IE) is a much rarer condition with an estimated annual incidence of 3–10 cases per 100,000 people.5 It is defined as an infection of the endocardium – the inner lining of the heart – often involving the heart valves. Classical predisposing conditions are summarised in table 1. IE represents a serious medical emergency and, as noted by William Osler, “few diseases present greater difficulties in the way of diagnosis than malignant endocarditis”.6 This underscores the clinical challenges IE presents, and reinforces the importance of exploring this potential link, as a common condition like AD may lead to pathways for pathogens to enter the bloodstream and colonise cardiac tissue.
IE can be categorised into two types: acute and subacute. In subacute IE, the pathogenesis typically begins with prodromal damage to the endocardial lining, often due to turbulent blood flow. This is often from an underlying valve abnormality, such as regurgitation or stenosis. This damage promotes platelet aggregation and fibrin deposition, which leads to a sterile thrombus known as a non-bacterial thrombotic endocarditis (NBTE). Then, if bacteria colonise this vegetation, it will lead to infective endocarditis.9 This can be from less virulent pathogens, such as Streptococcus viridans.10
In contrast, acute IE does not require any previous endothelial damage. This can occur due to extremely virulent species of pathogen, e.g. Staphylococcus aureus, that can directly adhere to the healthy heart endocardial tissue. From there, it causes damage by migration of neutrophils and macrophages, leading to the release of inflammatory cytokines. As the name suggests, acute IE has a rapid and aggressive time course in which a patient usually presents with a high-grade fever and rapid clinical deterioration. If not treated promptly, the patient can die in a matter of days.11
Patients with AD are more susceptible to pathogenic colonisation – particularly Staph. aureus. This is due to numerous different reasons, although the exact pathology is not yet fully understood. In summary, the skin of patients with AD have less filaggrin and filaggrin degradation products (FDPs), which results in weaker corneocytes and allows the pathogen to bind to the skin with greater affinity. Additionally, T-helper 2 (Th2) cytokines like interleukin-4 (IL-4) and interleukin-13 (IL-13) are overexpressed, leading to the downregulation of antimicrobial peptides, such as Leu-Leu 37 (LL-37) and human beta-defensin 3 (HBD-3),12 which directly kill Staph. aureus. Thus, the increased colonisation susceptibility is not only due to open wounds, but also because the principal skin of AD patients is intrinsically more vulnerable. Moreover, skin colonisation with Staph. aureus is estimated at 60–100% in patients with AD compared with 5–30% in healthy individuals.12,13
This literature review explores the current evidence linking AD, a common chronic inflammatory skin disease, and IE, a rare but potentially fatal cardiac infection. Here, we aim to map out the relevant research: epidemiological data, clinical case studies, and pathophysiological mechanisms. Understanding this relationship has important clinical implications given the stark juxtaposition between the commonality of AD and the severity of IE – with a one-year mortality rate of 30%.7 The current review examines the literature surrounding the mechanisms that connect these two distinct conditions, focusing on different patient subgroups and identifying gaps for further research on this unexpected connection.
Method
A literature search was conducted using Embase, CENTRAL, and MEDLINE from inception to 7 April 2025 using MeSH and free-text terms: (“atopic dermatitis” OR “eczema” OR “Dermatitis, Atopic”/MeSH) AND (“infective endocarditis” OR “bacterial endocarditis” OR “endocardit*” OR “Endocarditis, Bacterial”/MeSH). Limits were human studies and English language only. The relevant retrieved studies were imported into Covidence (Veritas Health Innovation, Melbourne, Australia) and were screened in order to identify the relevant studies for each of the different links between IE and AD. The majority of the studies were case reports, mainly of Japanese origin, and there were no systematic reviews linking the two. The included studies were grouped into three categories:
- A direct link between AD and IE with no other predisposing factors
- A direct link between IE and AD with additional predisposing factors
- Instances of IE due to complications/noncompliance with AD treatment.
Results
The results of the literature review are summarised in figure 1 and table 2. Those included 15 case reports and one retrospective cohort study, with a total number of 24 patients.
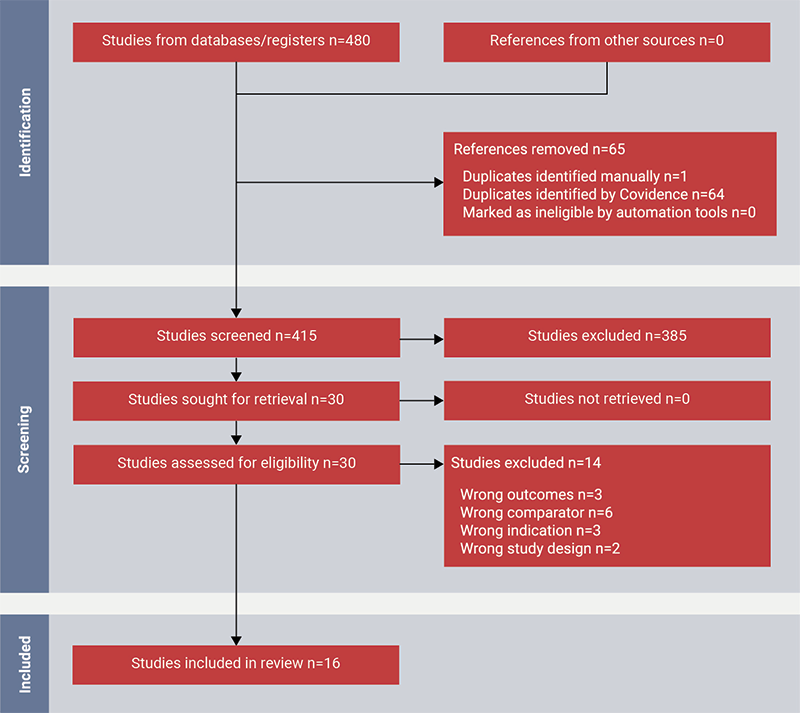
Table 2. Clinical characteristics of included cases of infective endocarditis (IE) associated with atopic dermatitis (AD) (n=24)
| Study | Age, years | Sex (M/F) | Micro-organism | Valve | Surgery (Y/N) | Outcome | Short summary |
| Mohiyiddeen et al.17 | 30 | M | MSSA | Mitral | Y | Survived | Severe AD with MSSA IE involving mitral valve; underwent successful valve replacement after initial medical therapy |
| Benenson et al.18 | 36 | F | MSSA | Mitral | Y | Survived | AD with MSSA IE causing mitral valve vegetation; required ICU admission; full recovery post-treatment |
| Park et al.19 | 22 | M | MSSA | Mitral | N | Survived | Developed MSSA mitral valve IE; treated with IV antibiotics; recovered without surgery |
| Park et al.19 | 42 | F | MSSA | Mitral | N | Survived | Developed MSSA mitral valve IE with cerebral and splenic emboli; managed medically with IV antibiotics |
| Patel and Jahnke20 | 17 | F | MRSA | Mitral | Y | Died | Severe AD developed MRSA mitral valve IE; underwent valve replacement but died later with many residual functional deficits |
| Tsuboi et al.21 | 42 | F | VRSA | Mitral | Y | Survived | AD with MSSA IE; ICU admission and mitral valve replacement; full recovery post-treatment |
| Woodun et al.22 | 29 | M | MSSA | Tricuspid | Y | Survived | AD managed by general practitioner; tricuspid valve repair; full recovery |
| Micallef and Ramphul23 | 39 | M | MSSA | Mitral | Y | Survived | AD with MSSA IE; underwent mitral valve repair and closure of patent foramen ovale; full recovery |
| Kitagata et al.24 | 26 | F | MSSA, MRSE | Pulmonary | Y | Survived | Congenital VSD; MSSA IE; full recovery post-surgery |
| Tamura and Abe25 | 18 | M | MSSA | Mitral | Y | Survived | Presented with 5-day fever and transient loss of consciousness; MSSA IE; full recovery post-surgery |
| Fukunaga et al.26 | 20 | M | MSSA | Mitral | Y | Survived | Pre-existing MR; underwent mitral valve plasty; post-surgery recovery |
| Fukunaga et al.26 | 20 | F | MRSA | Tricuspid | Y | Survived | Pre-existing TR; underwent tricuspid valve plasty; recovery post-surgery |
| Fukunaga et al.26 | 37 | M | MSSA | Mitral | Y | Survived | Pre-existing MR; underwent mitral valve plasty; recovery post-surgery |
| Fukunaga et al.26 | 31 | M | MSSA | Mitral | Y | Survived | Pre-existing MR; underwent mitral valve plasty; post-surgery recovery |
| Fukunaga et al.26 | 40 | M | MSSA | Mitral | Y | Survived | Pre-existing MR and ASD; underwent mitral valve plasty and ASD closure |
| Fukunaga et al.26 | 21 | F | Strep. viridans | Mitral | Y | Survived | Pre-existing MR; underwent mitral valve plasty; post-surgery recovery |
| Fukunaga et al.26 | 28 | M | MSSA | Mitral | Y | Survived | Pre-existing MR; underwent mitral valve plasty; post-surgery recovery |
| Fukunaga et al.26 | 30 | M | MSSA | Aortic + Mitral | Y | Survived | Pre-existing MR and AR; underwent mitral valve plasty and aortic valve replacement |
| Buckley27 | 15 | M | MSSA | Tricuspid | N | Survived | Fortnightly acupuncture into inflamed skin around knees; IV antibiotics; will require lifelong cardiology review |
| Furumura et al.29 | 41 | F | MSSA | Mitral | Y | Survived | Refused standard medical treatment; folk medicine instead such as spa therapy; mitral valve replacement surgery |
| Okada et al.28 | 23 | M | MSSA | Mitral | Y | Survived | Patient performed unsanitary cupping therapy; mitral valvuloplasty performed in the chronic phase after brain surgery |
| Hibi and Ito30 | 46 | F | MSSA | Mitral | N | Survived | Untreated AD since childhood; refused surgical intervention; 6-week antibiotic therapy |
| Aoyagi et al.31 | 24 | M | MSSA | Aortic | Y | Survived | Always refused topical steroid treatment for AD; developed MSSA aortic valve IE with cerebral embolism; underwent successful valve replacement and recovered well |
| Horimoto et al.32 | 34 | F | MSSA | Tricuspid | Y | Survived | Uncontrolled AD developed right-sided IE; underwent tricuspid valve replacement and recovered well post-surgery |
| Key: AR = aortic valve regurgitation; ASD = atrial septal defect; ICU = intensive care unit; IV = intravenous; MR = mitral valve regurgitation; MRSA = methicillin-resistant Staph. aureus; MRSE = methicillin-resistant Staph. epidermis; MSSA = methicillin-sensitive Staph. aureus; TR = tricuspid valve regurgitation; VRSA = vancomycin-resistant Staph. aureus; VSD = ventricular septal defect | |||||||
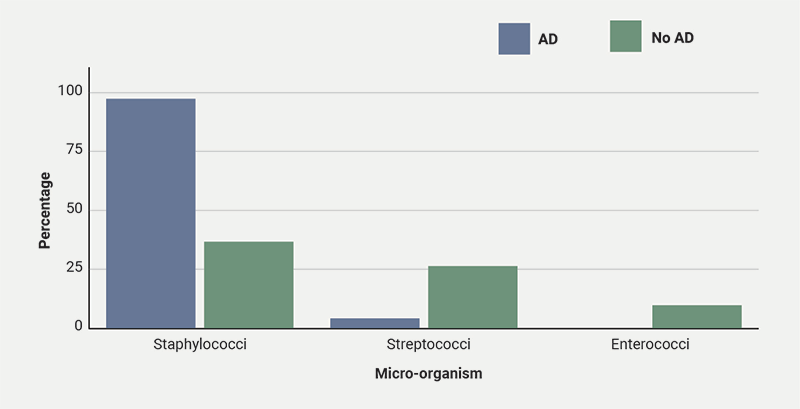
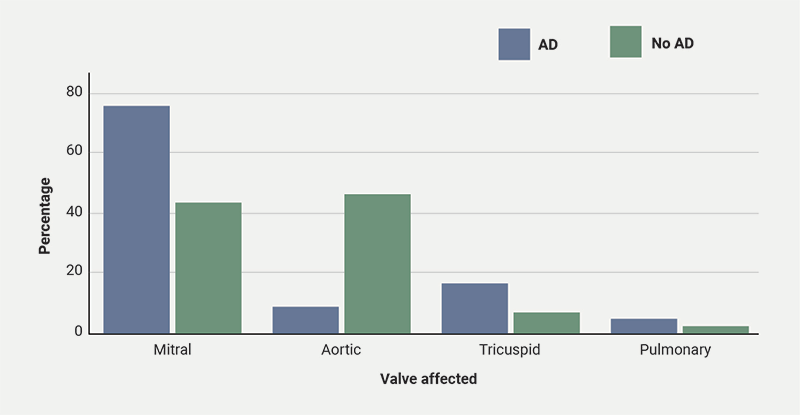
We extracted four main data points: patient age, micro-organism cultured, valve affected and patient outcomes. In patients with AD, the mitral valve was the most commonly affected (75.0%). On the other hand, in patients without AD, the aortic valve was more frequently affected (46.0%).14 This variation may reflect different patterns of bacterial seeding associated with AD. With regards to age, patients with AD have a much lower mean age compared with those without (29.6 vs. 60.0 years),15 suggesting that AD is associated with IE even in individuals with otherwise healthy heart tissue. In contrast, older patients without AD tend to develop IE due to having weaker immune systems and age-related cardiac vulnerabilities. Lastly, out of 24 patients with both AD and IE, 23 were infected with Staphylococci species and one patient with Streptococci species. However, in non-AD patients, the causative micro-organisms were more evenly spread among Staphylococci, Streptococci and Enterococci species.14 This finding aligns with the well-established increased presence of Staph. aureus colonisation on the skin of AD patients. Lastly, in terms of patient outcomes, we assessed two measures: valve surgery and mortality. Valve surgery was required in 20 of 24 patients (83.3%), which is much higher than the 32.4% reported in non-AD IE cohorts.16 In contrast, only one of 24 patients (4.17%) died, well below the approximate one-year mortality rate of 30% for patients without AD.7 This difference – more surgery but fewer deaths – may reflect the cohort’s younger age and lower comorbidity burden. Graphical representation of these findings can be seen in figures 2–4.
Direct link between IE and AD with no predisposing factors
The direct link between IE and AD in patients with no predisposing factors and who have controlled AD (no steroid phobia and are receiving the appropriate treatment) is arguably the most important group to observe, because they fall outside of a clinician’s usual high-risk radar. This may potentially lead to a delayed diagnosis and consequently a poorer prognosis for the patient. For this group, we included six studies.17–22 Across the six studies included in this category, all the patients shared similar characteristics: young age, no traditional IE risk factors, controlled AD and Staph. aureus as the only pathogen. Out of the seven included patients, six survived, but five required valve surgery.
Direct link between IE and AD with predisposing factors
Endocarditis typically develops when endocardial injury is followed by bacteraemia.7 The principal predisposing factors are summarised in table 1. In particular, lesions that lead to turbulent flow (e.g. structural cardiac abnormalities) promote platelet-fibrin deposition on the endocardium, which can be seeded during bacteraemia to form vegetations. For this subgroup, all four studies23–26 shared two key features: MSSA (methicillin-susceptible Staph. aureus) was the predominant colonising organism, and the patients were considerably younger than the typical IE patient – mean age of 28.2 years. For the 11 included patients, all underwent surgery and all survived.
Complications/non-compliance
This subgroup presents intriguing and unexpected connections between the unconventional treatment of AD or poorly controlled AD leading to IE. Six cases were identified,27–32 in which young patients with no traditional risk factors for IE (intravenous drug users, recent dental work, prosthetic valves or structural cardiac abnormalities) developed IE after either turning to alternative therapy for their AD or having untreated, long-standing AD. These include acupuncture,27 cupping therapy,28 folk medicine,29 and refusal of medical treatment.31 In each instance, the common link was an uncontrolled flare-up and exposure to non-sterile procedures – the acupuncture and cupping were conducted on the flared-up area of skin. These patients all developed MSSA IE, which underscores the dangers of how, if AD is mismanaged or left unmanaged, it can escalate into a life-threatening infection. In terms of outcomes, for the six patients, four underwent surgery but all survived.
Discussion
Table 3. Modified Duke criteria for infective endocarditis (IE)
| Major criteria | ||||||||
| Positive blood cultures for endocarditis | ||||||||
| Evidence of endocardial involvement | ||||||||
| Minor criteria | ||||||||
| Predispositiona | ||||||||
| Temperature >38.0°C | ||||||||
| Vascular phenomenab | ||||||||
| Immunologic phenomenac | ||||||||
| Microbiologic evidenced | ||||||||
| Definite IE | ||||||||
| Two major, or one major and three minor, or five minor | ||||||||
| Possible IE | ||||||||
| One major and one minor, or three minor | ||||||||
Adapted from Fowler et al.33 |
The modified Duke criteria are a set of clinical criteria used to aid in the diagnosis of IE (table 3), with an overall sensitivity of 80%.34 Among the minor criteria are predisposing heart conditions and intravenous drug users. The main, widely known and acknowledged risk factors include congenital or structural abnormalities of the heart, such as ventricular septal defects or bicuspid aortic valves. Intravenous drug use, often associated with nonsterile practices, increases the exposure to virulent pathogens. Also, recent dental procedures are a known risk factor, as commensal bacteria in the mouth flora may be introduced into the bloodstream, potentially leading to IE. However, severe AD is rarely considered a risk factor for IE, even though there is evidence to suggest it may increase the risk of IE, even in patients with no underlying heart defects. This highlights the importance of greater awareness among the medical professionals involved in managing IE: cardiologists, cardiothoracic surgeons, microbiologists, etc., as improved recognition may facilitate earlier diagnosis, better treatment progression and overall outcomes for patients.
The overwhelming predominance of case reports serves as a limitation, as they constitute the lowest level of evidence in the hierarchy of evidence.35 Because each case report involves just a single patient, no further statistical analysis can take place. Notably, nine of the 16 studies (56.3%) originated from Japan. Moreover, the only observational study on AD and IE was conducted in Japan, questioning the generalisability of the findings. There may be additional reasons to explain the abundance of literature from Japan, which are discussed next. Nakayama et al.36 conducted a cross-sectional study with 1,054 Japanese adults, evaluating the health literacy of the Japanese population against the European population. They found that, in general, health literacy was lower in Japan than in Europe. This coincides with the findings that medication adherence in Japanese patients with AD is positively correlated with their health literacy.37 One contributing factor may be the absence of a comprehensive national online platform to access reliable and understandable health information in Japan.36 Another explanation may be due to differences in the management of AD in Japan compared with other countries. Baron et al.38 compared the treatment preferences of dermatologists in Japan, USA and UK. They found that “USA and UK physicians were significantly more aggressive in prescribing systemic medications, such as steroids, antibiotics and immunosuppressants, compared with those in Japan”, and also “the use of alternative remedies was highest in Japan”. Hence, suboptimal control of AD due to differences in practice may increase the risk of infection, and, in turn, amplify the incidence of IE secondary to AD. Lastly, Japan’s strong research interest into this link may inflate the number of published cases. This suggests that wider awareness and documentation in other regions are needed to clarify whether this link appears in different patient demographics.
This synthesis of the current evidence highlights the rare, but important, link between AD and IE. It also stresses the importance of professional medical guidance in managing AD. Additionally, the general consensus is that there ought to be greater involvement of specialist dermatologists in the care of patients with severe AD, with some suggesting a role for prophylactic biologic therapy,22 especially if there is a history of underlying heart defects. The economic burden to the National Health Service (NHS) to treat IE – including hospital admissions, cost of treatment and potentially surgery – had a mean cost of £12,963 per patient in 2020.39 Raising awareness of this risk may support earlier diagnosis and consideration of prophylactic strategies, potentially improving outcomes for patients.
Conclusion
This review has examined the evidence linking IE and AD, suggesting that, although rare, there is a relevant connection between the two. Notably, the younger age of patients with AD who developed IE highlights a shift from the traditionally older demographic. Further observational studies and clinical trials are needed to clarify the nature of this association and inform updates to clinical guidelines and IE diagnostic criteria.
Key messages
- Atopic dermatitis may be an under-recognised risk factor for infective endocarditis; this review highlights an emerging link between severe atopic dermatitis and infective endocarditis, even in young, otherwise healthy individuals
- Skin barrier dysfunction in atopic dermatitis facilitates pathogenic entry, especially Staphylococcus aureus
- Infective endocarditis in patients with atopic dermatitis appears to affect younger patients, disproportionately affecting the mitral valve, while being predominantly caused by Staphylococcal species
- Mismanagement or non-adherence to treatment in atopic dermatitis can lead to potentially life-threatening outcomes. This underscores the need for proper dermatological management and patient education
- Greater clinical awareness and research are needed to address this link, given the serious implications and economic burden of infective endocarditis
Conflicts of interest
None declared.
Funding
None.
Acknowledgement
OL would like to express his sincere gratitude to Professor Terry McCormack who supervised him during the initial stages of this report, for all his guidance and support with this project.
References
1. National Institute of Arthritis and Musculoskeletal and Skin Diseases. Atopic dermatitis. Available at: https://www.niams.nih.gov/health-topics/atopic-dermatitis
2. National Eczema Association. Atopic dermatitis. Available at: https://nationaleczema.org/types-of-eczema/atopic-dermatitis/
3. Bieber T. Atopic dermatitis. Ann Dermatol 2010;22:125. https://doi.org/10.5021/ad.2010.22.2.125
4. Cunliffe T. Atopic eczema. Primary Care Dermatology Society. Available at: https://www.pcds.org.uk/clinical-guidance/atopic-eczema
5. Taduru SS. 30-year trends of incidence and mortality of infective endocarditis in the United States – unveiling the age- and gender-related and regional disparities. Am J Cardiol 2023;204:421–2. https://doi.org/10.1016/j.amjcard.2023.07.127
6. Osler W. The Gulstonian lectures on malignant endocarditis. BMJ 1885;1:522–6. https://doi.org/10.1136/bmj.1.1263.522
7. Cahill TJ, Prendergast BD. Infective endocarditis. Lancet 2016;387:882–93. https://doi.org/10.1016/S0140-6736(15)00067-7
8. Baddour LM, Wilson WR, Bayer AS et al. Infective endocarditis in adults: diagnosis, antimicrobial therapy, and management of complications. A scientific statement for healthcare professionals from the American Heart Association. Circulation 2015;132:1435–86. https://doi.org/10.1161/CIR.0000000000000296
9. Yallowitz AW, Decker LC. Infectious endocarditis. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2025. Available from: https://www.ncbi.nlm.nih.gov/books/NBK557641/
10. Doern CD, Burnham C-AD. It’s not easy being green: the viridans group streptococci, with a focus on pediatric clinical manifestations. J Clin Microbiol 2010;48:3829–35. https://doi.org/10.1128/JCM.01563-10
11. Armstrong GP. Infective endocarditis. MSD Manual Consumer Version, 2024. Available at: https://www.msdmanuals.com/home/heart-and-blood-vessel-disorders/endocarditis/infective-endocarditis
12. Kim J, Kim BE, Ahn K, Leung DYM. Interactions between atopic dermatitis and staphylococcus aureus infection: clinical implications. Allergy Asthma Immunol Res 2019;11:593–603. https://doi.org/10.4168/aair.2019.11.5.593
13. Park H-Y, Kim C-R, Huh I-S et al. Staphylococcus aureus colonization in acute and chronic skin lesions of patients with atopic dermatitis. Ann Dermatol 2013;25:410–16. https://doi.org/10.5021/ad.2013.25.4.410
14. Tzoumas A, Sagris M, Xenos D et al. Epidemiological profile and mortality of infective endocarditis over the past decade: a systematic review and meta-analysis of 133 studies. Am J Cardiol 2025;244:67–88. https://doi.org/10.1016/j.amjcard.2025.02.023
15. Ahtela E, Oksi J, Porela P, Ekström T, Rautava P, Kytö V. Trends in occurrence and 30-day mortality of infective endocarditis in adults: population-based registry study in Finland. BMJ Open 2019;9:e026811. https://doi.org/10.1136/bmjopen-2018-026811
16. Bin Abdulhak AA, Baddour LM, Erwin PJ et al. Global and regional burden of infective endocarditis, 1990–2010. Glob Heart 2014;9:131–43. https://doi.org/10.1016/j.gheart.2014.01.002
17. Mohiyiddeen G, Brett I, Jude E. Infective endocarditis caused by Staphylococcus aureus in a patient with atopic dermatitis: a case report. J Med Case Rep 2008;2:143. https://doi.org/10.1186/1752-1947-2-143
18. Benenson S, Zimhony O, Dahan D et al. Atopic dermatitis – a risk factor for invasive Staphylococcus aureus infections: two cases and review. Am J Med 2005;118:1048–51. https://doi.org/10.1016/j.amjmed.2005.03.040
19. Park BW, Shin YS, Cho EB, Park EJ, Kim KH, Kim KJ. Two cases of infective endocarditis in patients with atopic dermatitis. Ann Dermatol 2019;31:70. https://doi.org/10.5021/ad.2019.31.1.70
20. Patel D, Jahnke MN. Serious complications from Staphylococcal aureus in atopic dermatitis. Pediatr Dermatol 2015;32:792–6. https://doi.org/10.1111/pde.12665
21. Tsuboi I, Yumoto T, Toyokawa T et al. Staphylococcus aureus bacteremia complicated by psoas abscess and infective endocarditis in a patient with atopic dermatitis. Case Rep Infect Dis 2017;2017:4920182. https://doi.org/10.1155/2017/4920182
22. Woodun H, Bouayyad S, Sahib S, Elamin N, Hunter S, Al-Mohammad A. Tricuspid valve infective endocarditis in a non-IVDU patient with atopic dermatitis. Oxf Med Case Reports 2020;2020:omaa045. https://doi.org/10.1093/omcr/omaa045
23. Micallef MJ, Ramphul A. Infective endocarditis in a patient with atopic dermatitis. J Cardiol Cases 2016;13:153–4. https://doi.org/10.1016/j.jccase.2016.01.006
24. Kitagata Y, Tsuneyoshi H, Ito K et al. Surgical treatment of infective endocarditis with massive vegetations in a patient with a ventricular septal defect and atopic dermatitis: a case report. Gen Thorac Cardiovasc Surg Cases 2023;2:43. https://doi.org/10.1186/s44215-023-00056-z
25. Tamura Y, Abe T. Infective endocarditis associated with atopic dermatitis. Clin Case Rep 2023;11:e8321. https://doi.org/10.1002/ccr3.8321
26. Fukunaga N, Okada Y, Konishi Y, Murashita T, Koyama T. Pay attention to valvular disease in the presence of atopic dermatitis. Circ J 2013;77:1862–6. https://doi.org/10.1253/circj.CJ-12-1371
27. Buckley DA. Staphylococcus aureus endocarditis as a complication of acupuncture for eczema. Br J Dermatol 2011;164:1405–06. https://doi.org/10.1111/j.1365-2133.2011.10276.x
28. Okada M, Nakazato Y, Yamaguchi T, Tominaga E, Yamamoto T. Infectious endocarditis with subarachnoid hemorrhage and cerebral infarction in an atopic dermatitis patient during unsanitary cupping therapy. Cureus 2024;16:e71599. https://doi.org/10.7759/cureus.71599
29. Furumura Y, Nishida M, Imanishi A, Maekawa N, Fukai K. Infectious endocarditis with multiple cerebral infarctions in a patient with severe atopic dermatitis. J Dermatol 2019;46:e353–e354. https://doi.org/10.1111/1346-8138.14924
30. Hibi A, Ito C. Infective endocarditis due to methicillin-sensitive Staphylococcus aureus in a patient with untreated atopic dermatitis who was successfully treated without surgery. Oxf Med Case Reports 2018;2018:omx113. https://doi.org/10.1093/omcr/omx113
31. Aoyagi S, Oda T, Wada K, Nakamura E, Kosuga T, Yasunaga H. Infective endocarditis associated with atopic dermatitis. Int Heart J 2018;59:420–3. https://doi.org/10.1536/ihj.17-078
32. Horimoto K, Kubo T, Matsusaka H, Baba H, Umesue M. Right-sided infective endocarditis with a ruptured sinus of Valsalva and multiple septic pulmonary emboli in a patient with atopic dermatitis. Intern Med 2015;54:797–800. https://doi.org/10.2169/internalmedicine.54.3374
33. Fowler VG, Durack DT, Selton-Suty C et al. The 2023 Duke-International Society for Cardiovascular Infectious Diseases criteria for infective endocarditis: updating the modified Duke criteria. Clin Infect Dis 2023;77:518–26. https://doi.org/10.1093/cid/ciad271
34. Rajani R, Klein JL. Infective endocarditis: a contemporary update. Clin Med (Lond) 2020;20:31–5. https://doi.org/10.7861/clinmed.cme.20.1.1
35. Murad MH, Asi N, Alsawas M, Alahdab F. New evidence pyramid. Evid Based Med 2016;21:125–7. https://doi.org/10.1136/ebmed-2016-110401
36. Nakayama K, Osaka W, Togari T, Ishikawa H, Yonekura Y, Sekido A, Matsumoto M. Comprehensive health literacy in Japan is lower than in Europe: a validated Japanese-language assessment of health literacy. BMC Public Health 2015;15:505. https://doi.org/10.1186/s12889-015-1835-x
37. Kamei K, Hirose T, Yoshii N, Tanaka A. Burden of illness, medication adherence, and unmet medical needs in Japanese patients with atopic dermatitis: a retrospective analysis of a cross-sectional questionnaire survey. J Dermatol 2021;48:1491–8. https://doi.org/10.1111/1346-8138.16054
38. Baron ED, Barzilai D, Johnston G et al. Atopic dermatitis management: comparing the treatment patterns of dermatologists in Japan, U.S.A. and U.K. Br J Dermatol 2002;147:710–15. https://doi.org/10.1046/j.1365-2133.2002.04895.x
39. Lewer D, Hope VD, Harris M et al. Incidence and treatment costs of severe bacterial infections among people who inject heroin: a cohort study in South London, England. Drug Alcohol Depend 2020;212:108057. https://doi.org/10.1016/j.drugalcdep.2020.108057

