Up to one-third of patients with heart disease have diabetes. Cardiological status should be considered when deciding on treatment for diabetes. Patients with stable coronary disease can be treated with metformin, sulphonylureas or pioglitazone. Following an acute coronary syndrome, intensive insulin therapy with multi-dose insulin has been shown to reduce mortality, and longerterm treatment with pioglitazone may reduce recurrent events. There is little trial information for glycaemia control in patients with chronic heart failure, and metformin and insulin are both frequently used. Dipeptidylpeptidase-4 (DPP-4) inhibitors are new oral antidiabetic drugs, which are weight neutral, and the injected glucagon-like peptide-1 (GLP-1) receptor agonists reduce weight. Long-term outcome studies are awaited to see if they have cardiovascular advantages in any particular group of patients.
Introduction
 The incidence of diabetes is rising rapidly and patients with diabetes have a significantly increased risk of developing cardiovascular disease. The approach to the treatment of hyperglycaemia in patients with type 2 diabetes is evolving with the introduction of new medications. Intensive glycaemic control is desirable to limit microvascular complications,1 but, in those with established diabetes, aggressive blood-glucose lowering may be detrimental.2 The oral antidiabetic drug, rosiglitazone, has recently been removed from the European market due to its perceived lack of cardiovascular safety. In 2008, the Food and Drug Administration (FDA) issued recommendations that all new drugs for diabetes should provide data regarding cardiovascular safety, in addition to glycaemic control, prior to approval. It is important to consider both the impact on glycaemic control and cardiovascular risk when prescribing drugs for diabetes.
The incidence of diabetes is rising rapidly and patients with diabetes have a significantly increased risk of developing cardiovascular disease. The approach to the treatment of hyperglycaemia in patients with type 2 diabetes is evolving with the introduction of new medications. Intensive glycaemic control is desirable to limit microvascular complications,1 but, in those with established diabetes, aggressive blood-glucose lowering may be detrimental.2 The oral antidiabetic drug, rosiglitazone, has recently been removed from the European market due to its perceived lack of cardiovascular safety. In 2008, the Food and Drug Administration (FDA) issued recommendations that all new drugs for diabetes should provide data regarding cardiovascular safety, in addition to glycaemic control, prior to approval. It is important to consider both the impact on glycaemic control and cardiovascular risk when prescribing drugs for diabetes.
Stable coronary heart disease
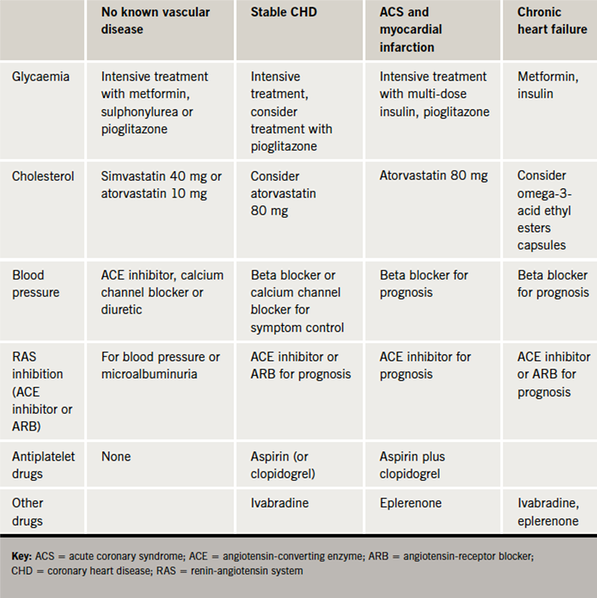
The approach to diabetes management in patients with stable, established coronary heart disease is similar to those with no cardiac history. The cardiovascular risk associated with diabetes means that use of statins and angiotensin-converting enzyme (ACE) inhibitors is standard above the age of 40 years (table 1). When considering glycaemic control, a glycosylated haemoglobin (HbA1c) target of 7% or 53 mmol/mol is generally accepted in the UK.3
Type 1 diabetes
Type 1 diabetes requires exogenous insulin treatment and metformin is used in some cases as an insulin-sensitising agent. Insulin can be delivered as a twice-daily fixed mixture or with multiple-daily injections (MDI) of basal and prandial insulin or even as a continuous subcutaneous insulin infusion (CSII). The newer insulin analogues allow people with type 1 diabetes improved glycaemic control with less hypoglycaemia but have not shown prognostic benefit in type 2 diabetes.4
The Diabetes Control and Complications Trial (DCCT) examined the impact of intensive glycaemic control in patients with type 1 diabetes for less than 10 years on microvascular and macrovascular events over an average of 6.5 years, compared with standard care.1 The Epidemiology of Diabetes Interventions and Complications (EDIC) trial followed this group for a further 10 years. The intensive group had a 42% reduction in any cardiovascular event and a 57% reduction in nonfatal myocardial infarction (MI), stroke or cardiovascular death.5 These benefits were retained despite returning to standard care after DCCT and have been referred to as ‘metabolic memory’ or ‘legacy effect’.
Type 2 diabetes
For patients with type 2 diabetes, treatment requires more consideration due to the increasing choice of medications available and the recent conflicting evidence regarding intensity of diabetes control. Additionally, there is now significant focus on cardiovascular risk management as well as glycaemic control (table 1).
The UK Prospective Diabetes Study (UKPDS) and its 10-year follow-up showed that early intensive glycaemic control, this time in type 2 diabetes, would also lead to later cardiovascular benefit or ‘legacy effect’ despite relaxation of HbA1c target outside of the study.6 Patients with coronary heart disease were excluded from the UKPDS at baseline, but on follow-up MI was the most common cause of death.
The Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial set out to assess whether intensive glycaemic control in patients with established type 2 diabetes, cardiovascular disease, or high cardiovascular risk would provide cardiovascular benefit. ACCORD was terminated prematurely due to increased mortality in the intensive control group.2 This trial has resulted in considerable debate because of this unexpected outcome. In drawing conclusions there are a few points to consider: there was widespread use of the thiazolidinedione rosiglitazone, which is no longer available on the European market due to cardiovascular risk, the intensive group were very aggressively treated and HbA1c lowered over a short time period with a target of 6% or 42 mmol/mol, and the people within the intensive group who had higher mortality were those who did not achieve target.7 Another large trial assessing intensive glycaemic control in a similar group of patients, the Action in Diabetes and Vascular disease: preterAx and diamicroN mr Controlled Evaluation (ADVANCE) trial, demonstrated benefit with intensive control on a composite microvascular and macrovascular end point.8 In this trial, the target HbA1c was 6.5% or 48 mmol/mol and the time allowed to achieve this was more relaxed. This approach may be more acceptable for patients with greater than 10 years of type 2 diabetes.
Management of type 2 diabetes must be holistic and includes education, lifestyle intervention and medication. There are now several newer antidiabetic drugs available, in addition to the well-established metformin, sulphonylureas and insulin. The newer agents include oral dipeptidylpeptidase-4 (DPP-4) inhibitors and injected glucagon-like peptide-1 (GLP-1) agonists.
Metformin is the first-choice medication in type 2 diabetes.9 It improves glycaemic control by increasing insulin sensitivity and reducing gluconeogenesis in the liver.10 Data from the UKPDS showed metformin to have similar effect on glycaemia to sulphonylurea and insulin, but it had a greater risk reduction for diabetes-related death and MI in overweight subjects.
Sulphonylureas, which stimulate pancreatic insulin secretion, and insulin are associated with hypoglycaemia and weight gain, both are side effects that can increase cardiac stress in diabetes.4,11 In UKPDS, the sulphonylurea gliclazide was found to have a non-significant risk reduction for MI. Insulin is reserved for third-line treatment in type 2 diabetes and when introduced should be a once-daily long-acting dose.
Glitazones
In 2010, rosiglitazone lost its European recommendation and the FDA significantly restricted its use in the USA. This was in response to data from a large meta-analysis which found that rosiglitazone was associated with a significant increase in MI.12 There are some conflicting data available as the Rosiglitazone Evaluated for Cardiovascular Outcomes in Oral Agent Combination Therapy for Type 2 Diabetes (RECORD) trial found an increased risk of heart failure with rosiglitazone but not an association with cardiovascular death, and the trial was inconclusive regarding MI.13
Pioglitazone, the other thiazolidinedione, acts on peroxisome proliferator-activated receptor-gamma (PPARγ) receptors to increase insulin sensitivity;14 it has had somewhat more favourable cardiovascular outcomes. The Prospective Pioglitazone Clinical Trial in Macrovascular Events (PROactive) sought to show cardiovascular benefit from pioglitazone. It did not meet its primary end point, but a main secondary end point of all-cause mortality, stroke, and MI was significantly reduced and subgroup analysis found benefit in high-risk patients who had previous MI.15,16 A common side effect of pioglitazone is fluid retention, and this may cause heart failure in those at risk, subgroup analysis did not show any associated increase in mortality.17
The FDA mandated a trial comparing rosiglitazone and pioglitazone on cardiovascular outcomes. The Thiazolidinedione Intervention with Vitamin D Evaluation (TIDE) trial had just started recruiting patients when it was stopped prematurely by the FDA: 16,000 patients were to be randomised using a 3 x 2 design (the other part of the study comparing Vitamin D and placebo). Only 1,332 subjects were in the study when it was terminated by the FDA because of ongoing concerns about the cardiovascular safety of rosiglitazone, so no conclusions can be made either about rosiglitazone compared with pioglitazone, or about possible benefits of vitamin D on cardiovascular outcomes in patients with diabetes.
Incretin-based therapies
The DPP-4 inhibitors and GLP-1 receptor agonists are both classes of drug working on the incretin system, which amplifies insulin secretion in response to oral carbohydrate intake.18,19 They are attractive treatment options for type 2 diabetes given that they have low risk of hypoglycaemia, similar effect on HbA1c and do not cause weight gain.
DPP-4 inhibitors, such as sitagliptin, vildagliptin and saxagliptin, were found to non-significantly reduce cardiovascular events in a recent meta-analysis.20 Several large randomised-controlled trials of cardiovascular safety are ongoing, including one designed to show non-inferiority of sitagliptin versus placebo and one to show superiority of saxagliptin over placebo.
GLP-1 receptor agonists, such as exenatide and liraglutide, require subcutaneous injection and commonly cause weight loss. Additional potential cardiovascular benefits exist as they have been shown to reduce blood pressure independently of weight loss, and may have other direct effects upon the cardiovascular system.21,22
Acute coronary syndromes
The Diabetes and Insulin-Glucose Infusion in Acute Myocardial Infarction (DIGAMI) study was a trial of intensive insulin following acute MI in patients with diabetes, which found a prognostic benefit of acute insulin-glucose infusion followed by at least three months of subcutaneous insulin.23 DIGAMI-2 was set up to establish whether the benefit came from the acute intravenous insulin or the three months of intensive subcutaneous insulin by having three trial groups. Unfortunately, no significant difference was demonstrated between the patients receiving routine management, those with acute insulin or those with three months of intensive insulin treatment, but trial numbers were small and by the end most of the patients in each group were on the same intensity of treatment.24
Recent National Institute for Health and Clinical Excellence (NICE) guidance has been published on the management of hyperglycaemia in acute coronary syndrome.25 Hyperglycaemia is common in patients who do not have a diagnosis of diabetes and is a risk factor for future development of diabetes. NICE advise that hyperglycaemia should be avoided with blood glucose target of less than 11 mmol/L for the 48-hour period following an acute event. The method for this glycaemic control is patient dependent, but a dose-adjusted insulin infusion should be considered with regular blood glucose monitoring and avoidance of hypoglycaemia.
Following the first 48 hours of care, the choice of antidiabetic drugs will depend on the clinical picture and pre-existing diabetes control. Patients with type 1 diabetes and poor control may have increased interest in intensifying their insulin, perhaps from twice-daily mixed insulin to multiple-daily injections with the option of carbohydrate counting. There will be a group of patients with type 2 diabetes who require insulin following acute coronary syndrome to achieve glycaemic control, but many will go back to their usual regimen.
NICE advises that patients with hyperglycaemia but no known diabetes should have HbA1c checked as an inpatient, then a fasting glucose following discharge (more than four days after event). Lifestyle advice should be given and warning about future risk of diabetes, which should be screened for annually.
Patients known to have diabetes will require a review of their medication. For those on metformin there is concern regarding the potential risk of lactic acidosis when given at times of tissue hypoxia, but there is no conclusive evidence to suggest metformin should not be used following MI.26 If metformin is the sole agent, it is also important to consider the glycaemic impact of removal peri-angiography when the contrast load in combination with metformin may potentially lead to renal impairment and lactic acidosis.
Other medications used in type 2 diabetes may require adjustment. Gliclazide and other sulphonylureas have not been associated with either increased or decreased mortality post-MI.27 Pioglitazone may reduce rates of recurrent MI but may also precipitate cardiac failure.16,17 There are no available data regarding GLP-1 agonists and DPP-4 inhibitors in patients following acute coronary syndromes.
Chronic heart failure
People with diabetes have a higher incidence of chronic heart failure and are more likely to develop heart failure at a younger age.28 Poor glycaemic control, blood pressure and obesity are modifiable factors that contribute to risk of cardiovascular disease. It may be opportune to consider glycaemic control when managing patients with heart failure, many of whom will have co-existent renal failure.
Concerns about the use of metformin in heart failure exist due to the potential to develop lactic acidosis in hypoperfused states, and it is common practice to discontinue metformin during episodes of acute heart failure. There are no randomised-controlled trials looking at metformin in heart failure, but observational data suggest a decrease in mortality and lower heart failure re-admission rates when compared with placebo.29 In a retrospective cohort study of more than 5,000 people starting medication for diabetes, high-dose sulphonylurea was found to produce a higher risk of developing heart failure than metformin or low-dose sulphonylurea.30
Thiazolidinediones are the main class of diabetes drug to be avoided in patients with cardiac failure due to side effects including fluid retention.14 A meta-analysis of thiazolidinediones found a relative risk of 1.72 for heart failure with greater risk associated with rosiglitazone than pioglitazone.31 Pioglitazone is associated with increased symptomatic heart failure, but, interestingly, not an increase in mortality.17
GLP-1 agonists have some promising data from animal studies suggesting an ability to improve left ventricular function. The addition of modest blood-pressure-lowering effects potentially elevates the desirability of these agents over insulin as an injectable therapy, although more studies are required.
Despite its associated weight gain, there is no direct evidence of insulin worsening heart failure in the majority of patients. Data from studies using acute insulin in cardiac patients such as DIGAMI, DIGAMI-2 and the Hyperglycaemia: Intensive Insulin Infusion in Infarction (HI-5) trial show either no increase in heart failure or a reduction in heart failure.23,24,32 Insulin is also likely to be used in type 2 diabetes in the presence of severe insulin resistance, which itself is associated with left ventricular dysfunction.33 There are a few case reports of individuals who had marked sodium and water retention when starting insulin therapy, but this is very rare.
Conclusion
Cardiovascular disease is arguably the most important complication of diabetes and is an important outcome in clinical trials as new drugs for diabetes must now prove their cardiovascular safety prior to approval. Many of the well-established diabetes medications do not have the benefit of strong cardiovascular safety data, rather they demonstrate the ability to control diabetes with the surrogate glycaemic marker HbA1c. Recent large trials assessing the impact of glycaemic control on cardiovascular risk highlight the importance of individualised care and addressing other modifiable cardiovascular risk factors in order to gain long-term benefit.
Conflict of interest
MF has served on advisory boards for AstraZeneca/BMS, Boehringer Ingelheim, Eli Lilly, GlaxoSmithKline, Merck Serono, MSD, Novartis, Novo Nordisk, Roche, Sanofi Aventis and Takeda. GAMcK has served on advisory boards for Boehringer Ingelheim and Eli Lilly. AW: none declared
Key messages
- Patients with diabetes and stable coronary disease should be treated to a target HbA1c of 7.0% (53 mmol/mol) avoiding hypoglycaemia and weight gain; pioglitazone may be useful but causes weight gain. Longer-term safety data for incretin-based therapies is awaited
- Following acute coronary syndromes, patients should be treated with intravenous insulin followed by multi-dose subcutaneous insulin for at least three months
- Glycaemic control in patients with diabetes and chronic heart failure requires further investigation; metformin can be used in stable patients
References
- The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications of insulin-dependent diabetes mellitus. N Engl J Med 1993;329:977–86. http://dx.doi.org/10.1056/NEJM199309303291401
- The Action to Control Cardiovascular Risk in Diabetes (ACCORD) Study Group. Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med 2008;358:2545–59. http://dx.doi.org/10.1056/NEJMoa0802743
- Scottish Intercollegiate Guidelines Network. Management of Diabetes. Guideline No. 116. Edinburgh:
SIGN, 2010. - Barwell N, McKay GA, Fisher M. Drugs for diabetes: part 7 insulin. Br J Cardiol 2011;18:224–8.
- The Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) Study Research Group. Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes. N Engl J Med 2005;353:2643–53. http://dx.doi.org/10.1056/NEJMoa052187
- Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HA. 10-Year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med 2008;359:1577–89. http://dx.doi.org/10.1056/NEJMoa0806470
- Riddle MC, Ambrosius WT, Brillon DJ et al.; for the ACCORD Investigators. Epidemiological relationships between A1C and all-cause mortality during a median 3.4 year follow-up of glycaemic treatment in the ACCORD trial. Diabetes Care 2010;33:983–90. http://dx.doi.org/10.2337/dc09-1278
- The ADVANCE Collaborative Group. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 2008;358:2560–72. http://dx.doi.org/10.1056/NEJMoa0802987
- National Institute for Health and Clinical Excellence. Type 2 diabetes: the management of type 2 diabetes. NICE clinical guideline 87. London: NICE, 2009.
- Boyle J, McKay G, Fisher M. Drugs for diabetes: part 1 metformin. Br J Cardiol 2010;17:231–4.
- Smith CJ, Fisher M, McKay GA. Drugs for diabetes: part 2 sulphonylureas. Br J Cardiol 2010;17:279–82.
- Nissen SE, Wolski K. Effect of rosiglitazone on the risk of myocardial infarction and death from cardiovascular causes. N Engl J Med 2007;356:2457–71. http://dx.doi.org/10.1056/NEJMoa072761
- Home PD, Pocock SJ, Beck-Neilsen H et al. Rosiglitazone evaluated for cardiovascular outcomes in oral agent combination therapy for type 2 diabetes (RECORD): a multi-centre randomised, open-label trial. Lancet 2009;373:2125–35. http://dx.doi.org/10.1016/S0140-6736(09)60953-3
- McGrane D, Fisher M, McKay GA. Drugs for diabetes: part 3 thiazolidinediones. Br J Cardiol 2011;18:24–7.
- Dormandy JA, Charbonnel B, Eckland DJ et al. Secondary prevention of macrovascular events in patients with type 2 diabetes in the PROactive Study (PROspective pioglitAzone Clinical Trial In macroVascular Events): a randomised controlled trial. Lancet 2005;366:1279–89. http://dx.doi.org/10.1016/S0140-6736(05)67528-9
- Erdmann E, Normandy JA, Charbonnel B, Massi-Benedetti M, Moules IK, Skene AM. The effect of pioglitazone on recurrent myocardial infarction in 2,445 patients with type 2 diabetes and previous myocardial infarction: results from the PROactive (PROactive 05) study. J Am Coll Cardiol 2007;49:1772–80. http://dx.doi.org/10.1016/j.jacc.2006.12.048
- Erdmann E, Charbonnel B, Wilcox RG et al. Pioglitazone use and heart failure in patients with type 2 diabetes and pre-existing cardiovascular disease: data from the PROactive (PROactive 08) study. Diabetes Care 2007;30:2773–8. http://dx.doi.org/10.2337/dc07-0717
- McDougall C, McKay GA, Fisher M. Drugs for diabetes: part 5 DPP-4 inhibitors. Br J Cardiol 2011;18:130–2.
- McDougall C, McKay GA, Fisher M. Drugs for diabetes: part 6 GLP-1 receptor agonists. Br J Cardiol 2011;18:167–9.
- Monami M, Iacomelli I, Marchionni N, Mannucci E. Dipeptidyl peptidase-4 inhibitors in type 2 diabetes: a meta-analysis of randomised controlled trials. Nutr Metab Cardiovasc Dis 2010;20:224–35. http://dx.doi.org/10.1016/j.numecd.2009.03.015
- Okerson T, Yan P, Stonehouse A et al. Effect of exenatide on systolic blood pressure in subjects with type 2 diabetes. Am J Hypertension 2010;3:334–9. http://dx.doi.org/10.1038/ajh.2009.245
- Zinman B, Gerich J, Buse JB et al. Efficacy and safety of the human glucagon-like peptide-1 analog liraglutide in combination with metformin and thiazolidinedione in patients with type 2 diabetes (LEAD-4 Met+TZD). Diabetes Care 2009;32:1224–30. http://dx.doi.org/10.2337/dc08-2124
- Malmberg K for the DIGAMI Study Group. Prospective randomised study of intensive insulin treatment on long-term survival after acute myocardial infarction in patients with diabetes mellitus. BMJ 1997;314:1512–15. http://dx.doi.org/10.1136/bmj.314.7093.1512
- Malmberg K, Ryden L, Wedel H et al. Intensive metabolic control by means of insulin in patients with acute myocardial infarction (DIGAMI 2): effects on mortality and morbidity. Eur Heart J 2005;26:650–61. http://dx.doi.org/10.1093/eurheartj/ehi199
- National Institute for Health and Clinical Excellence. Hyperglycaemia in acute coronary syndrome. NICE clinical guideline 130. London: NICE, 2011.
- Emslie-Smith AM, Boyle DI, Evans JM, Sullivan F, Morris AD. Contraindications to metformin therapy in patients with type 2 diabetes – a population-based study of adherence to prescribing guidelines. Diab Med 2001;18:483–8. http://dx.doi.org/10.1046/j.1464-5491.2001.00509.x
- Zeller M, Danchin N, Simon D et al. Impact of type of pre-admission sulphonylureas on mortality and cardiovascular outcomes in diabetic patients with acute myocardial infarction. J Clin Endocrinol Metab 2010;95:4993–5002. http://dx.doi.org/10.1210/jc.2010-0449
- Nichols GA, Gullion CM, Koro CE, Ephross SA, Brown JB. The incidence of congestive heart failure in type 2 diabetes. Diabetes Care 2004;27:1879–84. http://dx.doi.org/10.2337/diacare.27.8.1879
- Eurich DT, McAlister FA, Blackburn DF et al. Benefits and harms of antidiabetic agents in patients with diabetes and heart failure: systematic review. BMJ 2007;335:497–501. http://dx.doi.org/10.1136/bmj.39314.620174.80
- McAlister FA, Eurich DT, Majumdar SR, Johnson JA. The risk of heart failure in patients with type 2 diabetes treated with oral agent monotherapy. Eur J Heart Fail 2008;10:703–08. http://dx.doi.org/10.1016/j.ejheart.2008.05.013
- Lago RM, Singh PP, Nesto RW. Congestive heart failure and cardiovascular death in patients with prediabetes and type 2 diabetes given thiazolidinediones: a meta-analysis of randomized clinical trials. Lancet 2007;370:1129–36. http://dx.doi.org/10.1016/S0140-6736(07)61514-1
- Cheung NW, Wong VW, McLean M. The Hyperglycaemia: Intensive Insulin Infusion in Infarction (HI-5) study: a randomized controlled trial of insulin infusion therapy for myocardial infarction. Diabetes Care 2006;29:765–70. http://dx.doi.org/10.2337/diacare.29.04.06.dc05-1894
- Dinh W, Lankisch M, Nickl W et al. Insulin resistance and glycaemic abnormalities are associated with deterioration of left ventricular diastolic function: a cross-sectional study. Cardiovasc Diabetol 2010;9:63. http://dx.doi.org/10.1186/1475-2840-9-63
