With conflicting findings from studies of omega-3 fatty acids in cardiovascular disease, many healthcare professionals are uncertain of whether they show any benefit. BJC seminars are held to promote evidencebased practice and we recently convened a meeting of UK professionals working in cardiovascular disease to review the evidence for omega-3 fatty acids supplementation, as well as review some of the data relating to dietary fish oils. The panel considered how supplementation with omega-3 fatty acids might be used in the future. The meeting was sponsored with an unrestricted educational grant from Abbott Laboratories.
Background
Observations on fish consumption in general
Populations who consume large amounts of oily fish in their diet tend to have lower rates of coronary heart disease (CHD) and sudden cardiac death (SCD). Fish oils are rich in omega-3 polyunsaturated fatty acids (PUFAs), which have demonstrable cardioprotective properties. In line with these observations, extensive epidemiological data – including large meta-analyses – demonstrate clear associations between both increased fish consumption and increased omega-3 PUFA levels with a favourable cardiovascular prognosis.1-3
Most of the evidence for benefits has been observed in individuals with high tissue levels of eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), the long-chain fatty acids of the family, varying amounts of which are present in different types of fish (table 1).4 The clinical benefits seem to be most pronounced in CHD mortality and SCD, which is 50% lower in men who consume oily fish at least once a week.1-3,5,6
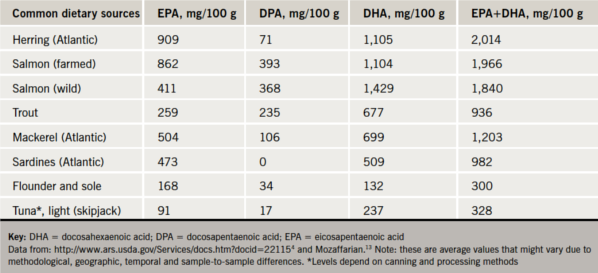
The national recommendation (for primary prevention) remains that people should eat at least two portions of fish per week, one portion of which should be oily fish (box 1).
Fish oil supplementation
For individuals who are unable to consume sufficient omega-3 fatty acids in their diet, or who wish to increase their consumption, there are several highly purified and concentrated fish oil preparations available. One omega-3 long chain PUFA preparation can be prescribed within the UK (Omacor®) for secondary prevention post-myocardial infarction (MI) at a dose of 1 g per day and as a supplement to diet for the management of hypertriglycidaemia.7
Box 1 includes a summary of some of the current guidelines for omega-3 PUFAs and shows there is no consensus on recommendations for supplementation with these agents leading to possible confusion among some healthcare professionals as to their benefit.

How do fish oils work?
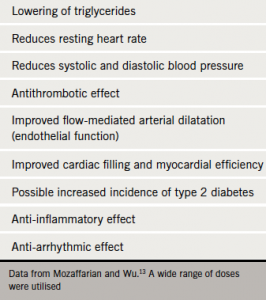
Omega-3 PUFAs are essential dietary components and have a number of biological actions. It is not known exactly how they exert their clinical benefits but they have a multitude of effects (table 2), reviewed in detail by Mozaffarian and Wu.13 These include lowering blood pressure, serum triglycerides, inflammatory markers and coagulability; reducing heart rate and increasing heart rate variability; enhancing insulin sensitivity and endothelial function; and anti-arrhythmic and plaque-stabilising effects, which have the potential to improve cardiovascular health and reduce the risk of clinical events. There is also indirect evidence that, by reducing the rate of telomere shortening, omega-3 PUFAs may slow biological ageing.
Triglyceride lowering
The well-recognised effect of omega-3 PUFAs on triglyceride lowering shows this to be a linear and dose-dependent effect, but with variable individual dose responses. Doses of at least 2 g/day are required to achieve a meaningful reduction, and greater absolute reductions are seen in those with higher baseline triglyceride levels.13
Antithrombotic effects
Omega-3 PUFAs are considered to have antithrombotic effects based on observation of increased bleeding times at very high doses (e.g. 15 g/day) but in clinical trials they have shown no consistent effects on platelet aggregation or coagulation factors.13
The panel felt that at doses of up to 4 g/day, antithrombotic effects are unlikely to be a major pathway for lower cardiovascular risk, but subtle effects cannot be excluded.13
Endothelial function
Omega-3 consumption increases nitric oxide (NO) bioavailability with several trials demonstrating improved flow-mediated arterial dilatation, a measure of improved endothelial function. Autonomic function, as a consequence of augmentation of vagal tone may also be improved.13
Heart failure
Increases in cardiac ejection fraction have been observed in heart failure patients taking omega-3 PUFAs.14,15
Insulin resistance/anti-inflammatory
The biological effects of omega-3 PUFAs on insulin resistance (marginally adverse or otherwise) are currently unclear. Similarly, it is not known whether their well-recognised anti-inflammatory effects are clinically meaningful.13
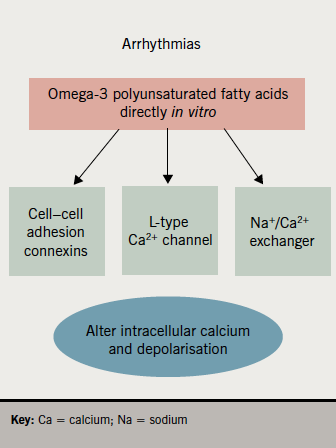
Anti-arrhythmic effects
The anti-arrhythmic activity of omega-3 PUFAs has been challenging to document in human studies.13 Animal experiments suggest that omega-3 PUFAs directly influence atrial and ventricular electrophysiology, and that, by altering the function of membrane ion channels (e.g. L-type Ca2+ channels, Na+/Ca2+ exchanger) or ‘cell–cell conexins’, they may contribute to reduced myocyte excitability, potentially reducing arrhythmia triggering (figure 1).
The panel concluded that the clinically relevant mechanisms by which omega-3 PUFAs influence arrhythmia are unknown. They noted the data showing a dose-effect with a fish-enriched diet and a reduction in arrhythmias. They agreed there was evidence of health benefits in randomised clinical trials with omega-3 PUFA supplementation, but noted many trials were not conducted on top of a background of contemporary therapy. In addition, the panel noted the use of a wide range of supplement preparations and dietary interventions which had provided variable doses of omega-3 intake, making the clinical relevance of their effects difficult to interpret, not only for patients, but also for some healthcare professionals.
What have the clinical trials of fish oils shown us?
The panel reviewed clinical studies with omega-3 PUFAs. Early promise shown in clinical studies, most notably in GISSI-P,16 has not been borne out in more recent studies, which – with the exception of the JELIS and GISSI-HF studies – have shown equivocal results.
The majority of intervention studies in those at high cardiovascular risk show a beneficial impact of omega-3 supplementation on major cardiovascular events, although heterogeneity between studies has been noted. Large-scale randomised trial data are most compelling for omega-3 supplementation in the post-MI setting.
GISSI-P
The largest of these, the GISSI-P (GISSI-Prevention) study (n=11,324 randomised to omega-3 PUFAs within three months of MI),16 demonstrated relative risk reductions in overall mortality, cardiac mortality and SCD of 20%, 30% and 45%, respectively, with 1 g/day of highly purified omega-3 acid ethyl esters (Omacor®) over a 3.5-year period. Absolute risk reductions over the same period were 2.1%, 2% and 1.6% for overall mortality, cardiac mortality and SCD, respectively. Significant benefits of supplementation emerged within three to four months and were most marked in those with more extensive left ventricular dysfunction. Considered together, these data suggest a reduction in ventricular arrhythmia as the likely mechanism of benefit.
JELIS
Results from JELIS (Japanese Eicosapentaenoic Acid Lipid Intervention Study),17 an open-label, blinded analysis conducted in Japan, showed that 1.8 g/day of EPA reduced combined cardiovascular events. About 36% of the subjects recruited were hypersensitive, 20% had coronary artery disease, and 15% had diabetes. Review of the individual component end points in JELIS demonstrated that the main driver of benefit was reduction in coronary instability, suggesting benefits on clinical lesion progression, destabilisation and thrombosis. No effect on the incidence of arrhythmia was noted, although such events were extremely rare in this Japanese population and the study was not powered to evaluate this end point.
OMEGA
Recommendations supporting intake of 1 g/day omega-3 PUFAs in those with established coronary artery disease, including the post-MI setting, are currently under review by NICE following publication of several recent studies. The German OMEGA (Effect of Omega 3-Fatty Acids on the Reduction of Sudden Cardiac Death After Myocardial Infarction) study randomised over 3,000 patients recruited early post-MI to 1 g Omacor® or placebo to examine the impact on SCD within the first year.18
Although the study outcome was neutral, it was substantially underpowered and the results should, therefore, be interpreted with caution. It was designed with the expectation of a 45% reduction in the relative risk of SCD after one year with omega-3 treatment as seen in GISSI-P (which had a duration of three years). But due to far lower than planned event rates (1.5% actual vs. 3.5%, which was expected based on the results from GISSI-P) and without changing the number of subjects recruited or the duration of follow-up, the study was estimated to have a power of only 45% or less to address the primary hypothesis.
Alpha Omega
Similarly, Alpha Omega (Study of Omega-3 Fatty Acids and Coronary Mortality),19 which studied omega-3 PUFA supplemented margarine in post-MI patients, was also neutral. This study was also underpowered. The study looked at relatively low-dose supplementation with either EPA+DHA or ALA (add doses), EPA+DHA+ALA or a control margarine in just under 5,000 patients who had suffered an MI an average of 2.5 years previously. The study was powered to show a 25% reduction in death due to CHD and it was reported in the online appendix that the final study power to address this end point was approximately 35%. The primary comparisons reported in the manuscript were also unusual, comparing outcomes in EPA+DHA versus ALA + placebo and ALA versus EPA+DHA + placebo rather than comparisons of the omega-3 supplement groups (alone or in combination) versus placebo alone.
GISSI-HF study
GISSI-HF (GISSI-Heart Failure),20 a large and adequately powered trial looked at omega-3 PUFAs in heart failure. Results demonstrated a statistically significant and clinically important reduction of all-cause mortality, despite the modest relative risk reduction, with 1 g Omacor® in a contemporary heart failure population.
Omega-3 PUFA supplementation showed an absolute risk reduction of 1.8% in all-cause mortality seen over two years of follow-up. It was felt that half of this benefit was due to a reduction in arrhythmic deaths with the other half due to reduction of admission for ventricular arrhythmia. There was little benefit in atherothrombotic events, such as MI and stroke. A subsequent pharmacoeconomic evaluation of the GISSI-HF data suggests that this dose of Omacor® is a cost-effective intervention in patients with heart failure.21
Cochrane meta-analysis
A Cochrane review, published in 2009,22 concluded that it was still not clear whether dietary or supplemental omega-3 PUFAs, either from fish or vegetable sources, did reduce total deaths or cardiovascular events in both patients with cardiovascular disease or those at risk of developing the disease. This conflicting evidence is reflected in the various meta-analyses on the possible outcomes of fish or omega-3 PUFA consumption and cardiovascular disease (CVD) outcomes.13 The wide range of study patient characteristics, together with the varying nature and dose of supplement, as well as trial size and quality, all contribute to the lack of clarity in this context.
The panel felt that recent post-MI studies have been inadequate in their design to elucidate the role of omega-3 PUFA supplements in a contemporary post-MI cohort. They noted recent studies in this area were often underpowered, were likely to be using suboptimal doses and may not have been conducted for long enough to show any benefit. It was reassuring, the panel felt, that the more recent JELIS and GISSI-HF studies, which were adequately powered and allowed several years of follow-up, demonstrated positive results.
What trials should be undertaken to address information ‘gaps’?
A number of trials are planned or underway that may fill some of the gaps in our understanding of intervention with omega-3 PUFAs in cardiovascular disease in contemporary populations.
Data from the ORIGIN (Outcome Reduction with an Initial Glargine Intervention) and the GISSI-R&P (Rischio & Prevenzione) studies, looking at 1 g/day Omacor® in at-risk patients with insulin resistance and high global CVD risk, respectively, should report later this year and may shed further light on the potential for additional event reduction with omega-3 PUFA supplementation in contemporary CVD prevention.
GISSI-R&P
The GISSI-R&P study, launched in 2004, is an ongoing large-scale, randomised-controlled trial conducted in Italian general practice to assess the efficacy and safety of omega-3 PUFAs in reducing cardiovascular mortality (including sudden death) and hospitalisation for cardiovascular reasons in patients at high CVD risk but with no history of MI. The secondary epidemiological aim is to assess the feasibility of adopting current guidelines in everyday clinical practice in order to optimise all available preventive strategies in people at high cardiovascular risk.
ASCEND
The randomised study ASCEND (A Study of Cardiovascular Events iN Diabetes), should provide the first reliable evidence about the effects of aspirin and of omega-3 PUFAs in type 1 and 2 diabetes. It has recruited 15,000 people with diabetes who were not known to have vascular disease. Funded by the British Heart Foundation, it is being coordinated by the University of Oxford Clinical Trial Service Unit and is scheduled to continue until 2017.
VITAL
 VITAL (VITamin D and OmegA-3 TriaL), underway at Brigham and Women’s Hospital and Harvard Medical School in Boston, Massachusetts, USA, is looking at supplementation in 20,000 men and women across the USA. It is investigating whether taking daily dietary supplements of vitamin D3 (2,000 IU) or omega-3 PUFAs reduces the risk for developing cancer, heart disease, and stroke in people who do not have a prior history of these illnesses. The trial is expected to be completed in 2016.
VITAL (VITamin D and OmegA-3 TriaL), underway at Brigham and Women’s Hospital and Harvard Medical School in Boston, Massachusetts, USA, is looking at supplementation in 20,000 men and women across the USA. It is investigating whether taking daily dietary supplements of vitamin D3 (2,000 IU) or omega-3 PUFAs reduces the risk for developing cancer, heart disease, and stroke in people who do not have a prior history of these illnesses. The trial is expected to be completed in 2016.
OPERA
OPERA (The Omega-3 Fatty Acids for the Prevention of Post-operative Atrial Fibrillation), due to complete later this year, is another ongoing large, randomised-controlled trial designed to investigate the effects of omega-3 PUFAs in the major public health challenge of atrial fibrillation.
Such large, well-designed studies in key selected patient populations will undoubtedly clarify the role of omega-3 PUFAs in the treatment and prevention of CVD.
The panel felt that due to the limitations of past research, randomised-controlled trials and studies should be revisited and the evidence re-assessed, particularly looking at subgroups of patients who may benefit from omega-3 PUFA supplementation.
Appropriate recommendations for the use of omega-3 PUFAs could only be made, they felt, from high-quality evidence in the form of meta-analysis or systematic reviews of randomised-controlled trials, or randomised-controlled trials with a low risk of bias.
How are fish oils currently used in the UK?
Since publication of the NICE guidance, the uptake of omega-3 PUFA supplementation for post-MI secondary prevention has been slow, but steady, with prescriptions increasing from 75,000 items per quarter in 2006 to approximately 180,000 per quarter in June 2011; this reflects an increase in NHS expenditure in this area from £1.7 million per quarter to over £4 million per quarter.23
Over the past two years, primary care trusts seeking to identify potential savings on the prescribing budget have begun to locally determine the role of omega-3 supplementation in practice. Some PCTs no longer recommend Omacor® for routine prescribing post-MI; although use for the management of hypertriglyceridaemia is still endorsed, more commonly as a second-line agent. A brief internet search carried out by the panel highlighted similar recommendations in many areas. This practice raised concern among the panel that sweeping general recommendations, driven primarily by prescribing cost-pressures, may lead to misinformation as well as restrict appropriate and potentially important treatment options at an individual patient level.
Prescribers and barriers to prescription

In clinical practice, it appears that the prescribing of Omacor® is inconsistent (table 3). Some cardiologists prescribe it in most patients post-MI, others use it only in selected patients (e.g. those with a low fish intake in their diet); while others do not prescribe it at all or cannot, due to local prescribing restrictions.
A number of barriers to omega-3 PUFA prescribing have been identified. Current barriers to use include:
- local guidelines prohibiting or discouraging primary care prescribing
- lack of clinical champions
- poor perception of the data in support of omega-3 supplementation, including lack of recent ‘good news’ stories
- QIPP plans to make savings on the prescribing budget, in which omega-3 supplements are seen as an ‘easy win’.
- While costs of prescribing omega-3 PUFAs in the post-MI setting may act as a disincentive to prescribing, some simple health economic modelling suggests a ‘return on investment’.
Applying the all-cause mortality end point from GISSI-P,16 which demonstrated a 2.1% average risk reduction (ARR), to a PCT population of 500,000, it can be estimated that total acute coronary syndrome (ACS) events would be circa 2,000 per annum (0.4% incidence) in this population. Assuming an uptake of Omacor® of 40% (i.e. 800 patients treated per annum), annual costs of treatment of this cohort would be £137,000 (or £478,000 over 3.5 years as per GISSI-P). Thus, the number of deaths avoided would be 17, making the cost per death avoided as £28,000. If the avoidance of second events is also taken into consideration, the cost per death avoided is even lower.
The panel felt that the current inconsistent use of omega-3 PUFAs in the UK was due to the emergence of recent equivocal evidence, due to poor trial design, and also local barriers to use. The panel thought that in clinical situations where healthcare professionals wanted to prescribe omega-3 PUFAs, there were good cost-effectiveness data to present to formulary groups to support this management strategy.
Considering the data from the GISSI-P and the GISSI-HF studies, the case for use of omega-3 supplementation in the post-MI setting would appear to be strongest for those patients with left ventricular (LV) dysfunction in terms of both clinical- and cost-effectiveness. The panel noted a recent review,24 which stated current guidelines suggest the potential value of omega-3 fatty acids supplementation in patients with coronary artery disease (CAD) or after MI, and possibly in those with heart failure.
Dietary recommendations for increased fish consumption, the panel felt, should continue to be encouraged. The panel noted that although fish intake is increasing slightly in the UK, a recent survey shows this remains low, particularly that of oily fish. During the four-day survey period, oily fish was not eaten by 64% and 77% of participants aged over 65 years, and 19–64 years, respectively, while mean consumption was well below one portion per week in all age groups. Where oily fish was consumed, mean intake per day across the entire sample was 43 g in adults over 65 years, and 45 g in 19–64 year olds.25
Key messages
- While we are uncertain how they work, there are persuasive data that omega-3 PUFAs reduce the risk of cardiac death
- Since many earlier trials were poorly designed or conducted before the availability of optimal background treatments (such as statins), re-assessment of randomised-controlled clinical trials is warranted to determine whether selected patient groups may particularly benefit from omega-3 PUFAs
- Larger studies in specific groups, such as patients with metabolic syndrome/diabetes or in heart failure post-MI, may clarify the cardiovascular benefits of prescribed omega-3 PUFAs
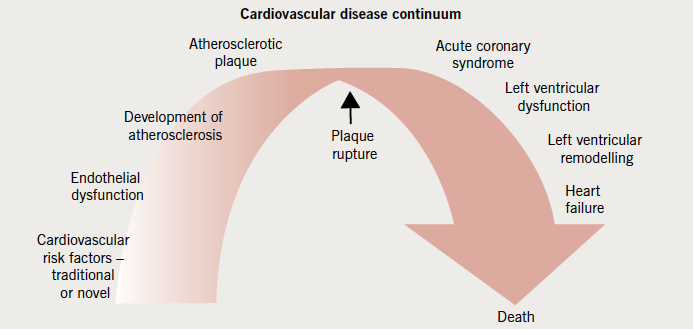
- These research ‘gaps’ along with cost issues in a tightening health economy, provide barriers to prescription of an agent, which may have many potential beneficial effects in the cardiovascular disease continuum (figure 2), that are yet to be elucidated
Conflict of interest
The meeting of this panel, honoraria and the development of this report was underwritten by an unrestricted educational grant from Abbott Laboratories Ltd. Abbott did not attend this meeting and had no input into its content or the development of this report but reviewed it for accuracy and ABPI compliance before publication.
AB has received honoraria from Abbott, AstraZeneca, BMS, Boehringer-Ingelheim, Merck, Pfizer, Roche and Servier. SC: none declared. JPH has received honoraria for advisory boards and lectures from Abbott, AstraZeneca, Boehringer-Ingelheim, BMS, Daiichi-Sankyo, Lilly, Merck, NovoNordisk, Pfizer, Roche and Sanofi-Aventis. AK has received honoraria for educational sessions from Abbott, Merck and Pfizer. LM: none declared. HP has received fees for lecturing and advisory board work from Abbott Healthcare, Merck, Menarini, Roche, Sanofi and Servier. KKR has received honoraria for advisory boards and lectures from Abbott, AstraZeneca, BMS, Kowa, Lilly, Merck, Novartis, NovoNordisk, Pfizer, Roche, Sankyo and Sanofi-Aventis. HW has received speaker honoraria and attended advisory boards for Abbott, AstraZeneca, BMS, Boehringer-Ingelheim, Daiichi-Sankyo, Pfizer, Sanofi-Aventis, Servier and Takeda. DY has received honoraria for lecturing and advisory services from Abbott Healthcare, and other cardiovascular companies including AZ, BMS, The Medicine Company, MSD & Pfizer.
References
- He K, Song Y, Daviglus ML et al. Accumulated evidence on fish consumption and coronary heart disease mortality: a meta-analysis of cohort studies. Circulation 2004;109:2705–11.
- Konig A, Bouzan C, Cohen JT et al. A quantitative analysis of fish consumption and coronary heart disease mortality. Am J Prev Med 2005;29:335–46.
- Whelton SP, He J, Whelton PK, Muntner P. Meta-analysis of observational studies on fish intake and coronary heart disease. Am J Cardiol 2004;93:1119–23.
- US Department of Agriculture. National Nutrition Database for Standard Reference, 2010. Release 23. Available from: http://www.ars.usda.gov/Services/docs.htm?docid=22115
- NHS Choices. Fish and shellfish. NHS Live Well. Available from: http://www.nhs.uk/Livewell/Goodfood/Pages/fish-shellfish.aspx
- Albert CM, Hennekens CH, O’Donnell CJ et al. Fish consumption and risk of sudden cardiac death. JAMA 1998;279:23–8.
- Electronic Medicines Compendium. Omacor (Abbott) Summary of Product Characteristics. Available from: http://www.medicines.org.uk/emc/medicine/10312/SPC/omacor [accessed 7th February 2012].
- National Institute for Health and Clinical Excellence. Secondary prevention in primary and secondary care for patients following a myocardial infarction. Clinical guideline 48. London: NICE, 2007. Available from: http://www.nice.org.uk/CG48 [accessed 7th February 2012].
- Scottish Intercollegiate Guidelines Network. Risk estimation and the prevention of cardiovascular disease. Guideline No. 97. Edinburgh: SIGN, 2007. Available from: http://www.sign.ac.uk/guidelines/fulltext/97/index.html [accessed 7th February 2007].
- van de Werf F, Bax J, Betriu A et al. Management of acute MI in patients presenting with persistent ST segment elevation MI. Eur Heart J 2008:29;2909–45. http://dx.doi.org/10.1093/eurheartj/ehn416
- Hooper L, Thompson RL, Harrison RA et al. Risks and benefits of omega 3 fats for mortality, cardiovascular disease, and cancer: systematic review. BMJ 2006;332:752. http://dx.doi.org/10.1136/bmj.38755.366331.2F
- Antman EM, Hand M, Armstrong PW et al. 2007 focused update of the ACC/AHA 2004 guidelines for the management of patients with ST-elevation myocardial infarction. Circulation 2008;117:296–329. http://dx.doi.org/10.1161/CIRCULATIONAHA.107.188209
- Mozaffarian D, Wu HY. Omega-3 fatty acids and cardiovascular disease. J Am Coll Cardiol 2011;58:2047–67. http://dx.doi.org/10.1016/j.jacc.2011.06.063
- Nodari S, Triggiani M, Campia U et al. Effects of n-3 polyunsaturated fatty acids on left ventricular function and functional capacity in patients with dilated cardiomyopathy. J Am Coll Cardiol 2011;57:870–9. http://dx.doi.org/10.1016/j.jacc.2010.11.017
- Ghio S, Scelsi L, Latini R et al. Effects of n-3 polyunsaturated fatty acids and of rosuvastatin on left ventricular function in chronic heart failure: a substudy of GISSI-HF trial. Eur J Heart Fail 2010;12:1345–53. http://dx.doi.org/10.1093/eurjhf/hfq172
- GISSI-Prevenzione Investigators. Dietary supplementation with n-3 polyunsaturated fatty acids and vitamin E after myocardial infarction: results of the GISSI-Prevenzione trial. Lancet 1999;354:447–55. http://dx.doi.org/10.1016/S0140-6736(99)07072-5
- Yokoyama M, Origasa H, Matsuzaki M et al. Effects of eicosapentaenoic acid on major coronary events in hypercholesterolaemic patients (JELIS): a randomised open-label, blinded endpoint analysis. Lancet 2007;369:1090–8. http://dx.doi.org/10.1016/S0140-6736(07)60527-3
- Rauch B, Schiele R, Schneider S et al. OMEGA, a randomized, placebo-controlled trial to test the effect of highly purified omega-3 fatty acids on top of modern guideline-adjusted therapy after myocardial infarction. Circulation 2010;122:2152–9. http://dx.doi.org/10.1161/CIRCULATIONAHA.110.948562
- Kromhout D, Giltay EJ, Geleijnse JM, for the Alpha Omega Trial Group. n–3 fatty acids and cardiovascular events after myocardial infarction. N Engl J Med 2010;363:2015–26. http://dx.doi.org/10.1056/NEJMoa1003603
- GISSI-HF Investigators. Effect of rosuvastatin in patients with chronic heart failure (the GISSI-HF trial): a randomised, double-blind, placebo-controlled trial. Lancet 2008;372:1223. http://dx.doi.org/10.1016/S0140-6736(08)61239-8
- Cowie MR, Cure S, Bianic F, McGuire A, Goodall G, Tavazzi L. Cost-effectiveness of highly purified omega-3 polyunsaturated fatty acid ethyl esters in the treatment of chronic heart failure: results of Markov modelling in a UK setting. Eur J Heart Fail 2011;13:681–9. http://dx.doi.org/10.1093/eurjhf/hfr023
- Hooper L, Harrison RA, Summerbell CD et al. Omega 3 fatty acids for prevention and treatment of cardiovascular disease. Cochrane Database Syst Rev 2009. http://summaries.cochrane.org/CD003177
- NHS Business Services Authority, Prescription Services 2011. Available from: http://www.nhsbsa.nhs.uk/PrescriptionServices/Documents/PPDPrescribingAnalysisCharts/Cardio_Nov_2011_National.pdf [accessed 9th January 2012].
- Kromhout D, Yasuda S, Geleijnse JM, Shimokawa H. Fish oil and omega-3 fatty acids in cardiovascular disease: do they really work? Eur Heart J 2012;33:436–43. http://dx.doi.org/10.1093/eurheartj/ehr362
- Department of Health. National Diet and Nutrition Survey: headline results from years 1 and 2 (combined) of the rolling programme 2008/9 – 2009/10. Available from: http://www.dh.gov.uk/en/Publicationsandstatistics/Publications/PublicationsStatistics/DH_128166 [accessed 6th February 2012].
