Metformin is one of the oldest oral treatments to reduce hyperglycaemia in people with diabetes. Gastrointestinal side effects are common, and metformin should be used with caution in patients with renal impairment because of the slight risk of lactic acidosis. In the United Kingdom Prospective Diabetes Study (UKPDS) patients treated with metformin had a significant reduction in myocardial infarction and mortality that was not demonstrated in patients treated with sulphonylureas or insulin. The fact that metformin significantly reduces cardiovascular events plus reduces weight has meant that metformin is the drug of first choice in guidelines for the treatment of type 2 diabetes. There are no longer concerns about using metformin in patients with chronic heart failure, other than in patients with associated renal failure, or during episodes of acute left ventricular failure when metformin should be temporarily stopped.
 Introduction
Introduction
The prevalence of type 2 diabetes mellitus (T2DM) is reaching epidemic proportions. Metformin therapy has been used in the treatment of T2DM for many years. Despite an increased armoury of agents for treating hyperglycaemia, only metformin has been shown to improve prognosis as a primary end point in a randomised-controlled trial.1 Clinical studies have shown that the effects of metformin go beyond improving glycosylated haemoglobin (HbA1c) and include reductions in cardiovascular end points. Despite the evidence base for the benefits of metformin, concerns remain about its side effects, including the perceived risk of lactic acidosis, particularly in the presence of cardiac and renal failure. Evidence confirming its safety, even in the setting of cardiac and renal failure, has resulted in metformin being accepted globally as the first-line treatment for overweight patients (body mass index [BMI] >25 kg/m2) with T2DM.
Pharmacology
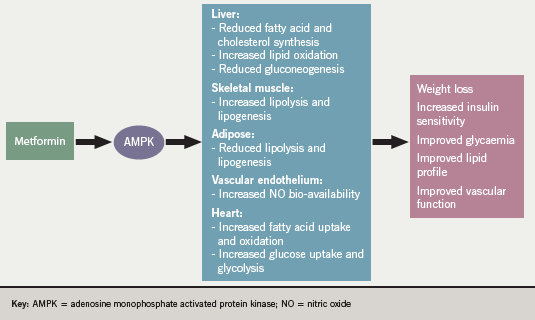
The proposed mechanisms of action of metformin, which are not fully understood, are illustrated in figure 1. It can lower blood glucose in several ways. It acts by countering insulin resistance, particularly in liver and skeletal muscle. It suppresses hepatic gluconeogenesis, increases peripheral insulin sensitivity in insulin sensitive tissues such as muscle and adipose tissue, and enhances peripheral glucose utilisation. The protective effect on the cardiovascular system cannot be fully explained by its blood glucose-lowering properties. These effects may be partly mediated via beneficial effects on circulating markers of endothelial function (vascular cell adhesion molecule-1 [VCAM-1], E-selectin), fibrinolysis (plasminogen activator inhibitor-1 [PAI-1]) and chronic inflammation (C-reactive protein [CRP]). There are several other potential mechanisms postulated to explain the cardiovascular benefits beyond blood glucose lowering, including disruption of respiratory chain oxidation in mitochondria and activation of the enzyme adenosine monophosphate (AMP)-activated protein kinase (AMPK).2,3 AMPK is a protein kinase ubiquitously expressed in mammalian tissues and involved in regulating energy balance. Activation of AMPK stimulates adenosine triphosphate (ATP)-producing catabolic pathways, while inhibiting ATP-consuming anabolic pathways, thereby, maintaining cellular energy stores. In skeletal muscle, activation of AMPK increases glucose uptake and lipid oxidation. In liver, activation of AMPK inhibits gluconeogenesis and lipid synthesis but increases lipid oxidation. Finally, in adipose tissue, activation of AMPK reduces both lipolysis and lipogenesis. Therefore, activation of AMPK in skeletal muscle, liver and adipose tissue results in decreased circulating glucose, lipids and ectopic fat accumulation, as well as enhanced insulin sensitivity.
Metformin has a short half-life of around six hours and undergoes renal excretion with 90% being eliminated within 24 hours. It can be prescribed as 500 mg or 850 mg tablets. It should be started at the 500 mg dose and increased in weekly increments until the maximum tolerated dose is achieved, normally 2 g/day. It should be taken with food to reduce the potential for gastrointestinal side effects. Occasionally higher doses are given depending on clinical response and tolerability. Hypoglycaemia is not usually a side effect of metformin therapy and it tends not to result in significant weight gain. The main side effect of concern is its association with lactic acidosis particularly in the setting of renal and cardiac failure.
Metformin is licensed to be given on its own in patients who have failed to improve with dietary modifications or it can be given in combination with sulphonylureas, thiazolidinediones, repaglinide/nateglinide, dipeptidyl peptidase-4 (DPP4) inhibitors (sitipliptin, vildagliptin, saxagliptin), glucagon-like peptide-1 (GLP-1) agonists (exenatide or liraglutide), acarbose or insulin. Indeed, given that the benefits of metformin are considered to go beyond glycaemic control, all overweight patients with T2DM should be on metformin, if tolerated. There is limited evidence to support the use of a slow-release preparation of metformin (Glucophage SR®) on the basis of fewer gastrointestinal side effects.
Trials of safety and efficacy
Evidence for improved glycaemic control
The United Kingdom Prospective Diabetes Study (UKPDS) study, initiated in 1977 and reported in 1998, demonstrated that improved glycaemic control was associated with a reduced incidence of microvascular complications (such as nephropathy and retinopathy).1 While no significant effect of lowering blood glucose was observed on cardiovascular complications, epidemiological analysis of the UKPDS data has demonstrated a continuous association between the risk of cardiovascular complications and glycaemic control. Metformin has been shown to improve glycaemic control compared with placebo or diet. When compared with placebo, metformin shows improvement in HbA1c and fasting plasma glucose, but there are no significant differences for BMI or weight, total cholesterol, high-density lipoprotein (HDL)-cholesterol, low-density lipoprotein (LDL)-cholesterol, triglycerides, or blood pressure.4 When compared with diet, metformin shows improvement in HbA1c and total cholesterol, but no difference for fasting plasma glucose, BMI or weight, HDL-cholesterol, LDL-cholesterol, triglycerides, or blood pressure. Metformin and sulphonylureas have similar effects on HbA1c, and there is no significant difference in HbA1c between those using metformin and those using insulin. UKPDS was the first randomised trial to demonstrate improved outcome with metformin treatment. The UKPDS trial allocated patients to either conventional (initial dietary modification with addition of a sulphonylurea for fasting plasma glucose >15 mmol/L) or a more intensive glycaemic control strategy (which could include metformin, sulphonylurea or insulin therapy).
Evidence for cardiovascular benefit
Within the UKPDS cohort there was a substudy looking at overweight patients (54% with obesity). Those allocated to metformin (n=342) had improved outcomes compared with those on conventional treatment (n=411). Randomisation to metformin was associated with dramatic relative risk reductions in diabetes-related death (–42%), myocardial infarction (–39%), stroke (–42%) any diabetes-related end point (–32%) and all-cause mortality (–36%) when compared with diet. This is in contrast to the relative risk reductions with sulphonylurea or insulin in diabetes-related death (–20%), myocardial infarction (–21%), stroke (+14%) any diabetes-related end point (–7%) and all-cause mortality (–8%) when compared with diet. There were no significant differences between metformin and other comparison arms for other outcomes such as stroke, peripheral arterial disease and microvascular disease. Mean HbA1c after follow-up in the diet group and metformin group were 8% and 7.4%, respectively, and there was no clear difference in the glucose-lowering effect between metformin, sulphonylurea and insulin.
Given that the glucose-lowering effects of metformin, sulphonylurea and insulin were similar, it has been proposed that metformin must confer additional vascular benefits beyond those of glycaemia alone. The UKPDS Post-Trial monitoring results suggest a legacy effect with continuing benefit of metformin therapy. There is maintenance of the relative risk reductions for any diabetes-related end point (–21%), myocardial infarction (–33%) and all-cause mortality (–27%), despite loss of within-trial blood glucose and therapy differences.5
Metformin-associated lactic acidosis
Although gastrointestinal upset is the most frequently reported adverse event with metformin therapy, concerns surround the risk of lactic acidosis with metformin therapy, especially in those patients with renal and cardiac failure. Lactic acidosis associated with metformin is a rare condition with an estimated prevalence of one to five cases per 100,000. Lactic acidosis associated with metformin has been traditionally thought to be secondary to accumulation, but the evidence for this is lacking. No patient in the UKPDS study developed lactic acidosis.
A systematic review of the risk of lactic acidosis with metformin found no cases of fatal or non-fatal lactic acidosis in 274 comparative trials and cohort studies, and estimated that the upper limit of the true incidence of lactic acidosis per 100,000 patient years was 5.1 with metformin and 5.8 without.6 In Scotland, analysis of a population-based study revealed that 24.5% of 1,847 patients treated with metformin had a documented contraindication. Despite 4,600 patient years of therapy, only one case of lactic acidosis was reported in a patient with myocardial infarction complicated by acute renal failure.7
Indeed, metformin does not affect lactate concentrations in patients with T2DM, and there is no difference in lactate levels for metformin compared with other treatments.8 Although 90% is renally excreted, accumulation is rare in the absence of moderate-to-severe renal impairment. Most cases of metformin-associated lactic acidosis documented in the literature happen during periods of tissue hypoxia such as myocardial infarction, acute left ventricular failure or septicaemia. Accumulation of metformin does not correlate with lactate concentrations or mortality. Given that mortality is predicted by the severity of underlying tissue hypoxia, metformin should always be discontinued in such a clinical scenario.
Although there is evidence to support insulin for the treatment of hyperglycaemia after myocardial infarction, there is no reason why metformin therapy could not be reinstated after stabilisation. While there are no randomised-controlled trials comparing metformin with placebo in patients with heart failure and diabetes, observational studies suggest that metformin is associated with a reduction in all-cause mortality when compared with placebo.9 This is reinforced by evidence that there is a significant improvement in outcome in heart failure patients allocated metformin compared with sulphonylurea therapy. Metformin has also been associated with a reduction in re-admission due to heart failure in observational studies.9 Furthermore, the withdrawal of metformin in stable chronic heart failure may have an adverse effect on glycaemic control. This evidence supports the view that metformin is no longer contraindicated in patients with chronic stable heart failure and diabetes.
The risk of metformin-related lactic acidosis in patients undergoing cardiac catheterisation has yet to be determined and is heavily influenced by the baseline creatinine and the amount of contrast used. Guidelines suggest that metformin should not be prescribed in patients with a serum creatinine of >150 µmol/L and/or estimated glomerular filtration rate (eGFR) <30 ml/min.10 For patients undergoing coronary angiography/percutaneous coronary intervention (PCI) or any radiological investigation involving the use of contrast, metformin should be withdrawn for 24 hours before and 48 hours after contrast medium containing iodine has been given, and treatment should be restarted after renal function has been checked.
Discussion
Clinical studies in T2DM have shown that the effects of metformin go beyond improving HbA1c and include reductions in cardiovascular end points. Metformin is considered to be the first drug of choice for the treatment of T2DM in overweight patients. There is increasing evidence to show that in the absence of tissue hypoxia and/or moderate-to-severe renal impairment, metformin will not result in lactic acidosis. Importantly, metformin should no longer be contraindicated in patients with chronic stable heart failure and may even have morbidity and mortality benefits. There is a clear need for prospective randomised-controlled studies of metformin therapy in patients with T2DM and the traditional contraindications of heart and renal failure. In the meantime, a pragmatic approach is recommended including temporarily stopping metformin during an acute illness where tissue hypoxia is suspected, which will include patients with an acute myocardial infarction, acute left ventricular failure or septicaemia, or when an individual is due to get radiological investigations using iodine-based contrast medium. Particular care should be taken in patients with chronic renal failure with a lower clinical threshold to temporarily discontinue in the aforementioned circumstances. However, chronic renal failure in itself is not an absolute contraindication to metformin use when the decision to prescribe or not is based on the potential prognostic benefits weighed against the likelihood of harm.
Conflict of interest
JGB and GAM: none declared. MF has received speaker’s honoraria from Merck Serono.
Editors’ note
This is the first article in a new series looking at the effects of drugs used in the treatment of diabetes discussed from a cardiology perspective. The next article in the series will look at sulphonylureas.
Key messages
- Stop metformin if serum concentration of creatinine is higher than 150 µmol/L and/or estimated glomerular filtration rate (eGFR) <30 ml/min
- Withdraw metformin during periods of suspected tissue hypoxia, such as myocardial infarction, acute left ventricular failure or septicaemia, and reinstate when clinically stable. (Have lower clinical threshold to stop completely if eGFR 30–45 ml/min)
- There is no absolute contraindication to the use of metformin in chronic stable heart failure
- For coronary angiography/percutaneous coronary intervention (PCI) or other procedures using iodine contrast, withdraw metformin for 24 hours before and 48 hours after contrast medium containing iodine has been given. Start treatment with metformin only after renal function has been checked
References
1. UK Prospective Diabetes Study (UKPDS) Group. Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). Lancet 1998;352:854–65.
2. Owen MR, Doran E, Halestrap AP. Evidence that metformin exerts its anti-diabetic effects through inhibition of complex 1 of the mitochondrial respiratory chain. Biochem J 2000;348:607–14.
3. Zhou G, Myers R, Li Y et al. Role of AMP-activated protein kinase in mechanism of metformin action. J Clin Invest 2001;108:1167–74.
4. Saenz A, Fernandez-Esteban I, Mataix A, Ausejo M, Roque M, Moher D. Metformin monotherapy for type 2 diabetes mellitus. Cochrane Database Syst Rev 2005;(3):CD002966.
5. Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HA. 10-year follow-up of intensive glucose control in type 2 diabetes (UKPDS 80). N Engl J Med 2008;359:1577–89.
6. Salpeter S, Greyber E, Pasternak G, Salpeter E. Risk of fatal and nonfatal lactic acidosis with metformin use in type 2 diabetes mellitus. Cochrane Database Syst Rev 2006;(1):CD002967.
7. Emslie-Smith AM, Boyle DI, Evans JM, Sullivan F, Morris AD. Contraindications to metformin therapy in patients with type 2 diabetes—a population-based study of adherence to prescribing guidelines. Diabet Med 2001;18:483–8.
8. Cusi K, Consoli A, DeFronzo RA. Metabolic effects of metformin on glucose and lactate metabolism in non insulin-dependent diabetes mellitus. J Clin Endocrinol Metabol 1996;81:4059–67.
9. Eurich DT, McAlister FA, Blackburn DF et al. Benefits and harms of antidiabetic agents in patients with diabetes and heart failure: systematic review. BMJ 2007;335:497–501.
10. National Institute for Health and Clinical Excellence. Type 2 diabetes: the management of type 2 diabetes (update). NICE Guideline CG66. London: NICE, 2008. Available from: http://www.nice.org.uk

